Agencija za lekove i medicinska sredstva Srbije Agencija za lekove i ...
Agencija za lekove i medicinska sredstva Srbije Agencija za lekove i ...
Agencija za lekove i medicinska sredstva Srbije Agencija za lekove i ...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
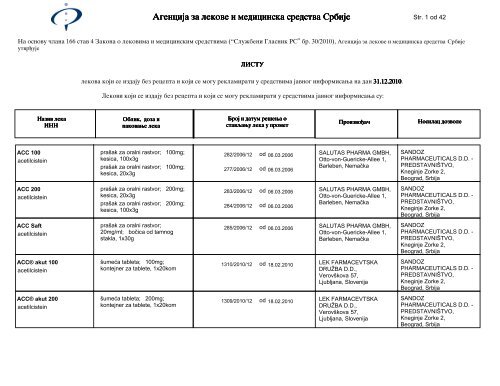
Str. 1 od 42<br />
Na osnovu ~lana 166 stav 4 Zakona o lekovima i medicinskim sredstvima (“Slu`beni Glasnik RS” br. 30/2010), <strong>Agencija</strong> <strong>za</strong> <strong>lekove</strong> i <strong>medicinska</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
utvr|uje<br />
ACC 100<br />
acetilcistein<br />
ACC 200<br />
acetilcistein<br />
ACC Saft<br />
acetilcistein<br />
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
ACC® akut 100<br />
acetilcistein<br />
ACC® akut 200<br />
acetilcistein<br />
LISTU<br />
LISTU<br />
lekova koji se izdaju bez recepta i koji se mogu reklamirati u sredstvima javnog informisawa na dan 31.12.2010 31.12.2010. 31.12.2010 31.12.2010<br />
Lekovi koji se izdaju bez recepta i koji se mogu reklamirati u sredstvima javnog informisawa su:<br />
Oblik Oblik, Oblik , do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe leka<br />
leka<br />
pra{ak <strong>za</strong> oralni rastvor; 100mg;<br />
kesica, 100x3g<br />
pra{ak <strong>za</strong> oralni rastvor; 100mg;<br />
kesica, 20x3g<br />
pra{ak <strong>za</strong> oralni rastvor; 200mg;<br />
kesica, 20x3g<br />
pra{ak <strong>za</strong> oralni rastvor; 200mg;<br />
kesica, 100x3g<br />
pra{ak <strong>za</strong> oralni rastvor;<br />
20mg/ml; bo~ica od tamnog<br />
stakla, 1x30g<br />
{ume}a tableta; 100mg;<br />
kontejner <strong>za</strong> tablete, 1x20kom<br />
{ume}a tableta; 200mg;<br />
kontejner <strong>za</strong> tablete, 1x20kom<br />
Broj Broj i i i datum datum datum re{ewa re{ewa oo<br />
o o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
282/2006/12<br />
277/2006/12<br />
283/2006/12<br />
284/2006/12<br />
285/2006/12<br />
1310/2010/12<br />
1309/2010/12<br />
od 06.03.2006<br />
od 06.03.2006<br />
od 06.03.2006<br />
od 06.03.2006<br />
od 06.03.2006<br />
od 18.02.2010<br />
od<br />
18.02.2010<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
SALUTAS PHARMA GMBH,<br />
Otto-von-Guericke-Allee 1,<br />
Barleben, Nema~ka<br />
SALUTAS PHARMA GMBH,<br />
Otto-von-Guericke-Allee 1,<br />
Barleben, Nema~ka<br />
SALUTAS PHARMA GMBH,<br />
Otto-von-Guericke-Allee 1,<br />
Barleben, Nema~ka<br />
LEK FARMACEVTSKA<br />
DRU@BA D.D.,<br />
Verov{kova 57,<br />
Ljubljana, Slovenija<br />
LEK FARMACEVTSKA<br />
DRU@BA D.D.,<br />
Verov{kova 57,<br />
Ljubljana, Slovenija<br />
SANDOZ<br />
PHARMACEUTICALS D.D. -<br />
PREDSTAVNI[TVO,<br />
Kneginje Zorke 2,<br />
Beograd, Srbija<br />
SANDOZ<br />
PHARMACEUTICALS D.D. -<br />
PREDSTAVNI[TVO,<br />
Kneginje Zorke 2,<br />
Beograd, Srbija<br />
SANDOZ<br />
PHARMACEUTICALS D.D. -<br />
PREDSTAVNI[TVO,<br />
Kneginje Zorke 2,<br />
Beograd, Srbija<br />
SANDOZ<br />
PHARMACEUTICALS D.D. -<br />
PREDSTAVNI[TVO,<br />
Kneginje Zorke 2,<br />
Beograd, Srbija<br />
SANDOZ<br />
PHARMACEUTICALS D.D. -<br />
PREDSTAVNI[TVO,<br />
Kneginje Zorke 2,<br />
Beograd, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
ACC® akut 600<br />
acetilcistein<br />
Acetisal pH 8®<br />
acetilsalicilna kiselina<br />
Aflubin®<br />
gentiana lutea trit. D1,<br />
aconitum napellus trit. D6,<br />
bryonia trit. D6, ferrum<br />
phosphoricum trit. D12,<br />
acidum sarcolacticum trit.<br />
D12<br />
Aknefug® Oxid Wash<br />
benzoil-peroksid<br />
Alfogel®<br />
aluminijum fosfat<br />
alli®<br />
orlistat<br />
alli®<br />
orlistat<br />
Oblik Oblik, Oblik , do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe leka<br />
leka<br />
{ume}a tableta; 600mg;<br />
kesica, 10x1kom<br />
film tableta; 500mg;<br />
blister, 2x10kom<br />
tableta; 3.6mg+37.2mg+<br />
37.2mg+37.2mg+37.2mg;<br />
blister, 1x12kom<br />
tableta; 3.6mg+37.2mg+<br />
37.2mg+37.2mg+37.2mg;<br />
blister, 4x12kom<br />
oralne kapi, rastvor; (1ml+10ml+<br />
10ml+10ml+10ml)/100ml;<br />
bo~ica sa kapaljkom, 1x20ml<br />
oralne kapi, rastvor; (1ml+10ml+<br />
10ml+10ml+10ml)/100ml;<br />
bo~ica sa kapaljkom, 1x50ml<br />
suspenzija <strong>za</strong> ko`u; 4%;<br />
tuba, 1x100g<br />
oralni gel; 8.8g; kesica, 20x16g<br />
kapsula, tvrda; 60mg;<br />
bo~ica plasti~na, 1x42kom<br />
kapsula, tvrda; 60mg;<br />
bo~ica plasti~na, 1x84kom<br />
kapsula, tvrda; 60mg;<br />
bo~ica plasti~na, 1x42kom<br />
kapsula, tvrda; 60mg;<br />
bo~ica plasti~na, 1x84kom<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
1311/2010/12<br />
3331/2009/12<br />
6295/2010/12<br />
6296/2010/12<br />
6299/2010/12<br />
6298/2010/12<br />
2148/2010/12<br />
108/2006/12<br />
2877/2010/12<br />
2878/2010/12<br />
2876/2010/12<br />
2879/2010/12<br />
od 18.02.2010<br />
od 15.06.2009<br />
od 03.11.2010<br />
od 03.11.2010<br />
od 03.11.2010<br />
od 03.11.2010<br />
od 22.03.2010<br />
od 02.02.2006<br />
od 06.05.2010<br />
od 06.05.2010<br />
od 06.05.2010<br />
od<br />
06.05.2010<br />
Str. 2 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
LEK FARMACEVTSKA<br />
DRU@BA D.D.,<br />
Verov{kova 57,<br />
Ljubljana, Slovenija<br />
GALENIKA AD, Batajni~ki<br />
drum b.b., Beograd, Srbija<br />
RICHARD BITTNER AG.,<br />
Ossiacher Strasse 7,<br />
Feldkirchen, Austrija<br />
DR. AUGUST WOLFF GMBH<br />
& CO. ARZNEIMITTEL,<br />
Sudbrackstrasse 56,<br />
Bielefeld, Nema~ka<br />
GALENIKA AD, Batajni~ki<br />
drum b.b., Beograd, Srbija<br />
CATALENT UK PACKAGING<br />
LIMITED, Sedge Close,<br />
Great Oakley, Corby,<br />
Northamptonshire,<br />
Velika Britanija<br />
FAMAR S.A.,<br />
49 KM Athens- Lamia,<br />
Avlona, Gr~ka<br />
SANDOZ<br />
PHARMACEUTICALS D.D. -<br />
PREDSTAVNI[TVO,<br />
Kneginje Zorke 2,<br />
Beograd, Srbija<br />
GALENIKA AD, Batajni~ki<br />
drum b.b., Beograd, Srbija<br />
FARMALOGIST D.O.O.,<br />
Bulevar Vojvode<br />
Mi{i}a 25-27,<br />
Beograd, Srbija<br />
EUROFARM D.O.O.,<br />
Bo{ka Petrovi}a 3a,<br />
Beograd, Srbija<br />
GALENIKA AD, Batajni~ki<br />
drum b.b., Beograd, Srbija<br />
GLAXOSMITHKLINE<br />
EXPORT LIMITED -<br />
PREDSTAVNI[TVO,<br />
Omladinskih brigada 88,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
GLAXOSMITHKLINE<br />
EXPORT LIMITED -<br />
PREDSTAVNI[TVO,<br />
Omladinskih brigada 88,<br />
Beograd - Novi Beograd,<br />
Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Altacid®<br />
aluminijum-hidroksid,<br />
magnezijum-oksid<br />
Ambroksol<br />
ambroksol<br />
Anbol®<br />
acetilsalicilna kiselina<br />
Andol®<br />
acetilsalicilna kiselina<br />
Andol® 100<br />
acetilsalicilna kiselina<br />
Antisept D®<br />
povidon<br />
Aqua pro injectione<br />
voda <strong>za</strong> injekcije<br />
Artelac®<br />
hipromelo<strong>za</strong><br />
Oblik Oblik, Oblik , do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe leka<br />
leka<br />
tableta <strong>za</strong> `vakanje;<br />
333.3mg+158.4mg;<br />
blister, 2x8kom<br />
oralna suspenzija;<br />
2249.5mg/5ml+425mg/5ml;<br />
bo~ica, 1x60ml<br />
{ume}a tableta; 30mg;<br />
fiola, 2x10kom<br />
{ume}a tableta; 60mg;<br />
fiola, 1x10kom<br />
sirup; 15mg/5ml;<br />
bo~ica, 1x100ml<br />
tableta; 300mg; strip, 3x6kom<br />
tableta; 300mg; strip, 2x10kom<br />
tableta; 300mg;<br />
blister, 2x10kom<br />
tableta; 100mg;<br />
blister, 2x10kom<br />
sprej <strong>za</strong> ko`u, rastvor; 10%;<br />
bo~ica plasti~na, 1x100ml<br />
rastvara~ <strong>za</strong> parenteralnu<br />
upotrebu; 2ml; ampula, 50x2ml<br />
rastvara~ <strong>za</strong> parenteralnu<br />
upotrebu; 5ml; ampula, 50x5ml<br />
rastvara~ <strong>za</strong> parenteralnu<br />
upotrebu; 10ml; ampula,<br />
50x10ml<br />
kapi <strong>za</strong> o~i, rastvor; 3.2mg/ml;<br />
bo~ica sa kapaljkom, 1x10ml<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
2106/2006/12<br />
88/2007/12<br />
2552/2007/12<br />
2528/2007/12<br />
2549/2007/12<br />
4470/2010/12<br />
2259/2008/12<br />
1134/2009/12<br />
1133/2009/12<br />
7398/2009/12<br />
63/2006/12<br />
64/2006/12<br />
2452/2009/12<br />
2297/2007/12<br />
od 14.11.2006<br />
od 11.01.2007<br />
od 22.10.2007<br />
od 22.10.2007<br />
od 22.10.2007<br />
od 08.10.2010<br />
od 17.07.2008<br />
od 01.04.2009<br />
od 01.04.2009<br />
od 24.11.2009<br />
od 20.01.2006<br />
od 20.01.2006<br />
od 30.04.2009<br />
od<br />
09.10.2007<br />
Str. 3 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
BOSNALIJEK D.D.,<br />
Juki}eva 53, Sarajevo,<br />
Bosna i Hercegovina<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
GALENIKA AD, Batajni~ki<br />
drum b.b., Beograd, Srbija<br />
PLIVA HRVATSKA D.O.O.,<br />
Prilaz baruna Filipovi}a 25,<br />
Zagreb, Hrvatska<br />
PLIVA HRVATSKA D.O.O.,<br />
Prilaz baruna Filipovi}a 25,<br />
Zagreb, Hrvatska<br />
BOSNALIJEK D.D.,<br />
Juki}eva 53, Sarajevo,<br />
Bosna i Hercegovina<br />
JUGOREMEDIJA A.D.<br />
ZRENJANIN,<br />
Pan~eva~ka bb,<br />
Zrenjanin, Srbija<br />
DR. GERHARD MANN,<br />
CHEM. - PHARM. FABRIK<br />
GMBH, Brunsbutteler Damm<br />
165 - 173, Berlin, Nema~ka<br />
BOSNALIJEK D.D. -<br />
PREDSTAVNI[TVO,<br />
Cara Nikolaja II 61/B,<br />
Beograd, Srbija<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
GALENIKA AD, Batajni~ki<br />
drum b.b., Beograd, Srbija<br />
UNIMED D.O.O.,<br />
De~anska 12/II,<br />
Beograd-Stari grad, Srbija<br />
UNIMED D.O.O.,<br />
De~anska 12/II,<br />
Beograd-Stari grad, Srbija<br />
BOSNALIJEK D.D. -<br />
PREDSTAVNI[TVO,<br />
Cara Nikolaja II 61/B,<br />
Beograd, Srbija II 61/B,<br />
Beograd,<br />
JUGOREMEDIJA A.D.<br />
ZRENJANIN,<br />
Pan~eva~ka bb,<br />
Zrenjanin, Srbija<br />
MEDILENS D.O.O.,<br />
Savski trg 9,<br />
Beograd, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Artelac® EDO®<br />
hipromelo<strong>za</strong><br />
Aspegic®<br />
lizin acetilsalicilat<br />
Aspegic® <strong>za</strong> odrasle<br />
lizin acetilsalicilat<br />
Aspirin® 500<br />
acetilsalicilna kiselina<br />
Aspirin® Complex<br />
pseudoefedrin,<br />
acetilsalicilna kiselina<br />
Aspirin® Migran<br />
acetilsalicilna kiselina<br />
Aspirin® plus C<br />
acetilsalicilna kiselina,<br />
askorbinska kiselina<br />
Oblik Oblik, Oblik Oblik,<br />
, do<strong>za</strong> do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe leka leka<br />
leka<br />
kapi <strong>za</strong> o~i, rastvor; 3.2mg/ml;<br />
kontejner jednodozni, 30x0.6ml<br />
kapi <strong>za</strong> o~i, rastvor; 3.2mg/ml;<br />
kontejner jednodozni, 10x0.6ml<br />
kapi <strong>za</strong> o~i, rastvor; 3.2mg/ml;<br />
kontejner jednodozni, 60x0.6ml<br />
pra{ak <strong>za</strong> oralni rastvor; 500mg;<br />
kesica, 20x1kom<br />
pra{ak <strong>za</strong> oralni rastvor;<br />
1000mg; kesica, 20x1kom<br />
tableta; 500mg;<br />
blister, 2x10kom<br />
granule <strong>za</strong> oralnu suspenziju;<br />
30mg+500mg; kesica, 10x1kom<br />
{ume}a tableta; 500mg;<br />
blister, 2x6kom<br />
{ume}a tableta; 400mg+240mg;<br />
strip, 5x2kom<br />
{ume}a tableta; 400mg+240mg;<br />
strip, 10x2kom<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
2299/2007/12<br />
2298/2007/12<br />
2300/2007/12<br />
1998/2008/12<br />
1997/2008/12<br />
199/2007/12<br />
2132/2008/12<br />
2283/2006/12<br />
200/2007/12<br />
1709/2010/12<br />
od 10.10.2007<br />
od<br />
od<br />
od<br />
od<br />
od<br />
od<br />
od<br />
od<br />
10.10.2007<br />
10.10.2007<br />
10.06.2008<br />
10.06.2008<br />
16.01.2007<br />
20.06.2008<br />
11.12.2006<br />
16.01.2007<br />
od<br />
15.03.2010<br />
Str. 4 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
DR. GERHARD MANN,<br />
CHEM. - PHARM. FABRIK<br />
GMBH, Brunsbutteler Damm<br />
165 - 173, Berlin, Nema~ka<br />
SANOFI WINTHROP<br />
INDUSTRIE, 196 avenue du<br />
Marechal Juin, Amilly,<br />
Francuska<br />
SANOFI WINTHROP<br />
INDUSTRIE, 196 avenue du<br />
Marechal Juin, Amilly,<br />
Francuska<br />
BAYER BITTERFELD GMBH,<br />
OT Greppin, Salegaster<br />
Chaussee 1, Bitterfeld-<br />
Wolfen, Nema~ka<br />
BAYER BITTERFELD GMBH,<br />
OT Greppin, Salegaster<br />
Chaussee 1, Bitterfeld-<br />
Wolfen, Nema~ka<br />
BAYER BITTERFELD GMBH,<br />
OT Greppin, Salegaster<br />
Chaussee 1, Bitterfeld-<br />
Wolfen, Nema~ka<br />
BAYER BITTERFELD GMBH,<br />
OT Greppin, Salegaster<br />
Chaussee 1, Bitterfeld-<br />
Wolfen, Nema~ka<br />
MEDILENS D.O.O.,<br />
Savski trg 9,<br />
Beograd, Srbija<br />
SANOFI-AVENTIS D.O.O.,<br />
Vladimira Popovi}a 40/IV,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
SANOFI-AVENTIS D.O.O.,<br />
Vladimira Popovi}a 40/IV,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
BAYER D.O.O., Omladinskih<br />
brigada 88b, Beograd - Novi<br />
Beograd, Srbija<br />
BAYER D.O.O., Omladinskih<br />
brigada 88b, Beograd - Novi<br />
Beograd, Srbija<br />
BAYER D.O.O., Omladinskih<br />
brigada 88b, Beograd - Novi<br />
Beograd, Srbija<br />
BAYER D.O.O., Omladinskih<br />
brigada 88b, Beograd - Novi<br />
Beograd, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Aspirin® protect<br />
acetilsalicilna kiselina<br />
Aviomarin®<br />
dimenhidrinat<br />
B-Complex<br />
tiamin, riboflavin, piridoksin,<br />
nikotinamid, kalcijumpantotenat,<br />
cijanokobalamin,<br />
paba<br />
Bekunis®<br />
plod tinevelijske sene<br />
(Sennae fructus angustifoliae),<br />
suvi vodeni ekstakt<br />
Bekunis®<br />
list tinevelijske sene<br />
Bekunis®<br />
list tinevelijske sene<br />
Bekunis®<br />
plod aleksandrijske i<br />
tinevelijske sene, suvi<br />
vodeni ekstrakt<br />
Ben-Gay® Sport Balm<br />
metilsalicilat, mentol<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
gastrorezistentna tableta;<br />
100mg; blister, 3x10kom<br />
gastrorezistentna tableta;<br />
300mg; blister, 3x10kom<br />
tableta; 50mg; blister, 1x5kom<br />
oblo`ena tableta; 4mg+5mg+<br />
2mg+25mg+5mg+0.001mg+<br />
20mg; kutija, 1x1000kom<br />
oblo`ena tableta; 4mg+5mg+<br />
2mg+25mg+5mg+0.001mg+<br />
20mg; blister, 2x15kom<br />
granule; (4mg+2mg+2mg+<br />
20mg+2mg)/5g; folija, 1x70g<br />
gastrorezistentna oblo`ena tableta;<br />
20mg; kontejner, 1x45kom<br />
gastrorezistentna oblo`ena tableta;<br />
20mg; kontejner, 1x100kom<br />
biljni ~aj; 25mg/g; kutija, 1x80g<br />
biljni ~aj; 25mg/g;<br />
kesica, 12x1g<br />
instant biljni ~aj; 30.8-<br />
51.3g/100g; tegla, 1x17.6g<br />
krem; 28%+10%; tuba, 1x35g<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
2781/2008/12<br />
2782/2008/12<br />
4275/2010/12<br />
1197/2007/12<br />
1196/2007/12<br />
1198/2007/12<br />
2781/2010/12<br />
2782/2010/12<br />
3718/2010/12<br />
4199/2010/12<br />
2963/2010/12<br />
575/2007/12<br />
od 26.08.2008<br />
od 26.08.2008<br />
od 16.09.2010<br />
od 29.05.2007<br />
od 29.05.2007<br />
od 29.05.2007<br />
od 27.04.2010<br />
od<br />
od<br />
od<br />
od<br />
27.04.2010<br />
16.07.2010<br />
09.09.2010<br />
10.05.2010<br />
od<br />
01.03.2007<br />
Str. 5 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
BAYER BITTERFELD GMBH,<br />
OT Greppin, Salegaster<br />
Chaussee 1, Bitterfeld-<br />
Wolfen, Nema~ka<br />
PLIVA HRVATSKA D.O.O.,<br />
Prilaz baruna Filipovi}a 25,<br />
Zagreb, Hrvatska<br />
KRKA, TOVARNA ZDRAVIL,<br />
D.D., [marje{ka cesta 6,<br />
Novo Mesto, Slovenija<br />
ROHA ARZNEIMITTEL<br />
GMBH, Rockwinkeler<br />
Heerstrase 100,<br />
Bremen, Nema~ka<br />
ROHA ARZNEIMITTEL<br />
GMBH, Rockwinkeler Heerstrase<br />
100, Bremen, Nema~ka<br />
ZDRAVLJE A.D.,<br />
Vlajkova 199,<br />
Leskovac, Srbija<br />
ZDRAVLJE A.D.,<br />
Vlajkova 199,<br />
Leskovac, Srbija<br />
PFIZER ILACLARI LTD. STI.,<br />
Muallim Naci Cad. No.55,<br />
Ortakoy, Istanbul, Turska<br />
BAYER D.O.O., Omladinskih<br />
brigada 88b, Beograd - Novi<br />
Beograd, Srbija<br />
UNIMED D.O.O.,<br />
De~anska 12/II,<br />
Beograd-Stari grad, Srbija<br />
KRKA FARMA D.O.O.,<br />
Jurija Gagarina 26/V/II,<br />
Beograd, Srbija<br />
ACTAVIS D.O.O., \or|a<br />
Stanojevi}a 12, Beograd -<br />
Novi Beograd, Srbija<br />
ACTAVIS D.O.O., \or|a<br />
Stanojevi}a 12, Beograd -<br />
Novi Beograd, Srbija<br />
ZDRAVLJE A.D.,<br />
Vlajkova 199,<br />
Leskovac, Srbija<br />
ZDRAVLJE A.D.,<br />
Vlajkova 199,<br />
Leskovac, Srbija<br />
JOHNSON & JOHNSON S.E.<br />
INC.- PREDSTAVNI[TVO,<br />
Omladinskih brigada 88 b,<br />
Beograd - Novi Beograd,<br />
Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
BENGAY® Greaseless<br />
metilsalicilat, mentol<br />
Bepanthen®<br />
dekspantenol<br />
Bepanthen® Plus<br />
dekspantenol, hlorheksidin<br />
Berlithion® 300<br />
tioktinska kiselina<br />
Berlithion® 600<br />
tioktinska kiselina<br />
Betadine®<br />
povidon<br />
Bilobil®<br />
ginko (Ginkgo biloba), suvi<br />
ekstrakt lista<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
krem; 15%+10%; tuba, 1x50g<br />
krem; 5%; tuba, 1x30g<br />
mast; 5%; tuba, 1x30g<br />
krem; 5%+0.5%; tuba, 1x30g<br />
kapsula, meka; 300mg;<br />
blister, 1x15kom<br />
kapsula, meka; 300mg;<br />
blister, 2x15kom<br />
kapsula, meka; 600mg;<br />
blister, 2x15kom<br />
kapsula, meka; 600mg;<br />
blister, 1x15kom<br />
vagitorija; 200mg; strip, 2x7kom<br />
rastvor <strong>za</strong> ko`u; 10%;<br />
bo~ica, 1x100ml<br />
rastvor <strong>za</strong> grgljanje; 1%;<br />
bo~ica, 1x100ml<br />
kapsula, tvrda; 40mg;<br />
blister, 2x10kom<br />
kapsula, tvrda; 40mg;<br />
blister, 6x10kom<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
3963/2009/12<br />
2478/2008/12<br />
2479/2008/12<br />
2480/2008/12<br />
3109/2008/12<br />
3112/2008/12<br />
3113/2008/12<br />
3114/2008/12<br />
314/2008/12<br />
316/2008/12<br />
1216/2008/12<br />
2313/2006/12<br />
2314/2006/12<br />
od 24.07.2009<br />
od 04.08.2008<br />
od 04.08.2008<br />
od 04.08.2008<br />
od 10.09.2008<br />
od 10.09.2008<br />
od<br />
od<br />
od<br />
od<br />
od<br />
10.09.2008<br />
10.09.2008<br />
06.02.2008<br />
06.02.2008<br />
26.03.2008<br />
od 12.12.2006<br />
od<br />
12.12.2006<br />
Str. 6 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
JANSSEN-CILAG,<br />
Domaine de Maigremont,<br />
Val-De-Reuil, Francuska<br />
GP GRENZACH<br />
PRODUKTIONS GMBH, Emil-<br />
Barell-Strasse 7, Gren<strong>za</strong>ch-<br />
Wyhlen, Nema~ka<br />
GP GRENZACH<br />
PRODUKTIONS GMBH, Emil-<br />
Barell-Strasse 7, Gren<strong>za</strong>ch-<br />
Wyhlen, Nema~ka<br />
BERLIN-CHEMIE AG<br />
(MENARINI GROUP),<br />
Glienicker Weg 125, Berlin,<br />
Nema~ka<br />
BERLIN-CHEMIE AG<br />
(MENARINI GROUP),<br />
Glienicker Weg 125, Berlin,<br />
Nema~ka<br />
ALKALOID AD, Bul.<br />
Aleksandar Makedonski 12,<br />
Skopje, Makedonija<br />
u saradnji sa<br />
MUNDIPHARMA AG,<br />
St. Alban-Rheinweg 74,<br />
Bazel, [vajcarska<br />
KRKA, TOVARNA ZDRAVIL,<br />
D.D., [marje{ka cesta 6,<br />
Novo Mesto, Slovenija<br />
JOHNSON & JOHNSON S.E.<br />
INC.- PREDSTAVNI[TVO,<br />
Omladinskih brigada 88 b,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
BAYER D.O.O., Omladinskih<br />
brigada 88b, Beograd - Novi<br />
Beograd, Srbija<br />
BAYER D.O.O., Omladinskih<br />
brigada 88b, Beograd - Novi<br />
Beograd, Srbija<br />
BERLIN-CHEMIE AG<br />
(MENARINI GROUP) -<br />
PREDSTAVNI[TVO,<br />
Prilepska 1,<br />
Beograd, Srbija<br />
BERLIN-CHEMIE AG<br />
(MENARINI GROUP) -<br />
PREDSTAVNI[TVO,<br />
Prilepska 1,<br />
Beograd, Srbija<br />
ALKALOID D.O.O.,<br />
Prahovska 3, Beograd -<br />
Savski Venac, Srbija<br />
KRKA FARMA D.O.O.,<br />
Jurija Gagarina 26/V/II,<br />
Beograd, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Bilobil® forte<br />
ginko (Ginkgo biloba), suvi<br />
ekstrakt lista<br />
Bronchicum® eliksir S<br />
timijan (Thymus vulgaris),<br />
te~ni ekstrakt, jagor~evina<br />
(Primula veris), te~ni<br />
ekstrakt<br />
Bronchicum® pastile<br />
timijan (Thymus vulgaris),<br />
te~ni ekstrakt herbe<br />
Bronchicum® sirup S<br />
timijan (Thymus vulgaris),<br />
te~ni ekstrakt herbe<br />
Brufen®<br />
ibuprofen<br />
Bubil®<br />
piretrin<br />
Ca-C 1000 Sandoz®<br />
askorbinska kiselina,<br />
kalcijum-karbonat, kalcijumlaktat-glukonat<br />
Caffebol<br />
paracetamol, kofein, kodein<br />
Caffebol<br />
paracetamol, kofein, kodein<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
kapsula, tvrda; 80mg;<br />
blister, 2x10kom<br />
kapsula, tvrda; 80mg;<br />
blister, 6x10kom<br />
oralni rastvor; 50mg/g+25mg/g;<br />
bo~ica, 1x130g<br />
komprimovana lozenga; 100mg;<br />
blister, 2x10kom<br />
sirup; 150mg/ml;<br />
bo~ica, 1x100ml<br />
oblo`ena tableta; 200mg;<br />
blister, 3x10kom<br />
krem; 10%; tuba, 1x50g<br />
{ampon; 720mg/60ml; bo~ica<br />
staklena, 1x60ml<br />
{ume}a tableta;<br />
1000mg+327mg+1000mg; fiola,<br />
1x10kom<br />
tableta; 460mg+50mg+10mg;<br />
folija, 50x10kom<br />
tableta; 460mg+50mg+10mg;<br />
blister, 2x10kom<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
636/2006/12<br />
635/2006/12<br />
1480/2007/12<br />
2877/2007/12<br />
1481/2007/12<br />
226/2008/12<br />
4347/2010/12<br />
3254/2010/12<br />
3658/2008/12<br />
3316/2010/12<br />
2094/2010/12<br />
od 17.05.2006<br />
od 17.05.2006<br />
od 10.07.2007<br />
od 21.11.2007<br />
od 10.07.2007<br />
od 31.01.2008<br />
od 27.09.2010<br />
od 04.06.2010<br />
od 15.10.2008<br />
od 07.06.2010<br />
od<br />
16.03.2010<br />
Str. 7 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
KRKA, TOVARNA ZDRAVIL,<br />
D.D., [marje{ka cesta 6,<br />
Novo Mesto, Slovenija<br />
A. NATTERMANN & CIE.<br />
GMBH, Nattermannallee 1,<br />
Keln, Nema~ka<br />
A. NATTERMANN & CIE.<br />
GMBH, Nattermannallee 1,<br />
Keln, Nema~ka<br />
A. NATTERMANN & CIE.<br />
GMBH, Nattermannallee 1,<br />
Keln, Nema~ka<br />
GALENIKA AD, Batajni~ki<br />
drum b.b., Beograd, Srbija u<br />
saradnji sa ABBOTT GMBH &<br />
CO.KG, Max Planck Ring 2,<br />
Wiesbaden, Nema~ka<br />
JUGOREMEDIJA A.D.<br />
ZRENJANIN, Pan~eva~ka bb,<br />
Zrenjanin, Srbija<br />
FAMAR FRANCE, 1, Avenue<br />
du Champs de Mars, Orleans<br />
Cedex 2, Francuska <strong>za</strong><br />
NOVARTIS CONSUMER<br />
HEALTH SA, Route de<br />
l'Etraz, Nyon, [vajcarska<br />
HEMOFARM AD, Beogradski<br />
put bb, Vr{ac, Srbija<br />
HEMOFARM D.O.O. [ABAC,<br />
Hajduk Veljkova bb,<br />
[abac, Srbija<br />
KRKA FARMA D.O.O.,<br />
Jurija Gagarina 26/V/II,<br />
Beograd, Srbija<br />
SANOFI-AVENTIS D.O.O.,<br />
Vladimira Popovi}a 40/IV,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
SANOFI-AVENTIS D.O.O.,<br />
Vladimira Popovi}a 40/IV,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
SANOFI-AVENTIS D.O.O.,<br />
Vladimira Popovi}a 40/IV,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
GALENIKA AD,<br />
Batajni~ki drum b.b.,<br />
Beograd, Srbija<br />
JUGOREMEDIJA A.D.<br />
ZRENJANIN, Pan~eva~ka bb,<br />
Zrenjanin, Srbija<br />
MEDIS INTERNATIONAL<br />
D.O.O., Bulevar Mihaila<br />
Pupina 10d/1 pp7, Beograd -<br />
Novi Beograd, Srbija<br />
HEMOFARM AD, Beogradski<br />
put bb, Vr{ac, Srbija<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Caffetin cold®<br />
paracetamol, pseudoefedrin,<br />
dekstrometorfan,<br />
askorbinska kiselina<br />
Caffetin menstrual®<br />
ibuprofen<br />
Caffetin®<br />
paracetamol, kofein, kodein,<br />
propifenazon<br />
Calcium-Sandoz forte® 500<br />
kalcijum-laktat-glukonat,<br />
kalcijum-karbonat<br />
CalciumvitaC®<br />
askorbinska kiselina,<br />
kalcijum (kalcijum-laktatglukonat,<br />
kalcijum-karbonat)<br />
Calgel®<br />
lidokain, cetilpiridinijumhlorid<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
film tableta;<br />
500mg+30mg+15mg+60mg;<br />
blister, 1x10kom<br />
film tableta; 200mg;<br />
blister, 1x10kom<br />
tableta;<br />
250mg+50mg+10mg+210mg;<br />
strip, 1x6kom<br />
tableta;<br />
250mg+50mg+10mg+210mg;<br />
strip, 2x6kom<br />
tableta;<br />
250mg+50mg+10mg+210mg;<br />
strip, 50x10kom<br />
tableta;<br />
250mg+50mg+10mg+210mg;<br />
strip, 1x10kom<br />
{ume}a tableta;<br />
1132mg+875mg; fiola, 1x10kom<br />
{ume}a tableta;<br />
1132mg+875mg; fiola, 1x20kom<br />
{ume}a tableta; 500mg+260mg;<br />
fiola, 1x10kom<br />
gel <strong>za</strong> desni; 0.33%+0.1%;<br />
tuba, 1x10g<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
2170/2006/12<br />
3879/2009/12<br />
1556/2006/12<br />
1557/2006/12<br />
1567/2006/12<br />
1568/2006/12<br />
2516/2008/12<br />
2231/2008/12<br />
413/2006/12<br />
3757/2009/12<br />
od 24.11.2006<br />
od 15.07.2009<br />
od 31.08.2006<br />
od 31.08.2006<br />
od 05.09.2006<br />
od 05.09.2006<br />
od 07.08.2008<br />
od 25.08.2008<br />
od 05.04.2006<br />
od<br />
08.07.2009<br />
Str. 8 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
ALKALOID AD, Bul.<br />
Aleksandar Makedonski 12,<br />
Skopje, Makedonija<br />
ALKALOID AD, Bul.<br />
Aleksandar Makedonski 12,<br />
Skopje, Makedonija<br />
ALKALOID AD, Bul.<br />
Aleksandar Makedonski 12,<br />
Skopje, Makedonija<br />
FAMAR FRANCE, 1, Avenue<br />
du Champs de Mars, Orleans<br />
Cedex 2, Francuska <strong>za</strong><br />
NOVARTIS CONSUMER<br />
HEALTH SA, Route de<br />
l'Etraz, Nyon, [vajcarska<br />
KRKA, TOVARNA ZDRAVIL,<br />
D.D., [marje{ka cesta 6,<br />
Novo Mesto, Slovenija<br />
GLAXOSMITHKLINE<br />
PHARMACEUTICALS S.A.,<br />
Ul. Grunwaldzka 189,<br />
Poznan, Poljska<br />
ALKALOID D.O.O.,<br />
Prahovska 3, Beograd -<br />
Savski Venac, Srbija<br />
ALKALOID D.O.O.,<br />
Prahovska 3, Beograd -<br />
Savski Venac, Srbija<br />
ALKALOID D.O.O.,<br />
Prahovska 3, Beograd -<br />
Savski Venac, Srbija<br />
MEDIS INTERNATIONAL<br />
D.O.O., Bulevar Mihaila<br />
Pupina 10d/1 pp7, Beograd -<br />
Novi Beograd, Srbija<br />
KRKA FARMA D.O.O.,<br />
Jurija Gagarina 26/V/II,<br />
Beograd, Srbija<br />
GLAXOSMITHKLINE<br />
EXPORT LIMITED -<br />
PREDSTAVNI[TVO,<br />
Omladinskih brigada 88,<br />
Beograd - Novi Beograd,<br />
Srbija
Canesten®<br />
klotrimazol<br />
Canesten®<br />
klotrimazol<br />
Canesten®<br />
klotrimazol<br />
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Canesten® 1<br />
klotrimazol<br />
Canesten® 3<br />
klotrimazol<br />
Canesten® 3<br />
klotrimazol<br />
Cardiopirin®<br />
acetilsalicilna kiselina<br />
Cefabene®<br />
stabljike razvodnika, te~ni<br />
ekstrakt<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
pra{ak <strong>za</strong> ko`u; 1%; bo~ica<br />
plasti~na, 1x30g<br />
krem; 1%; tuba, 1x20g<br />
rastvor <strong>za</strong> ko`u; 1%;<br />
bo~ica sa kapaljkom, 1x20ml<br />
vaginalna tableta; 500mg;<br />
blister, 1x1kom<br />
vaginalna tableta; 200mg;<br />
blister, 1x3kom<br />
vaginalni krem; 2%;<br />
tuba, 1x20g<br />
gastrorezistentna tableta;<br />
100mg; blister, 3x10kom<br />
mast; 10%; tuba, 1x25g<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
68/2007/12<br />
66/2007/12<br />
1553/2007/12<br />
69/2007/12<br />
70/2007/12<br />
71/2007/12<br />
7188/2010/12<br />
2511/2010/12<br />
od 11.01.2007<br />
od 11.01.2007<br />
od 24.07.2007<br />
od 11.01.2007<br />
od 11.01.2007<br />
od 11.01.2007<br />
od<br />
29.12.2010<br />
od<br />
14.04.2010<br />
Str. 9 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
BAYER HEALTHCARE<br />
MANUFACTURING S.R.L.,<br />
Via delle Groane 126,<br />
Garbagnate, Milanese, Italija<br />
KERN PHARMA, S.L.,<br />
Poligono Industrial Colon II,<br />
Venus, 72, Terrassa<br />
(Barcelona), [panija<br />
KVP PHARMA UND<br />
VETERINÄR PRODUKTE<br />
GMBH, Projensdorfer Strasse<br />
324, Kiel, Nema~ka<br />
BAYER SCHERING<br />
PHARMA AG, 51368,<br />
Leverkusen, Nema~ka<br />
BAYER SCHERING<br />
PHARMA AG, 51368,<br />
Leverkusen, Nema~ka<br />
KERN PHARMA, S.L.,<br />
Poligono Industrial Colon II,<br />
Venus, 72, Terrassa<br />
(Barcelona), [panija<br />
G.L. PHARMA GMBH,<br />
Schlossplatz 1,<br />
Lannach, Austrija<br />
CEFAK KG,<br />
Ostbahnhofstraße 15,<br />
Kempten, Nema~ka<br />
BAYER D.O.O., Omladinskih<br />
brigada 88b, Beograd - Novi<br />
Beograd, Srbija<br />
BAYER D.O.O., Omladinskih<br />
brigada 88b, Beograd - Novi<br />
Beograd, Srbija<br />
BAYER D.O.O., Omladinskih<br />
brigada 88b, Beograd - Novi<br />
Beograd, Srbija<br />
BAYER D.O.O., Omladinskih<br />
brigada 88b, Beograd - Novi<br />
Beograd, Srbija<br />
BAYER D.O.O., Omladinskih<br />
brigada 88b, Beograd - Novi<br />
Beograd, Srbija<br />
BAYER D.O.O., Omladinskih<br />
brigada 88b, Beograd - Novi<br />
Beograd, Srbija<br />
PHARMASWISS D.O.O.,<br />
Vojvode Stepe 18,<br />
Beograd, Srbija<br />
MEDICO PHARM SERVIS,<br />
Save Kova~evi}a 113A,<br />
Beograd - Kalu|erica, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Cefagil®<br />
Turnera diffusa (Damiana),<br />
Ø D1<br />
Cefakliman<br />
rizom cimicifuge, suvi<br />
standardizovani ekstrakt<br />
Cefamadar<br />
Calotropis gigantea (Madar),<br />
triturat D4<br />
Cefan<br />
valerijana (Valeriana<br />
officinalis), suvi ekstrakt<br />
korena<br />
Cefanorm®<br />
plod konopljike, suvi<br />
ekstrakt<br />
Cefasabal<br />
testerasta palmica (Sabal<br />
serrulata), suvi ekstrakt ploda,<br />
zlatica (Solidago canadensis),<br />
suvi ekstrakt herbe, divlji kesten<br />
(Aesculus hippocastanum),<br />
suvi ekstrakt semena<br />
Cefasinu<br />
Chininum arsenicosum trit.<br />
D12, Cinnabaris trit. D4,<br />
Ferrum phosphoricum trit.<br />
D3, Mercurius solubilis<br />
Hahn. trit. D5<br />
Chymoral®<br />
tripsin, kombinacije<br />
Cinnabsin®<br />
Cinnabaris, triturat D3,<br />
Echinacea, triturat D1,<br />
Kalijum-dihromat, triturat<br />
D3, Hydrastis, triturat D3<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
tableta; 25mg; blister, 5x20kom<br />
kapsula, tvrda; 2.675mg;<br />
blister, 5x10kom<br />
tableta; 250mg;<br />
blister, 5x20kom<br />
oblo`ena tableta; 441.35mg;<br />
blister, 1x20kom<br />
oblo`ena tableta; 441.35mg;<br />
blister, 2x25kom<br />
kapsula, tvrda; 4mg;<br />
blister, 3x10kom<br />
tableta;<br />
1.25mg+3.75mg+6.25mg;<br />
blister, 5x20kom<br />
tableta;<br />
30mg+10mg+30mg+130mg;<br />
blister, 2x10kom<br />
gastrorezistentna tableta;<br />
100000Aj; blister, 2x12kom<br />
tableta;<br />
25mg+25mg+25mg+25mg;<br />
blister, 5x20kom<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
798/2007/12<br />
730/2009/12<br />
1/2006/1500<br />
1768/2006/12<br />
3596/2008/12<br />
6429/2009/12<br />
4581/2010/12<br />
6310/2010/12<br />
1298/2007/12<br />
od 11.04.2007<br />
od 25.02.2009<br />
od 10.02.2006<br />
od 02.10.2006<br />
od 10.10.2008<br />
od 19.10.2009<br />
od 18.10.2010<br />
od<br />
od<br />
03.11.2010<br />
12.06.2007<br />
4/2006/1500 od 10.02.2006<br />
Str. 10 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
CEFAK KG,<br />
Ostbahnhofstraße 15,<br />
Kempten, Nema~ka<br />
CEFAK KG,<br />
Ostbahnhofstraße 15,<br />
Kempten, Nema~ka<br />
CEFAK KG,<br />
Ostbahnhofstraße 15,<br />
Kempten, Nema~ka<br />
CEFAK KG,<br />
Ostbahnhofstraße 15,<br />
Kempten, Nema~ka<br />
CEFAK KG,<br />
Ostbahnhofstraße 15,<br />
Kempten, Nema~ka<br />
CEFAK KG,<br />
Ostbahnhofstraße 15,<br />
Kempten, Nema~ka<br />
CEFAK KG,<br />
Ostbahnhofstraße 15,<br />
Kempten, Nema~ka<br />
GALENIKA AD, Batajni~ki<br />
drum b.b., Beograd, Srbija<br />
Deutsche Homöopathie-<br />
Union DHU-Arzneimittel<br />
GmbH & Co. KG.,<br />
Ottostrasse 24, 76227<br />
Karlsruhe, Nema~ka<br />
MEDICO PHARM SERVIS,<br />
Save Kova~evi}a 113A,<br />
Beograd - Kalu|erica, Srbija<br />
MEDICO PHARM SERVIS,<br />
Save Kova~evi}a 113A,<br />
Beograd - Kalu|erica, Srbija<br />
MEDICO PHARM SERVIS,<br />
Save Kova~evi}a 113A,<br />
Beograd - Kalu|erica, Srbija<br />
MEDICO PHARM SERVIS,<br />
Save Kova~evi}a 113A,<br />
Beograd - Kalu|erica, Srbija<br />
MEDICO PHARM SERVIS,<br />
Save Kova~evi}a 113A,<br />
Beograd - Kalu|erica, Srbija<br />
MEDICO PHARM SERVIS,<br />
Save Kova~evi}a 113A,<br />
Beograd - Kalu|erica, Srbija<br />
MEDICO PHARM SERVIS,<br />
Save Kova~evi}a 113A,<br />
Beograd - Kalu|erica, Srbija<br />
GALENIKA AD, Batajni~ki<br />
drum b.b., Beograd, Srbija<br />
LIBRA BGD D.O.O.-<br />
Obalskih radnika 4 g<br />
Beograd, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Coldrex®<br />
paracetamol, kofein,<br />
fenilefrin, terpin, askorbinska<br />
kiselina<br />
Coldrex®<br />
paracetamol, kofein,<br />
fenilefrin, terpin, askorbinska<br />
kiselina<br />
Coldrex® Caplets<br />
paracetamol, kofein,<br />
fenilefrin<br />
Coldrex® Caplets<br />
paracetamol, kofein,<br />
fenilefrin<br />
Coldrex® HotRem<br />
Blackcurrant<br />
fenilefrin, paracetamol,<br />
askorbinska kiselina<br />
Coldrex® HotRem Lemon<br />
fenilefrin, paracetamol,<br />
askorbinska kiselina<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
tableta;<br />
500mg+25mg+5mg+20mg+30mg;<br />
blister, 1x12kom<br />
tableta;<br />
500mg+25mg+5mg+20mg+30mg;<br />
blister, 2x12kom<br />
tableta;<br />
500mg+25mg+5mg+20mg+30mg;<br />
blister, 1x12kom<br />
tableta;<br />
500mg+25mg+5mg+20mg+30mg;<br />
blister, 2x12kom<br />
film tableta; 500mg+25mg+5mg;<br />
blister, 2x8kom<br />
film tableta; 500mg+25mg+5mg;<br />
blister, 2x8kom<br />
pra{ak <strong>za</strong> oralni rastvor;<br />
10mg+750mg+60mg;<br />
kesica, 5x5g<br />
pra{ak <strong>za</strong> oralni rastvor;<br />
10mg+750mg+60mg;<br />
kesica, 10x5g<br />
pra{ak <strong>za</strong> oralni rastvor;<br />
10mg+750mg+60mg;<br />
kesica, 5x5g<br />
pra{ak <strong>za</strong> oralni rastvor;<br />
10mg+750mg+60mg;<br />
kesica, 10x5g<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
<strong>Agencija</strong> <strong>za</strong> <strong>lekove</strong> i <strong>medicinska</strong> <strong>sredstva</strong> <strong>Srbije</strong> Str. 11 od 42<br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
3765/2009/12<br />
3766/2009/12<br />
814/2010/12<br />
815/2010/12<br />
804/2009/12<br />
4510/2008/12<br />
6062/2009/12<br />
6063/2009/12<br />
3767/2009/12<br />
3768/2009/12<br />
od 08.07.2009<br />
od 08.07.2009<br />
od 03.02.2010<br />
od 03.02.2010<br />
od 15.12.2008<br />
od 15.12.2008<br />
od 09.10.2009<br />
od 09.10.2009<br />
od 08.07.2009<br />
od<br />
08.07.2009<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
GLAXOSMITHKLINE<br />
DUNGARVAN LTD,<br />
Knockbrack, Dungarvan,<br />
Co.Waterford, Irska<br />
HBM PHARMA S.R.O.,<br />
Sklabinska 30,<br />
Martin, Slova~ka<br />
GLAXOSMITHKLINE<br />
CONSUMER HEALTHCARE<br />
GMBH & CO., KG,<br />
Benzstrasse 25,<br />
Herrenberg, Nema~ka<br />
GLAXOSMITHKLINE<br />
DUNGARVAN LTD,<br />
Knockbrack, Dungarvan,<br />
Co.Waterford, Irska<br />
SMITHKLINE BEECHAM<br />
S.A., Ctra. de Ajalvir, Km<br />
2.500, Alcala de Henares,<br />
Madrid, [panija<br />
SMITHKLINE BEECHAM<br />
S.A., Ctra. de Ajalvir, Km<br />
2.500, Alcala de Henares,<br />
Madrid, [panija<br />
GLAXOSMITHKLINE<br />
EXPORT LIMITED -<br />
PREDSTAVNI[TVO,<br />
Omladinskih brigada 88,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
GLAXOSMITHKLINE<br />
EXPORT LIMITED -<br />
PREDSTAVNI[TVO,<br />
Omladinskih brigada 88,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
GLAXOSMITHKLINE<br />
EXPORT LIMITED -<br />
PREDSTAVNI[TVO,<br />
Omladinskih brigada 88,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
GLAXOSMITHKLINE<br />
EXPORT LIMITED -<br />
PREDSTAVNI[TVO,<br />
Omladinskih brigada 88,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
GLAXOSMITHKLINE<br />
EXPORT LIMITED -<br />
PREDSTAVNI[TVO,<br />
Omladinskih brigada 88,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
GLAXOSMITHKLINE<br />
EXPORT LIMITED -<br />
PREDSTAVNI[TVO,<br />
Omladinskih brigada 88,<br />
Beograd - Novi Beograd,<br />
Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Coldrex® HotRem med i<br />
limun<br />
fenilefrin, paracetamol,<br />
askorbinska kiselina<br />
Coldrex® Junior HotRem<br />
fenilefrin, paracetamol,<br />
askorbinska kiselina<br />
Contractubex®<br />
heparin-natrijum, alantoin,<br />
te~ni ekstrakt crnog luka<br />
Deep Freeze Cold Gel<br />
mentol<br />
Deep Heat Rub<br />
metilsalicilat, mentol,<br />
etarsko ulje eukaliptusa,<br />
terpentinsko ulje<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
pra{ak <strong>za</strong> oralni rastvor;<br />
10mg+750mg+60mg;<br />
kesica, 10x5g<br />
pra{ak <strong>za</strong> oralni rastvor;<br />
5mg+300mg+20mg;<br />
kesica, 10x3g<br />
gel; 50i.j./g+0.01g/g+0.1g/g;<br />
tuba, 1x20g<br />
gel; 2%; tuba, 1x35g<br />
gel; 2%; tuba, 1x100g<br />
krem;<br />
12.8%+5.91%+1.97%+1.47%;<br />
tuba, 1x15g<br />
krem;<br />
12.8%+5.91%+1.97%+1.47%;<br />
tuba, 1x35g<br />
krem;<br />
12.8%+5.91%+1.97%+1.47%;<br />
tuba, 1x67g<br />
krem;<br />
12.8%+5.91%+1.97%+1.47%;<br />
tuba, 1x100g<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
4232/2008/12<br />
4233/2008/12<br />
2983/2010/12<br />
1995/2008/12<br />
1996/2008/12<br />
888/2008/12<br />
889/2008/12<br />
890/2008/12<br />
891/2008/12<br />
od 25.11.2008<br />
od 25.11.2008<br />
od 11.05.2010<br />
od<br />
od<br />
10.06.2008<br />
10.06.2008<br />
od 04.03.2008<br />
od 04.03.2008<br />
od 04.03.2008<br />
od<br />
04.03.2008<br />
Str. 12 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
SMITHKLINE BEECHAM<br />
S.A., Ctra. de Ajalvir, Km<br />
2.500, Alcala de Henares,<br />
Madrid, [panija<br />
SMITHKLINE BEECHAM<br />
S.A., Ctra. de Ajalvir, Km<br />
2.500, Alcala de Henares,<br />
Madrid, [panija<br />
MERZ PHARMA GMBH &<br />
CO. KGaA, Ludwigstrasse 22,<br />
Reinheim, Nema~ka<br />
THE MENTHOLATUM<br />
COMPANY LIMITED,<br />
1 Redwood Avenue,<br />
Peel Park Campus,<br />
East Kilbride,<br />
Velika Britanija<br />
THE MENTHOLATUM<br />
COMPANY LIMITED,<br />
1 Redwood Avenue,<br />
Peel Park Campus,<br />
East Kilbride, Velika Britanija<br />
GLAXOSMITHKLINE<br />
EXPORT LIMITED -<br />
PREDSTAVNI[TVO,<br />
Omladinskih brigada 88,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
GLAXOSMITHKLINE<br />
EXPORT LIMITED -<br />
PREDSTAVNI[TVO,<br />
Omladinskih brigada 88,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
LIBRA BGD D.O.O.,<br />
Obalskih radnika 4 g,<br />
Beograd, Srbija<br />
OKTAL PHARMA D.O.O.,<br />
Ju`ni bulevar 142,<br />
Beograd, Srbija<br />
OKTAL PHARMA D.O.O.,<br />
Ju`ni bulevar 142,<br />
Beograd, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Deep Heat Spray<br />
metilsalicilat, metilnikotinat,<br />
etilsalicilat,<br />
hidroksietilsalicilat<br />
Deep Relief<br />
ibuprofen, levomentol<br />
Defrinol®<br />
pseudoefedrin, ibuprofen<br />
Dentinox® N<br />
tinktura kamilice, lidokain,<br />
makrogol lauriletar<br />
Dentokind<br />
Belladonna trit. D6,<br />
Chamomilla trit. D6, Ferrum<br />
phosphoricum trit. D6,<br />
Hepar sulfuris trit. D12,<br />
Pulsatilla trit. D6<br />
Dermodrin<br />
difenhidramin<br />
Diclofenac Duo 4% Spray<br />
Gel<br />
diklofenak<br />
Diklofenak<br />
diklofenak<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
sprej <strong>za</strong> ko`u, rastvor;<br />
1%+1.6%+5%+5%; kontejner<br />
pod pritiskom, 1x150ml<br />
gel; 5%+3%; tuba, 1x50g<br />
gel; 5%+3%; tuba, 1x30g<br />
gel; 5%+3%; tuba, 1x15g<br />
film tableta; 30mg+200mg;<br />
blister, 2x10kom<br />
sirup; 30mg/5ml+100mg/5ml;<br />
boca staklena, 1x100ml<br />
rastvor <strong>za</strong> desni;<br />
150mg/g+3.4mg/g+3.2mg/g;<br />
bo~ica, 1x10g<br />
tableta;<br />
15mg+15mg+15mg+15mg+15mg;<br />
bo~ica staklena, 1x150kom<br />
krem; 20mg/g; tuba, 1x20g<br />
krem; 20mg/g; tuba, 1x50g<br />
sprej <strong>za</strong> ko`u, rastvor; 4%;<br />
bo~ica sa raspr{iva~em, 1x12.5g<br />
sprej <strong>za</strong> ko`u, rastvor; 4%;<br />
bo~ica sa raspr{iva~em, 1x25g<br />
gel; 1%; tuba, 1x40g<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
892/2008/12<br />
125/2008/12<br />
126/2008/12<br />
127/2008/12<br />
2042/2006/12<br />
2043/2006/12<br />
1439/2008/12<br />
3247/2010/12<br />
1422/2007/12<br />
1423/2007/12<br />
118/2007/12<br />
119/2007/12<br />
3037/2007/12<br />
od 04.03.2008<br />
od 22.01.2008<br />
od 22.01.2008<br />
od 22.01.2008<br />
od 06.11.2006<br />
od 06.11.2006<br />
od 17.04.2008<br />
od 04.06.2010<br />
od 05.07.2007<br />
od 05.07.2007<br />
od 11.01.2007<br />
od 11.01.2007<br />
od<br />
18.12.2007<br />
Str. 13 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
THE MENTHOLATUM<br />
COMPANY LIMITED,<br />
1 Redwood Avenue,<br />
Peel Park Campus,<br />
East Kilbride, Velika Britanija<br />
THE MENTHOLATUM<br />
COMPANY LIMITED,<br />
1 Redwood Avenue,<br />
Peel Park Campus,<br />
East Kilbride, Velika Britanija<br />
GALENIKA AD,<br />
Batajni~ki drum b.b.,<br />
Beograd, Srbija<br />
DENTINOX GESELLSCHAFT<br />
FÜR PHARMAZEUTISCHE<br />
PRÄPARATE LENK &<br />
SCHUPPAN, Nunsdorfer Ring<br />
19, Berlin, Nema~ka<br />
Deutsche Homöopathie-Union<br />
DHU-Arzneimittel GmbH &<br />
Co. KG., Ottostrasse 24,<br />
76227 Karlsruhe, Nema~ka<br />
PHARMAZEUTISCHE<br />
FABRIK MONTAVIT<br />
GES.M.B.H.,<br />
Salzbergstrasse 96, Absam,<br />
Tirol, Austrija<br />
PHARBIL WALTROP GMBH,<br />
Im Wirrigen 25,<br />
Waltrop, Nema~ka<br />
HEMOFARM AD, Beogradski<br />
put bb, Vr{ac, Srbija<br />
OKTAL PHARMA D.O.O.,<br />
Ju`ni bulevar 142,<br />
Beograd, Srbija<br />
OKTAL PHARMA D.O.O.,<br />
Ju`ni bulevar 142,<br />
Beograd, Srbija<br />
GALENIKA AD,<br />
Batajni~ki drum b.b.,<br />
Beograd, Srbija<br />
NYCOMED GMBH -<br />
PREDSTAVNI[TVO,<br />
Krunska 24/14,<br />
Beograd, Srbija<br />
LIBRA BGD D.O.O.,<br />
Obalskih radnika 4 g,<br />
Beograd, Srbija<br />
PHARMAZEUTISCHE<br />
FABRIK MONTAVIT<br />
GES.M.B.H. -<br />
PREDSTAVNI[TVO,<br />
Lipova~ki put 390,<br />
Beograd-Barajevo, Srbija<br />
PHARMASWISS D.O.O.,<br />
Vojvode Stepe 18,<br />
Beograd, Srbija<br />
HEMOFARM AD, Beogradski<br />
put bb, Vr{ac, Srbija
Diklofen®<br />
diklofenak<br />
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Dimigal®<br />
dimenhidrinat<br />
Dona®<br />
gluko<strong>za</strong>min<br />
Dona®<br />
gluko<strong>za</strong>min<br />
Dona®<br />
gluko<strong>za</strong>min<br />
Dramina®<br />
dimenhidrinat<br />
Duphalac®<br />
laktulo<strong>za</strong><br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
gel; 1%; tuba, 1x50g<br />
tableta; 50mg; blister, 1x10kom<br />
pra{ak <strong>za</strong> oralni rastvor;1500mg; kesica, 4x 1500mg<br />
pra{ak <strong>za</strong> oralni rastvor;1500mg;kesica, 30x1500mg<br />
pra{ak <strong>za</strong> oralni rastvor;1500mg;kesica, 20x1500mg<br />
pra{ak <strong>za</strong> oralni rastvor;1500mg;<br />
kesica, 30x1500mg<br />
pra{ak <strong>za</strong> oralni rastvor;1500mg;<br />
kesica, 20x1500mg<br />
pra{ak <strong>za</strong> oralni rastvor;1500mg;kesica, 30x1500mg<br />
pra{ak <strong>za</strong> oralni rastvor;1500mg;kesica, 20x1500mg<br />
Dormikind<br />
tableta; 15mg+20mg+15mg;<br />
Cypripedium pubescens D4,<br />
Magnesium carbonicum D10,<br />
bo~ica staklena, 1x150kom<br />
Zincum valerianicum D12<br />
tableta; 50mg; blister, 1x10kom<br />
oralni rastvor; 667g/l;<br />
boca plasti~na, 1x200ml<br />
oralni rastvor; 667g/l;<br />
kesica, 10x15ml<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
1226/2008/12<br />
2265/2006/12<br />
4165/2009/12<br />
1519/2008/12<br />
1517/2008/12<br />
895/2008/12<br />
894/2008/12<br />
1520/2008/12<br />
1518/2008/12<br />
3248/2010/12<br />
1877/2006/12<br />
241/2009/12<br />
242/2009/12<br />
od<br />
od<br />
od<br />
od<br />
od<br />
od<br />
od<br />
od<br />
od<br />
od<br />
od<br />
28.03.2008<br />
06.12.2006<br />
31.07.2009<br />
05.05.2008<br />
05.05.2008<br />
04.03.2008<br />
04.03.2008<br />
05.05.2008<br />
05.05.2008<br />
04.06.2010<br />
18.10.2006<br />
od 28.01.2009<br />
od 28.01.2009<br />
Str. 14 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
GALENIKA AD, Batajni~ki<br />
drum b.b., Beograd, Srbija<br />
GALENIKA AD, Batajni~ki<br />
drum b.b., Beograd, Srbija<br />
ROTTAPHARM LTD.,<br />
Damastown Industrial Park,<br />
Mulhuddart, Dublin, Irska<br />
ROTTAPHARM S.P.A., Via<br />
Valosa di Sopra, 9, Mon<strong>za</strong>,<br />
Milano, Italija<br />
SIGMAR ITALIA S.P.A.,<br />
Via Sombreno,11, Alme,<br />
Bergamo, Italija<br />
Deutsche Homöopathie-Union<br />
DHU-Arzneimittel GmbH &<br />
Co. KG., Ottostrasse 24,<br />
76227 Karlsruhe, Nema~ka<br />
JADRAN GALENSKI<br />
LABORATORIJ D.D.,<br />
Pulac bb, Rijeka, Hrvatska<br />
SOLVAY BIOLOGICALS B.V.,<br />
Veerweg 12, Olst, Holandija<br />
GALENIKA AD, Batajni~ki<br />
drum b.b., Beograd, Srbija<br />
GALENIKA AD, Batajni~ki<br />
drum b.b., Beograd, Srbija<br />
GOODWILL PHARMA<br />
D.O.O., Matije Gubca 14,<br />
Subotica, Srbija<br />
GOODWILL PHARMA<br />
D.O.O., Matije Gubca 14,<br />
Subotica, Srbija<br />
GOODWILL PHARMA<br />
D.O.O., Matije Gubca 14,<br />
Subotica, Srbija<br />
LIBRA BGD D.O.O.,<br />
Obalskih radnika 4 g,<br />
Beograd, Srbija<br />
JADRAN GALENSKI<br />
LABORATORIJ D.D -<br />
PREDSTAVNI[TVO,<br />
Mirijevski bulevar 37 a,<br />
Beograd, Srbija<br />
UNIFARM MEDICOM D.O.O.,<br />
Cara Du{ana 264,<br />
Beograd, Srbija
Efferalgan®<br />
paracetamol<br />
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Efferalgan®<br />
paracetamol<br />
Efferalgan® sa<br />
vitaminom C<br />
paracetamol, askorbinska<br />
kiselina<br />
Ehido®<br />
standardizovani suvi<br />
ekstrakt korena purpurne<br />
ehinacee, tinktura timijana,<br />
macerat korena belog sle<strong>za</strong><br />
Elevit pronatal<br />
retinol, tiamin, riboflavin,<br />
piridoksin, cijanokobalamin,<br />
askorbinska kiselina,<br />
holekalciferol, alfa-tokoferil<br />
acetat, kalcijum-pantotenat,<br />
biotin, folna kiselina,<br />
nikotinamid, gvo`|e,<br />
kalcijum, bakar,<br />
magnezijum, mangan,<br />
fosfor, cink<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
supozitorija; 80mg; blister,<br />
2x5kom<br />
supozitorija; 150mg; blister,<br />
2x5kom<br />
{ume}a tableta; 500mg; strip,<br />
4x4kom<br />
oralni rastvor; 30mg/ml; bo~ica,<br />
1x90ml<br />
supozitorija; 80mg; blister,<br />
2x5kom<br />
supozitorija; 150mg; blister,<br />
2x5kom<br />
oralni rastvor; 30mg/ml; bo~ica,<br />
1x90ml<br />
{ume}a tableta; 500mg; strip,<br />
4x4kom<br />
{ume}a tableta; 330mg+200mg;<br />
fiola, 1x10kom<br />
{ume}a tableta; 330mg+200mg;<br />
fiola, 1x20kom<br />
sirup;<br />
8.4mg/g+10mg/g+332mg/g;<br />
bo~ica, 1x150g<br />
film tableta; 3600i.j.+1.6mg+<br />
1.8mg+2.6mg+4mcg+100mg+<br />
500i.j.+15i.j.+10mg+200mcg+<br />
0.8mg+19mg+60mg+125mg+<br />
1mg+100mg+1mg+125mg+<br />
7.5mg; blister, 3x10kom<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
2288/2006/12<br />
2289/2006/12<br />
547/2007/12<br />
133/2008/12<br />
2295/2008/12<br />
2296/2008/12<br />
2297/2008/12<br />
3512/2010/12<br />
821/2007/12<br />
822/2007/12<br />
440/2006/12<br />
6597/2010/12<br />
od 11.12.2006<br />
od 11.12.2006<br />
od 27.02.2007<br />
od 22.01.2008<br />
od 15.07.2008<br />
od 15.07.2008<br />
od 15.07.2008<br />
od 05.07.2010<br />
od 13.04.2007<br />
od 28.07.2007<br />
od 06.04.2006<br />
od 22.11.2010<br />
Str. 15 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
BRISTOL-MYERS SQUIBB,<br />
304, avenue Jean Bru,<br />
Agen, Francuska<br />
BRISTOL-MYERS SQUIBB,<br />
979 Avenue des Pyrénées,<br />
Le Passage, Francuska<br />
BRISTOL-MYERS SQUIBB,<br />
304, avenue Jean Bru, Agen,<br />
Francuska<br />
PHARMANOVA<br />
VELEPRODAJA D.O.O., Bore<br />
Stankovi}a 2, P.Box 21,<br />
Beograd, Srbija<br />
ROTTENDORF<br />
PHARMA GMBH,<br />
Ostenfelder Strasse 51 - 61,<br />
Ennigerloh, Nema~ka<br />
PHARMASWISS D.O.O.,<br />
Vojvode Stepe 18,<br />
Beograd, Srbija<br />
PHARMASWISS D.O.O.,<br />
Vojvode Stepe 18,<br />
Beograd, Srbija<br />
PHARMASWISS D.O.O.,<br />
Vojvode Stepe 18,<br />
Beograd, Srbija<br />
PHARMANOVA D.O.O.,<br />
Industrijska 8,<br />
Obrenovac, Srbija<br />
BAYER D.O.O.,<br />
Omladinskih brigada 88b,<br />
Beograd - Novi<br />
Beograd, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Enterokind<br />
Chamomilla dil. D6, Cina dil.<br />
D6, Colocynthis dil. D6, Lac<br />
defloratum dil. D6,<br />
Magnesium chloratum dil.<br />
D6<br />
Escapelle®<br />
levonorgestrel<br />
Espumisan®<br />
simetikon<br />
Espumisan® L<br />
simetikon<br />
Essentiale® forte N<br />
esencijalni fosfolipidi<br />
Evitol®<br />
tokoferol<br />
Febricet<br />
paracetamol<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
oralni rastvor;<br />
(10g+10g+10g+10g+10g)/100g;<br />
bo~ica staklena, 1x20ml<br />
tableta; 1.5mg; blister, 1x1kom<br />
kapsula, meka; 40mg;<br />
blister, 1x25kom<br />
kapsula, meka; 40mg;<br />
blister, 4x25kom<br />
kapsula, meka; 40mg;<br />
blister, 2x25kom<br />
oralna emulzija; 40mg/ml;<br />
boca staklena, 1x30ml<br />
kapsula, tvrda; 300mg;<br />
blister, 3x10kom<br />
oblo`ena tableta; 100mg;<br />
blister, 3x10kom<br />
supozitorija; 200mg;<br />
strip, 1x5kom<br />
sirup; 120mg/5ml;<br />
boca staklena, 1x100ml<br />
{ume}a tableta; 500mg;<br />
fiola, 1x10kom<br />
{ume}a tableta; 500mg;<br />
fiola, 2x10kom<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
<strong>Agencija</strong> <strong>za</strong> <strong>lekove</strong> i <strong>medicinska</strong> <strong>sredstva</strong> <strong>Srbije</strong> Str. 16 od 42<br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
4730/2009/12<br />
4508/2008/12<br />
695/2006/12<br />
697/2006/12<br />
696/2006/12<br />
657/2006/12<br />
1327/2008/12<br />
1772/2006/12<br />
1511/2008/12<br />
4137/2008/12<br />
3057/2007/12<br />
3052/2007/12<br />
od 14.09.2009<br />
od 15.12.2008<br />
od 25.05.2006<br />
od 25.05.2006<br />
od 25.05.2006<br />
od 18.05.2006<br />
od 04.04.2008<br />
od 02.10.2006<br />
od 05.05.2008<br />
od 14.11.2008<br />
od 18.12.2007<br />
od<br />
18.12.2007<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
Deutsche Homöopathie-Union<br />
DHU-Arzneimittel GmbH &<br />
Co. KG., Ottostrasse 24,<br />
76227 Karlsruhe, Nema~ka<br />
GEDEON RICHTER PLC,<br />
Gyömrõi út. 19-21,<br />
Budimpe{ta, Ma|arska<br />
BERLIN-CHEMIE AG<br />
(MENARINI GROUP),<br />
Glienicker Weg 125,<br />
Berlin, Nema~ka<br />
BERLIN-CHEMIE AG<br />
(MENARINI GROUP),<br />
Glienicker Weg 125,<br />
Berlin, Nema~ka<br />
A. NATTERMANN &CIE.<br />
GMBH, Nattermannallee 1,<br />
Keln, Nema~ka<br />
KRKA, TOVARNA ZDRAVIL,<br />
D.D., [marje{ka cesta 6,<br />
Novo Mesto, Slovenija<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
LIBRA BGD D.O.O.,<br />
Obalskih radnika 4 g,<br />
Beograd, Srbija<br />
RICHTER GEDEON NYRT-<br />
PREDSTAVNI[TVO,<br />
Vladimira Popovi}a 6,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
BERLIN-CHEMIE AG<br />
(MENARINI GROUP) -<br />
PREDSTAVNI[TVO,<br />
Prilepska 1,<br />
Beograd, Srbija<br />
BERLIN-CHEMIE AG<br />
(MENARINI GROUP) -<br />
PREDSTAVNI[TVO,<br />
Prilepska 1, Beograd, Srbija<br />
SANOFI-AVENTIS D.O.O.,<br />
Vladimira Popovi}a 40/IV,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
KRKA FARMA D.O.O.,<br />
Jurija Gagarina 26/V/II,<br />
Beograd, Srbija<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Febricet C<br />
paracetamol, askorbinska<br />
kiselina<br />
Febricet Cold<br />
feniramin, paracetamol,<br />
askorbinska kiselina<br />
Febricet Cold Junior<br />
feniramin, paracetamol,<br />
askorbinska kiselina<br />
Fenistil<br />
dimetinden<br />
Fitoven®<br />
te~ni etanolni ekstrakt<br />
semena divljeg kestena,<br />
te~ni etanolni ekstrakt<br />
kokoca, etarsko ulje<br />
ruzmarina, etarsko ulje<br />
lavande<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
{ume}a tableta; 330mg+200mg;<br />
fiola, 1x10kom<br />
pra{ak <strong>za</strong> oralni rastvor;<br />
25mg+500mg+200mg;<br />
kesica, 8x4g<br />
pra{ak <strong>za</strong> oralni rastvor;<br />
10mg+280mg+100mg;<br />
kesica, 8x5g<br />
gel; 0.1%; tuba, 1x30g<br />
gel;<br />
350mg/g+20mg/g+2mg/g+1mg/g;<br />
tuba, 1x45g<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
314/2007/12<br />
od 23.01.2007<br />
357/2008/12 od 11.02.2008<br />
355/2008/12<br />
1476/2008/12<br />
od<br />
od<br />
11.02.2008<br />
8410/2009/12 od 24.12.2009<br />
24.04.2008<br />
Str. 17 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
NOVARTIS CONSUMER<br />
HEALTH SA,<br />
Route de l'Etraz,<br />
Nyon, [vajcarska<br />
ZDRAVLJE A.D.,<br />
Vlajkova 199,<br />
Leskovac, Srbija<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
MEDIS INTERNATIONAL<br />
D.O.O., Bulevar Mihaila<br />
Pupina 10d/1 pp7, Beograd -<br />
Novi Beograd, Srbija<br />
ZDRAVLJE A.D.,<br />
Vlajkova 199,<br />
Leskovac, Srbija
Flavamed®<br />
ambroksol<br />
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Flonivin-BS®<br />
Bacillus IP 5832<br />
Fluimucil® 100<br />
acetilcistein<br />
Fluimucil® 200<br />
acetilcistein<br />
Fluimucil® 600<br />
acetilcistein<br />
Fluorogal®<br />
natrijum-fluorid<br />
Gastal®<br />
aluminijum-hidroksidmagnezijum-karbonat,<br />
magnezijum-hidroksid<br />
Gastrokind<br />
Arsenicum album trit. D6,<br />
Croton tiglium trit. D6,<br />
Okoubaka trit. D4, Veratrum<br />
album trit. D12<br />
Gasvan<br />
dimetikon<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
oralni rastvor; 15mg/5ml;<br />
bo~ica, 1x100ml<br />
oralni rastvor; 15mg/5ml;<br />
bo~ica, 1x60ml<br />
kapsula, tvrda; 35mg;<br />
blister, 2x8kom<br />
granule <strong>za</strong> oralni rastvor; 100mg;<br />
kesica, 30x1g<br />
granule <strong>za</strong> oralni rastvor; 200mg;<br />
kesica, 30x1g<br />
{ume}a tableta; 600mg;<br />
strip, 5x2kom<br />
tableta; 1mg;<br />
plasti~na fiola, 1x100kom<br />
tableta; 0.25mg;<br />
plasti~na fiola, 1x250kom<br />
tableta; 450mg+300mg;<br />
blister, 8x6kom<br />
tableta; 450mg+300mg;<br />
blister, 4x6kom<br />
tableta;<br />
20mg+20mg+20mg+20mg;<br />
bo~ica staklena, 1x150kom<br />
tableta <strong>za</strong> `vakanje; 80mg;<br />
bo~ica plasti~na, 1x20kom<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
1108/2008/12<br />
1109/2008/12<br />
5251/2009/12<br />
814/2007/12<br />
815/2007/12<br />
816/2007/12<br />
295/2009/12<br />
296/2009/12<br />
5831/2009/12<br />
5830/2009/12<br />
4731/2009/12<br />
3375/2010/12<br />
od 18.03.2008<br />
od 18.03.2008<br />
od 22.09.2009<br />
od 13.04.2007<br />
od 13.04.2007<br />
od 13.04.2007<br />
od 06.02.2009<br />
od 06.02.2009<br />
od 02.10.2009<br />
od 02.10.2009<br />
od 14.09.2009<br />
od<br />
11.06.2010<br />
Str. 18 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
BERLIN-CHEMIE AG<br />
(MENARINI GROUP),<br />
Glienicker Weg 125,<br />
Berlin, Nema~ka<br />
GALENIKA CRNA GORA<br />
D.O.O., 8.marta 55-A,<br />
Podgorica, Crna Gora<br />
ZAMBON SWITZERLAND<br />
LTD, Via Industria, 13,<br />
Cadempino, [vajcarska<br />
ZAMBON SWITZERLAND<br />
LTD, Via Industria, 13,<br />
Cadempino, [vajcarska<br />
ZAMBON SWITZERLAND<br />
LTD, Via Industria, 13,<br />
Cadempino, [vajcarska<br />
GALENIKA AD,<br />
Batajni~ki drum b.b.,<br />
Beograd, Srbija<br />
PLIVA HRVATSKA D.O.O.,<br />
Prilaz baruna Filipovi}a 25,<br />
Zagreb, Hrvatska<br />
Deutsche Homöopathie-Union<br />
DHU-Arzneimittel GmbH &<br />
Co. KG., Ottostrasse 24,<br />
76227 Karlsruhe, Nema~ka<br />
SRBOLEK A.D., Sarajevska<br />
84, Beograd, Srbija<br />
BERLIN-CHEMIE AG<br />
(MENARINI GROUP) -<br />
PREDSTAVNI[TVO,<br />
Prilepska 1, Beograd, Srbija<br />
GALENIKA AD,<br />
Batajni~ki drum b.b.,<br />
Beograd, Srbija<br />
BONIFAR D.O.O.,<br />
Bulevar Zorana \in|i}a 87/1,<br />
Beograd, Srbija<br />
BONIFAR D.O.O.,<br />
Bulevar Zorana \in|i}a 87/1,<br />
Beograd, Srbija<br />
BONIFAR D.O.O.,<br />
Bulevar Zorana \in|i}a 87/1,<br />
Beograd, Srbija<br />
GALENIKA AD,<br />
Batajni~ki drum b.b.,<br />
Beograd, Srbija<br />
UNIMED D.O.O.,<br />
De~anska 12/II,<br />
Beograd - Stari grad, Srbija<br />
LIBRA BGD D.O.O.,<br />
Obalskih radnika 4 g,<br />
Beograd, Srbija<br />
SRBOLEK A.D., Sarajevska<br />
84, Beograd, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
GeloMyrtol® forte<br />
sme{a etarskih ulja<br />
eukaliptusa, slatke<br />
narand`e, mirte i limuna,<br />
standardizovana na 1,8cineol,<br />
d-limonen i (+)-alfapinen<br />
Gelusil® - Lac<br />
aluminijum-magnezijumsilikat<br />
Gentos®<br />
populus tremuloides trit. D1,<br />
serenoa repens trit. D6,<br />
conium maculatum trit. D6,<br />
kalium iodatum trit. D12,<br />
ferrum picrinicum trit. D12<br />
Ginko Sandoz®<br />
list ginka, suvi<br />
standardizovani ekstrakt<br />
Ginkor Fort®<br />
trokserutin, list ginka, suvi<br />
standardizovani ekstrakt,<br />
heptaminol<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
gastrorezistentna kapsula, meka;<br />
300mg; blister, 2x10kom<br />
tableta; 500mg;<br />
blister, 4x10kom<br />
tableta; 74.5mg+37.2mg+<br />
37.2mg+37.2mg+37.2mg;<br />
blister, 2x20kom<br />
tableta; 74.5mg+37.2mg+<br />
37.2mg+37.2mg+37.2mg;<br />
blister, 1x20kom<br />
oralne kapi, rastvor; (7ml+10ml+<br />
10ml+10ml+10ml)/100ml;<br />
bo~ica sa kapaljkom, 1x50ml<br />
oralne kapi, rastvor; (7ml+10ml+<br />
10ml+10ml+10ml)/100ml;<br />
bo~ica sa kapaljkom, 1x20ml<br />
film tableta; 40mg;<br />
blister, 3x10kom<br />
film tableta; 80mg;<br />
blister, 3x10kom<br />
film tableta; 120mg;<br />
blister, 3x10kom<br />
film tableta; 40mg;<br />
blister, 9x10kom<br />
kapsula, tvrda;<br />
300mg+14mg+300mg;<br />
blister, 3x10kom<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
347/2008/12<br />
1078/2007/12<br />
6307/2010/12<br />
6306/2010/12<br />
6308/2010/12<br />
6309/2010/12<br />
4215/2008/12<br />
4216/2008/12<br />
4217/2008/12<br />
4218/2008/12<br />
2867/2009/12<br />
od 07.02.2008<br />
od 23.05.2007<br />
od 03.11.2010<br />
od 03.11.2010<br />
od 03.11.2010<br />
od 03.11.2010<br />
od 25.11.2008<br />
od 25.11.2008<br />
od 25.11.2008<br />
od 25.11.2008<br />
od<br />
27.05.2009<br />
Str. 19 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
G. POHL-BOSKAMP GMBH<br />
& CO. KG, Kieler Straße 11,<br />
Hohenlockstedt, Nema~ka<br />
HEMOFARM AD, Beogradski<br />
put bb, Vr{ac, Srbija u<br />
saradnji sa PFIZER H.C.P.<br />
CORPORATION, 235 East<br />
42nd Street, New York, USA<br />
RICHARD BITTNER AG.,<br />
Ossiacher Strasse 7,<br />
Feldkirchen, Austrija<br />
LEK FARMACEVTSKA<br />
DRU@BA D.D.,<br />
Verov{kova 57,<br />
Ljubljana, Slovenija<br />
BEAUFOUR IPSEN<br />
INDUSTRIE, Rue d'Ethe-<br />
Virthon, Dreux, Francuska<br />
MEDIS PHARMA D.O.O.,<br />
Bulevar Mihaila Pupina 10 d/1<br />
pp7, Beograd - Novi Beograd,<br />
Srbija<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
FARMALOGIST D.O.O.,<br />
Bulevar Vojvode<br />
Mi{i}a 25- 27,<br />
Beograd, Srbija<br />
SANDOZ<br />
PHARMACEUTICALS D.D. -<br />
PREDSTAVNI[TVO,<br />
Kneginje Zorke 2,<br />
Beograd, Srbija<br />
PHARMASWISS D.O.O.,<br />
Vojvode Stepe 18,<br />
Beograd, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Grippostad® C<br />
paracetamol, askorbinska<br />
kiselina, kofein, hlorfenamin<br />
Harpadol<br />
koren |avolje kand`e,<br />
pra{ak<br />
Hemodrops<br />
hipromelo<strong>za</strong><br />
Hepalip forte®<br />
esencijalni fosfolipidi,<br />
nikotinamid, piridoksin,<br />
riboflavin, tiamin,<br />
tokoferolacetat<br />
Hepalpan®<br />
heparin-natrijum,<br />
dekspantenol<br />
Hepan®<br />
heparin-natrijum,<br />
dekspantenol<br />
Hepathrombin<br />
heparin-natrijum<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
kapsula, tvrda;<br />
200mg+150mg+25mg+2.5mg;<br />
blister, 2x10kom<br />
kapsula, tvrda; 435mg;<br />
bo~ica plasti~na, 1x45kom<br />
kapi <strong>za</strong> o~i, rastvor; 0.5%;<br />
bo~ica staklena, 1x10ml<br />
kapsula, tvrda; 300mg+30mg+<br />
6mg+6mg+6mg+6mg;<br />
blister, 3x10kom<br />
gel; 300i.j./g+2.5mg/g;<br />
tuba, 1x40g<br />
krem; 300i.j./g+4mg/g;<br />
tuba, 1x40g<br />
gel; 500i.j./g+5mg/g;<br />
tuba, 1x50g<br />
krem; 300i.j./g; tuba, 1x40g<br />
krem; 500i.j./g; tuba, 1x40g<br />
gel; 300i.j./g; tuba, 1x40g<br />
gel; 500i.j./g; tuba, 1x40g<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
1344/2010/12<br />
731/2009/12<br />
4382/2008/12<br />
3116/2008/12<br />
4016/2010/12<br />
4017/2010/12<br />
642/2009/12<br />
1487/2007/12<br />
1488/2007/12<br />
1489/2007/12<br />
1490/2007/12<br />
od<br />
od<br />
22.02.2010<br />
25.02.2009<br />
od 08.12.2008<br />
od 10.09.2008<br />
od 09.08.2010<br />
od 09.08.2010<br />
od 23.02.2009<br />
od 10.07.2007<br />
od 10.07.2007<br />
od 10.07.2007<br />
od<br />
10.07.2007<br />
Str. 20 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
STADA ARZNEIMITTEL AG,<br />
Stadastrasse 2-18, Bad<br />
Vilbel, Nema~ka<br />
LABORATOIRES<br />
ARKOPHARMA, Lid de<br />
Carros Le Broc 1ere avenue,<br />
2709m, Carros, Francuska<br />
HEMOMONT D.O.O.,<br />
8. marta 55a, Podgorica,<br />
Crna Gora<br />
BOSNALIJEK D.D.,<br />
Juki}eva 53, Sarajevo,<br />
Bosna i Hercegovina<br />
GALENIKA AD,<br />
Batajni~ki drum b.b.,<br />
Beograd, Srbija<br />
JADRAN GALENSKI<br />
LABORATORIJ D.D.,<br />
Pulac bb,<br />
Rijeka, Hrvatska<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
ABELA PHARM D.O.O.,<br />
Bulevar Kralja<br />
Aleksandra 137,<br />
Beograd, Srbija<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
BOSNALIJEK D.D. -<br />
PREDSTAVNI[TVO,<br />
Cara Nikolaja II 61/B,<br />
Beograd, Srbija<br />
GALENIKA AD,<br />
Batajni~ki drum b.b.,<br />
Beograd, Srbija<br />
JADRAN GALENSKI<br />
LABORATORIJ D.D -<br />
PREDSTAVNI[TVO,<br />
Mirijevski bulevar 37 a,<br />
Beograd, Srbija<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Hepathrombin H<br />
heparin-natrijum,<br />
prednizolon, polidokanol<br />
Hexoral®<br />
heksetidin<br />
Holyplant Septogal®<br />
nana (Mentha piperita),<br />
etarsko ulje, eukaliptus<br />
(Eucalyptus spp), etarsko<br />
ulje, levomentol, timol,<br />
ben<strong>za</strong>lkonijum-hlorid<br />
Holyplant Septogal® bez<br />
{e}era<br />
nana (Mentha piperita),<br />
etarsko ulje, eukaliptus<br />
(Eucalyptus spp), etarsko<br />
ulje, levomentol,<br />
ben<strong>za</strong>lkonijum-hlorid<br />
Iberogast®<br />
cela sve`a biljka divljeg<br />
renca, etanolni ekstrakt,<br />
koren an|elike, etanolni<br />
ekstrakt, list mati~njaka,<br />
etanolni ekstrakt, plod kima,<br />
etanolni ekstrakt, nadzemni<br />
deo ruse, etanolni ekstrakt,<br />
koren sladi}a, etanolni<br />
ekstrakt, cvet kamilice,<br />
etanolni ekstrakt, list nane,<br />
etanolni ekstrakt, plod gujine<br />
trave, etanolni ekstrakt<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
supozitorija;<br />
120i.j.+1.675mg+30mg;<br />
blister, 1x10kom<br />
rektalna mast;<br />
65i.j./g+2.233mg/g+30mg/g;<br />
tuba sa aplikatorom, 1x20g<br />
rastvor <strong>za</strong> usnu sluznicu;<br />
1mg/ml; boca staklena, 1x200ml<br />
komprimovana lozenga;<br />
1mg+0.6mg+3mg+0.6mg+1mg;<br />
blister, 3x10kom<br />
komprimovana lozenga;<br />
1mg+0.6mg+3mg+1mg;<br />
blister, 3x10kom<br />
oralne kapi, rastvor;<br />
(15ml+10ml+10ml+10ml+10ml+<br />
10ml+20ml+5ml+10ml)/100ml;<br />
bo~ica staklena, 1x20ml<br />
oralne kapi, rastvor;<br />
(15ml+10ml+10ml+10ml+10ml+<br />
10ml+20ml+5ml+10ml)/100ml;<br />
bo~ica staklena, 1x50ml<br />
oralne kapi, rastvor;<br />
(15ml+10ml+10ml+10ml+10ml+<br />
10ml+20ml+5ml+10ml)/100ml;<br />
bo~ica staklena, 1x100ml<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
2062/2007/12<br />
2063/2007/12<br />
196/2009/12<br />
7401/2009/12<br />
7402/2009/12<br />
732/2009/12<br />
733/2009/12<br />
734/2009/12<br />
od 25.09.2007<br />
od 25.09.2007<br />
od 27.01.2009<br />
od 24.11.2009<br />
od 24.11.2009<br />
od 25.02.2009<br />
od 25.02.2009<br />
od<br />
25.02.2009<br />
Str. 21 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
HEMOFARM AD, Beogradski<br />
put bb, Vr{ac, Srbija u<br />
saradnji sa PFIZER H.C.P.<br />
CORPORATION, 235 East<br />
42nd Street, New York, USA<br />
JADRAN GALENSKI<br />
LABORATORIJ D.D.,<br />
Pulac bb,<br />
Rijeka, Hrvatska<br />
JADRAN GALENSKI<br />
LABORATORIJ D.D.,<br />
Pulac bb,<br />
Rijeka, Hrvatska<br />
STEIGERWALD<br />
ARZNEIMITTELWERK<br />
GMBH, Havelstrasse 5,<br />
Darmstadt, Nema~ka<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
JADRAN GALENSKI<br />
LABORATORIJ D.D -<br />
PREDSTAVNI[TVO,<br />
Mirijevski bulevar 37 a,<br />
Beograd, Srbija<br />
JADRAN GALENSKI<br />
LABORATORIJ D.D -<br />
PREDSTAVNI[TVO,<br />
Mirijevski bulevar 37 a,<br />
Beograd, Srbija<br />
LIBRA BGD D.O.O.,<br />
Obalskih radnika 4 g,<br />
Beograd, Srbija
Ibuprofen<br />
ibuprofen<br />
Ibuprofen<br />
ibuprofen<br />
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Immunal®<br />
osu{eni sok herbe<br />
purpurne ehinacee<br />
Immunokind<br />
Calcium carbonicum<br />
Hahnemanni trit. D6,<br />
Calcium fluoratum trit. D6,<br />
Calcium phosphoricum trit.<br />
D6, Sulfur jodatum trit. D12<br />
Influcid®<br />
Aconitum napellus, triturat<br />
D3, Bryonia, triturat D2,<br />
Eupatorium perfoliatum,<br />
triturat D1, Gelsemium<br />
sempervirens, triturat D3,<br />
Ipecacuanha, triturat D3,<br />
Fosfor, triturat D5<br />
Influrex®<br />
fenilefrin, paracetamol,<br />
askorbinska kiselina<br />
Isopto® Tears<br />
hipromelo<strong>za</strong><br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
{ume}a tableta; 200mg;<br />
fiola, 2x10kom<br />
film tableta; 200mg;<br />
blister, 3x10kom<br />
tableta; 80mg; blister, 1x20kom<br />
oralni rastvor; 0.8ml/ml;<br />
bo~ica, 1x50ml<br />
tableta;<br />
20mg+20mg+20mg+20mg;<br />
bo~ica staklena, 1x150kom<br />
tableta;<br />
25mg+25mg+25mg+25mg+25mg<br />
+25mg; blister, 3x20kom<br />
pra{ak <strong>za</strong> oralni rastvor;<br />
10mg+750mg+60mg;<br />
kesica, 5x1kom<br />
kapi <strong>za</strong> o~i, rastvor; 5mg/ml;<br />
bo~ica, 1x15ml<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
2947/2007/12<br />
3789/2009/12<br />
1475/2008/12<br />
957/2007/12<br />
4732/2009/12<br />
6/2006/1500<br />
306/2009/12<br />
2038/2007/12<br />
od 29.11.2007<br />
od<br />
od<br />
09.07.2009<br />
24.04.2008<br />
od 09.05.2007<br />
od 14.09.2009<br />
od 10.02.2006<br />
od 06.02.2009<br />
od<br />
25.09.2007<br />
Str. 22 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
HEMOFARM AD, Beogradski<br />
put bb, Vr{ac, Srbija<br />
JUGOREMEDIJA A.D.<br />
ZRENJANIN,<br />
Pan~eva~ka bb,<br />
Zrenjanin, Srbija<br />
LEK FARMACEVTSKA<br />
DRU@BA D.D.,<br />
Verov{kova 57,<br />
Ljubljana, Slovenija<br />
Deutsche Homöopathie-Union<br />
DHU-Arzneimittel GmbH &<br />
Co. KG., Ottostrasse 24,<br />
76227 Karlsruhe, Nema~ka<br />
Deutsche Homöopathie-Union<br />
DHU-Arzneimittel GmbH &<br />
Co. KG., Ottostrasse 24,<br />
76227 Karlsruhe, Nema~ka<br />
IVAN^I] I SINOVI D.O.O.,<br />
Bra}e Trnini} bb, Stari<br />
Banovci, Srbija<br />
ALCON-COUVREUR N.V.,<br />
Rijksweg 14,<br />
Puurs, Belgija<br />
HEMOFARM AD, Beogradski<br />
put bb, Vr{ac, Srbija<br />
JUGOREMEDIJA A.D.<br />
ZRENJANIN, Pan~eva~ka bb,<br />
Zrenjanin, Srbija<br />
SANDOZ<br />
PHARMACEUTICALS D.D. -<br />
PREDSTAVNI[TVO,<br />
Kneginje Zorke 2,<br />
Beograd, Srbija<br />
LIBRA BGD D.O.O.,<br />
Obalskih radnika 4 g,<br />
Beograd, Srbija<br />
LIBRA BGD D.O.O.,<br />
Obalskih radnika 4 g,<br />
Beograd, Srbija<br />
IVAN^I] I SINOVI D.O.O.,<br />
Palmoti}eva 13,<br />
Beograd, Srbija<br />
ALCON<br />
PHARMACEUTICALS LTD -<br />
PREDSTAVNI[TVO,<br />
Gospodar Jevremova 59,<br />
Beograd, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Kalii chloridi<br />
kalijum-hlorid<br />
Kalijum hlorid Jadran<br />
kalijum-hlorid<br />
Kamagel®<br />
aluminijum-acetotartarat,<br />
kamilica (Matricaria<br />
Chamomilla), te~ni ekstrakt<br />
cvasti<br />
Kamfart®<br />
kamfor, metilsalicilat,<br />
benzilnikotinat, mentol<br />
Kansen®<br />
klotrimazol<br />
Ketonal®<br />
ketoprofen<br />
Kindinorm<br />
Calcium hypophosphorosum<br />
dil. D4, Chamomilla dil. D12,<br />
Cuprum metallicum dil. D10,<br />
Kalium phosphoricum dil.<br />
D6, Staphisagria dil. D12,<br />
Valeriana dil. D6<br />
Kofan® instant<br />
paracetamol,propifenazon,<br />
kofein<br />
Oblik Oblik do<strong>za</strong> do<strong>za</strong> do<strong>za</strong> i<br />
i<br />
pakovawe pakovawe leka<br />
leka<br />
oralni pra{ak; 1g; kesica, 10x1g<br />
tableta <strong>za</strong> oralni rastvor; 500mg;<br />
fiola, 1x20kom<br />
gel; 0.05g/g+0.01g/g; tuba,<br />
1x40g<br />
krem; 20mg/g+10mg/g+<br />
10mg/g+10mg/g;<br />
tuba, 1x40g<br />
krem; 1%; tuba, 1x20g<br />
gel; 2.5%; tuba, 1x50g<br />
granule; (0.0167g+0.0167g+<br />
0.0167g+0.0167g+0.0167g+<br />
0.0167g)/10g;<br />
bo~ica staklena, 1x10g<br />
tableta; 200mg+200mg+50mg;<br />
blister, 2x10kom<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa oo<br />
o<br />
stavqawu stavqawu stavqawu leka leka u u u promet<br />
promet<br />
1863/2007/12<br />
1179/2006/12<br />
27/2008/12<br />
2738/2007/12<br />
65/2009/12<br />
3376/2010/12<br />
4938/2009/12<br />
1208/2008/12<br />
od 04.09.2007<br />
od 13.07.2006<br />
od 11.01.2008<br />
od 08.11.2007<br />
od 19.01.2009<br />
od 14.06.2010<br />
od 21.09.2009<br />
od<br />
26.03.2008<br />
Str. 23 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
UFAR D.O.O.,<br />
Lipovi~ki Put 308 b,<br />
Beograd- Barajevo, Srbija<br />
JADRAN GALENSKI<br />
LABORATORIJ D.D.,<br />
Pulac bb,<br />
Rijeka, Hrvatska<br />
KRKA, TOVARNA ZDRAVIL,<br />
D.D., [marje{ka cesta 6,<br />
Novo Mesto, Slovenija<br />
BOSNALIJEK D.D.,<br />
Juki}eva 53, Sarajevo,<br />
Bosna i Hercegovina<br />
ZDRAVLJE A.D., Vlajkova<br />
199, Leskovac, Srbija<br />
SALUTAS PHARMA GMBH,<br />
Lange Göhren 3, Sülzetal,<br />
Osterweddingen, Nema~ka<br />
Deutsche Homöopathie-Union<br />
DHU-Arzneimittel GmbH &<br />
Co. KG., Ottostrasse 24,<br />
76227 Karlsruhe, Nema~ka<br />
BOSNALIJEK D.D.,<br />
Juki}eva 53, Sarajevo,<br />
Bosna i Hercegovina<br />
UFAR D.O.O.,<br />
Lipovi~ki Put 308 b,<br />
Beograd- Barajevo, Srbija<br />
JADRAN GALENSKI<br />
LABORATORIJ D.D -<br />
PREDSTAVNI[TVO,<br />
Mirijevski bulevar 37 a,<br />
Beograd, Srbija<br />
KRKA FARMA D.O.O.,<br />
Jurija Gagarina 26/V/II,<br />
Beograd, Srbija<br />
BOSNALIJEK D.D. -<br />
PREDSTAVNI[TVO,<br />
Cara Nikolaja II 61/B,<br />
Beograd, Srbija<br />
ZDRAVLJE A.D., Vlajkova<br />
199, Leskovac, Srbija<br />
SANDOZ<br />
PHARMACEUTICALS D.D. -<br />
PREDSTAVNI[TVO,<br />
Kneginje Zorke 2,<br />
Beograd, Srbija<br />
LIBRA BGD D.O.O.,<br />
Obalskih radnika 4 g,<br />
Beograd, Srbija<br />
BOSNALIJEK D.D. -<br />
PREDSTAVNI[TVO,<br />
Cara Nikolaja II 61/B,<br />
Beograd, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Kombikaf®<br />
acetilsalicilna kiselina,<br />
paracetamol, kofein<br />
Kombinovani pra{ak protiv<br />
bolova<br />
paracetamol, kofein<br />
Lamisil® Once 1%<br />
terbinafin<br />
Lekadol® Plus C<br />
paracetamol, askorbinska<br />
kiselina<br />
Linex®<br />
mle~nokiselinske bakterije,<br />
`ive liofilizovane<br />
Linola®<br />
masne kiseline, ne<strong>za</strong>si}ene<br />
Linola® Fett<br />
masne kiseline, ne<strong>za</strong>si}ene<br />
Linola® Fett N<br />
parafin, te~ni i masni<br />
proizvodi<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
oralni pra{ak;<br />
200mg+300mg+50mg; kesica,<br />
10x1kom<br />
oralni pra{ak;<br />
200mg+300mg+50mg; kesica,<br />
500x1kom<br />
oralni pra{ak; 400mg+50mg;<br />
kesica, 200x850mg<br />
oralni pra{ak; 400mg+50mg;<br />
kesica, 10x850mg<br />
rastvor <strong>za</strong> ko`u; 10mg/g;<br />
tuba, 1x4g<br />
granule <strong>za</strong> oralni rastvor;<br />
500mg+300mg; kesica, 10x5g<br />
kapsula, tvrda; 1.2x 10exp7;<br />
blister, 2x8kom<br />
kapsula, tvrda; 1.2x 10exp7;<br />
bo~ica, 1x16kom<br />
krem; 5mg/g; tuba, 1x50g<br />
krem; 8.15mg/g; tuba, 1x50g<br />
dodatak <strong>za</strong> kupku; 480mg/g+<br />
440mg/g+10mg/g+60mg/g;<br />
boca, 1x200ml<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
3237/2010/12<br />
3238/2010/12<br />
2082/2010/12<br />
3299/2010/12<br />
3606/2009/12<br />
8172/2009/12<br />
3842/2009/12<br />
654/2006/12<br />
763/2007/12<br />
923/2007/12<br />
922/2007/12<br />
od 04.06.2010<br />
od 04.06.2010<br />
od 16.03.2010<br />
od 07.06.2010<br />
od 29.06.2009<br />
od 17.12.2009<br />
od 03.08.2009<br />
od 17.05.2006<br />
od 02.04.2007<br />
od 04.05.2007<br />
od<br />
04.05.2007<br />
Str. 24 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
B.G.-PHARM D.O.O.,<br />
Milosava Vlaji}a 110A,<br />
Sopot, Srbija<br />
IVAN^I] I SINOVI D.O.O.,<br />
Bra}e Trnini} bb,<br />
Stari Banovci, Srbija<br />
NOVARTIS CONSUMER<br />
HEALTH SA, Route de<br />
l'Etraz, Nyon, [vajcarska<br />
LEK FARMACEVTSKA<br />
DRU@BA D.D.,<br />
Verov{kova 57,<br />
Ljubljana, Slovenija<br />
LEK FARMACEVTSKA<br />
DRU@BA D.D.,<br />
Verov{kova 57,<br />
Ljubljana, Slovenija<br />
DR. AUGUST WOLFF GMBH<br />
& CO. ARZNEIMITTEL,<br />
Sudbrackstrasse 56,<br />
Bielefeld, Nema~ka<br />
DR. AUGUST WOLFF GMBH<br />
& CO. ARZNEIMITTEL,<br />
Sudbrackstrasse 56,<br />
Bielefeld, Nema~ka<br />
DR. AUGUST WOLFF GMBH<br />
& CO. ARZNEIMITTEL,<br />
Sudbrackstrasse 56,<br />
Bielefeld, Nema~ka<br />
B.G-PHARM D.O.O.,<br />
Visokog Stevana 31,<br />
Beograd, Srbija<br />
IVAN^I] I SINOVI D.O.O.,<br />
Palmoti}eva 13,<br />
Beograd, Srbija<br />
MEDIS INTERNATIONAL<br />
D.O.O., Bulevar Mihaila<br />
Pupina 10d/1 pp7, Beograd -<br />
Novi Beograd, Srbija<br />
SANDOZ<br />
PHARMACEUTICALS D.D. -<br />
PREDSTAVNI[TVO,<br />
Kneginje Zorke 2,<br />
Beograd, Srbija<br />
SANDOZ<br />
PHARMACEUTICALS D.D. -<br />
PREDSTAVNI[TVO,<br />
Kneginje Zorke 2,<br />
Beograd, Srbija<br />
EUROFARM D.O.O.,<br />
Bo{ka Petrovi}a 3a,<br />
Beograd, Srbija<br />
EUROFARM D.O.O.,<br />
Bo{ka Petrovi}a 3a,<br />
Beograd, Srbija<br />
EUROFARM D.O.O.,<br />
Bo{ka Petrovi}a 3a,<br />
Beograd, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Linola® Gamma<br />
no}urak, (Oenothere spp),<br />
masno ulje<br />
Linola® Urea<br />
urea<br />
Lioton 1000<br />
heparin-natrijum<br />
Lysobact®<br />
lizozim-hidrohlorid,<br />
piridoksin<br />
Magne B6<br />
magnezijum-laktat,<br />
piridoksin<br />
Midol<br />
acetilsalicilna kiselina<br />
Midol C<br />
acetilsalicilna kiselina,<br />
askorbinska kiselina<br />
Milgamma® 100<br />
benfotiamin, piridoksin<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
krem; 200mg/g; tuba, 1x50g<br />
krem; 120mg/g; tuba, 1x50g<br />
gel; 1000i.j./g; tuba, 1x100g<br />
gel; 1000i.j./g; tuba, 1x50g<br />
gel; 1000i.j./g; tuba, 1x30g<br />
gel; 1000i.j./g; tuba, 1x20g<br />
komprimovana lozenga;<br />
20mg+10mg; blister, 3x10kom<br />
oblo`ena tableta; 470mg+5mg;<br />
blister, 5x10kom<br />
tableta; 100mg; blister, 2x10kom<br />
tableta; 300mg; blister, 2x10kom<br />
{ume}a tableta; 400mg+240mg;<br />
fiola, 1x10kom<br />
oblo`ena tableta;<br />
100mg+100mg; blister, 2x15kom<br />
oblo`ena tableta;<br />
100mg+100mg; blister, 4x15kom<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
764/2007/12<br />
1444/2007/12<br />
1087/2007/12<br />
1088/2007/12<br />
1089/2007/12<br />
1090/2007/12<br />
2062/2006/12<br />
597/2005/12<br />
4022/2009/12<br />
1504/2010/12<br />
122/2008/12<br />
78/2007/12<br />
80/2007/12<br />
od 02.04.2007<br />
od 06.07.2007<br />
od 23.05.2007<br />
od 23.05.2007<br />
od 23.05.2007<br />
od 23.05.2007<br />
od 13.11.2006<br />
od 22.02.2006<br />
od<br />
od<br />
29.07.2009<br />
01.03.2010<br />
od 22.01.2008<br />
od 11.01.2007<br />
od<br />
11.01.2007<br />
Str. 25 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
DR. AUGUST WOLFF GMBH<br />
& CO. ARZNEIMITTEL,<br />
Sudbrackstrasse 56,<br />
Bielefeld, Nema~ka<br />
DR. AUGUST WOLFF GMBH<br />
& CO. ARZNEIMITTEL,<br />
Sudbrackstrasse 56,<br />
Bielefeld, Nema~ka<br />
A.MENARINI,<br />
MANUFACTURING<br />
LOGISTICS AND SERVICES<br />
S.R.L., Via Sette Santi 3,<br />
Firenca, Italija<br />
BOSNALIJEK D.D., Juki}eva<br />
53, Sarajevo, Bosna i<br />
Hercegovina<br />
SANOFI WINTHROP<br />
INDUSTRIE, 1, rue de la<br />
Vierge, Ambares et Lagrave,<br />
Carbon Blanc, Francuska<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
MAUERMANN-<br />
ARZNEIMITTEL KG,<br />
Heinrich-Knote-Straße 2,<br />
Pöcking, Nema~ka <strong>za</strong><br />
WÖRWAG PHARMA GMBH<br />
& CO. KG, Calwer Straße 7,<br />
Böblingen, Nema~ka<br />
EUROFARM D.O.O.,<br />
Bo{ka Petrovi}a 3a,<br />
Beograd, Srbija<br />
EUROFARM D.O.O.,<br />
Bo{ka Petrovi}a 3a,<br />
Beograd, Srbija<br />
BERLIN-CHEMIE AG<br />
(MENARINI GROUP) -<br />
PREDSTAVNI[TVO,<br />
Prilepska 1,<br />
Beograd, Srbija<br />
BOSNALIJEK D.D. -<br />
PREDSTAVNI[TVO,<br />
Cara Nikolaja II 61/B,<br />
Beograd, Srbija<br />
SANOFI-AVENTIS D.O.O.,<br />
Vladimira Popovi}a 40/IV,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
WORWAG PHARMA GMBH<br />
& CO.KG -<br />
PREDSTAVNI[TVO,<br />
Zahumska 23 a/1,<br />
Beograd-Zvezdara, Srbija
Mucodyne®<br />
karbocistein<br />
Mucodyne®<br />
karbocistein<br />
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Mucofalk® Pomorand`a<br />
bokvica (Plantago ovata),<br />
semenja~a semena<br />
Mucoplant<br />
list mu{ke bokvice, te~ni<br />
ekstrakt<br />
Mucosolvan®<br />
ambroksol<br />
Mucosolvan®<br />
ambroksol<br />
Mucosolvan®<br />
ambroksol<br />
Mucosolvan® Junior<br />
ambroksol<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
sirup; 25mg/ml; bo~ica od<br />
tamnog stakla, 1x100ml<br />
sirup; 50mg/ml; boca od<br />
tamnog stakla, 1x200ml<br />
sirup; 50mg/ml; bo~ica od<br />
tamnog stakla, 1x100ml<br />
sirup; 25mg/ml; bo~ica od<br />
tamnog stakla, 1x200ml<br />
granule <strong>za</strong> oralnu suspenziju;<br />
3.25g/5g; kesica, 20x5g<br />
sirup; 5g/100g;<br />
boca staklena, 1x100ml<br />
sirup; 30mg/5ml;<br />
bo~ica staklena, 1x100ml<br />
tableta; 30mg; blister, 2x10kom<br />
sirup; 30mg/5ml;<br />
bo~ica staklena, 1x100ml<br />
sirup; 15mg/5ml;<br />
bo~ica staklena, 1x100ml<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
<strong>Agencija</strong> <strong>za</strong> <strong>lekove</strong> i <strong>medicinska</strong> <strong>sredstva</strong> <strong>Srbije</strong> Str. 26 od 42<br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
3309/2010/12<br />
3310/2010/12<br />
500/2006/12<br />
499/2006/12<br />
2321/2006/12<br />
3624/2010/12<br />
6578/2009/12<br />
1278/2006/12<br />
6541/2009/12<br />
6577/2009/12<br />
od 07.06.2010<br />
od 07.06.2010<br />
od<br />
od<br />
10.04.2006<br />
10.04.2006<br />
od 12.12.2006<br />
od 09.07.2010<br />
od 22.10.2009<br />
od 02.08.2006<br />
od 22.10.2009<br />
od<br />
22.10.2009<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
HEMOFARM D.O.O. [ABAC,<br />
Hajduk Veljkova bb,<br />
[abac, Srbija<br />
DR. FALK PHARMA GMBH,<br />
Leinenweberstrasse 5,<br />
Freiburg, Nema~ka<br />
DR. THEISS NATURWAREN<br />
GMBH, Michelinstrasse 10,<br />
Homburg, Nema~ka<br />
BOEHRINGER INGELHEIM<br />
ESPANA S.A., Prat de la<br />
Riba, s/n, Sant Cugat del<br />
Valles, Barcelona, [panija<br />
BOEHRINGER INGELHEIM<br />
HELLAS S.A., 2 Ellinikou,<br />
Elliniko, Atina, Gr~ka<br />
DELPHARM REIMS, 10 rue<br />
Colonel Charbonneaux,<br />
Reims, Francuska<br />
BOEHRINGER INGELHEIM<br />
ESPANA S.A., Prat de la<br />
Riba, s/n, Sant Cugat del<br />
Valles, Barcelona, [panija<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
SEPHORALEK DOO,<br />
Mile{evska 37,<br />
Beograd, Srbija<br />
SEPHORALEK DOO,<br />
Mile{evska 37,<br />
Beograd, Srbija<br />
BOEHRINGER INGELHEIM<br />
SERBIA D.O.O. BEOGRAD,<br />
Ljube Stojanovi}a 15,<br />
Po{tanski fah 365,<br />
Beograd, Srbija<br />
BOEHRINGER INGELHEIM<br />
SERBIA D.O.O. BEOGRAD,<br />
Ljube Stojanovi}a 15,<br />
Po{tanski fah 365,<br />
Beograd, Srbija<br />
BOEHRINGER INGELHEIM<br />
SERBIA D.O.O. BEOGRAD,<br />
Ljube Stojanovi}a 15,<br />
Po{tanski fah 365,<br />
Beograd, Srbija<br />
BOEHRINGER INGELHEIM<br />
SERBIA D.O.O. BEOGRAD,<br />
Ljube Stojanovi}a 15,<br />
Po{tanski fah 365,<br />
Beograd, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Mucosolvan® Junior<br />
ambroksol<br />
Mycoseb<br />
ketokonazol<br />
Naklofen®<br />
diklofenak<br />
nasic <strong>za</strong> decu<br />
ksilometazolin,<br />
dekspantenol<br />
nasic®<br />
ksilometazolin,<br />
dekspantenol<br />
Neo-angin®<br />
amilmetakrezol, 2,4dihlorbenzil<br />
alkohol,<br />
levomentol<br />
Neo-angin®<br />
amilmetakrezol, 2,4dihlorbenzil<br />
alkohol,<br />
levomentol<br />
Neo-angin® bez {e}era<br />
amilmetakrezol, 2,4dihlorbenzil<br />
alkohol,<br />
levomentol<br />
neo-bronchol®<br />
ambroksol<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
sirup; 15mg/5ml;<br />
bo~ica staklena, 1x100ml<br />
{ampon; 2%;<br />
bo~ica plasti~na, 1x100ml<br />
gel; 10mg/g; tuba, 1x60g<br />
sprej <strong>za</strong> nos, rastvor; 0.05%+5%;<br />
bo~ica sa pumpom <strong>za</strong> doziranje,<br />
1x10ml<br />
sprej <strong>za</strong> nos, rastvor; 0.1%+5%;<br />
bo~ica sa pumpom <strong>za</strong> doziranje,<br />
1x10ml<br />
pastila; 0.6mg+1.2mg+5.9mg;<br />
blister, 2x12kom<br />
sprej <strong>za</strong> usnu sluznicu;<br />
2.92mg/ml+14.58mg/ml+0.87mg/<br />
ml; boca staklena, 1x30ml<br />
pastila; 0.6mg+1.2mg+5.72mg;<br />
blister, 2x12kom<br />
lozenga; 15mg;<br />
blister, 2x10kom<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
6542/2009/12<br />
15/2010/12<br />
8001/2009/12<br />
252/2009/12<br />
251/2009/12<br />
640/2006/12<br />
2980/2010/12<br />
645/2006/12<br />
125/2010/12<br />
od 22.10.2009<br />
od 11.01.2010<br />
od 11.12.2009<br />
od 28.01.2009<br />
od 28.01.2010<br />
od 17.05.2006<br />
od 11.05.2010<br />
od 17.05.2006<br />
od<br />
15.01.2010<br />
Str. 27 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
DELPHARM REIMS, 10 rue<br />
Colonel Charbonneaux,<br />
Reims, Francuska<br />
HEMOFARM AD, Beogradski<br />
put bb, Vr{ac, Srbija<br />
KRKA, TOVARNA ZDRAVIL,<br />
D.D., [marje{ka cesta 6,<br />
Novo Mesto, Slovenija<br />
KLOSTERFRAU BERLIN<br />
GMBH, Motzener Str. 41,<br />
Berlin, Nema~ka<br />
KLOSTERFRAU BERLIN<br />
GMBH, Motzener Str. 41,<br />
Berlin, Nema~ka<br />
DIVAPHARMA GMBH,<br />
Motzener Str. 41,<br />
Berlin, Nema~ka<br />
DIVAPHARMA GMBH,<br />
Motzener Str. 41,<br />
Berlin, Nema~ka<br />
DIVAPHARMA GMBH,<br />
Motzener Str. 41,<br />
Berlin, Nema~ka<br />
DIVAPHARMA GMBH,<br />
Motzener Str. 41,<br />
Berlin, Nema~ka<br />
BOEHRINGER INGELHEIM<br />
SERBIA D.O.O. BEOGRAD,<br />
Ljube Stojanovi}a 15,<br />
Po{tanski fah 365,<br />
Beograd, Srbija<br />
HEMOFARM AD, Beogradski<br />
put bb, Vr{ac, Srbija<br />
KRKA FARMA D.O.O.,<br />
Jurija Gagarina 26/V/II,<br />
Beograd, Srbija<br />
CLINRES FARMACIJA<br />
D.O.O., Molerova 36/III,<br />
Beograd, Srbija<br />
CLINRES FARMACIJA<br />
D.O.O., Molerova 36/III,<br />
Beograd, Srbija<br />
POLIFARM D.O.O.,<br />
Zlatiborska 2V,<br />
Beograd, Srbija<br />
POLIFARM D.O.O.,<br />
Zlatiborska 2V,<br />
Beograd, Srbija<br />
POLIFARM D.O.O.,<br />
Zlatiborska 2V,<br />
Beograd, Srbija<br />
POLIFARM D.O.O.,<br />
Zlatiborska 2V,<br />
Beograd, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Nicorette® Freshfruit<br />
nikotin<br />
Nicorette® Freshmint<br />
nikotin<br />
Nicorette® Microtab Lemon<br />
nikotin<br />
NiQuitin®<br />
nikotin<br />
Normogin<br />
Liofilizirana kultura<br />
izolovanog soja Doederlein<br />
bacillus (Lactobacillus<br />
rhamnosus)<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
lekovita guma <strong>za</strong> `vakanje; 4mg;<br />
blister, 7x15kom<br />
lekovita guma <strong>za</strong> `vakanje; 2mg;<br />
blister, 7x15kom<br />
lekovita guma <strong>za</strong> `vakanje; 4mg;<br />
blister, 2x15kom<br />
lekovita guma <strong>za</strong> `vakanje; 2mg;<br />
blister, 2x15kom<br />
lekovita guma <strong>za</strong> `vakanje; 4mg;<br />
blister, 7x15kom<br />
lekovita guma <strong>za</strong> `vakanje; 4mg;<br />
blister, 2x15kom<br />
lekovita guma <strong>za</strong> `vakanje; 2mg;<br />
blister, 7x15kom<br />
lekovita guma <strong>za</strong> `vakanje; 2mg;<br />
blister, 2x15kom<br />
sublingvalna tableta; 2mg;<br />
blister, 3x10kom<br />
sublingvalna tableta; 4mg;<br />
blister, 3x10kom<br />
sublingvalna tableta; 4mg;<br />
blister, 10x10kom<br />
sublingvalna tableta; 2mg;<br />
blister, 10x10kom<br />
transdermalni flaster; 21mg/24h;<br />
kesica, 7x1kom<br />
transdermalni flaster; 14mg/24h;<br />
kesica, 7x1kom<br />
transdermalni flaster; 7mg/24h;<br />
kesica, 7x1kom<br />
vaginalna tableta; 40mg;<br />
bo~ica staklena, 1x6kom<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
1511/2009/12<br />
1513/2009/12<br />
1514/2009/12<br />
1515/2009/12<br />
1510/2009/12<br />
1517/2009/12<br />
1516/2009/12<br />
1512/2009/12<br />
1518/2009/12<br />
1519/2009/12<br />
1520/2009/12<br />
1521/2009/12<br />
3592/2009/12<br />
3591/2009/12<br />
3590/2009/12<br />
1492/2009/12<br />
od 11.05.2009<br />
od 11.05.2009<br />
od 11.05.2009<br />
od 11.05.2009<br />
od 11.05.2009<br />
od 11.05.2009<br />
od 11.05.2009<br />
od 11.05.2009<br />
od 11.05.2009<br />
od 11.05.2009<br />
od 11.05.2009<br />
od 11.05.2009<br />
od 29.06.2009<br />
od 29.06.2009<br />
od 29.06.2009<br />
od<br />
11.05.2009<br />
Str. 28 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
MCNEIL AB,<br />
Norrbroplatsen 2,<br />
Helsingborg, [vedska<br />
MCNEIL AB,<br />
Norrbroplatsen 2,<br />
Helsingborg, [vedska<br />
MCNEIL AB,<br />
Norrbroplatsen 2,<br />
Helsingborg, [vedska<br />
CATALENT UK PACKAGING<br />
LIMITED, Sedge Close,<br />
Great Oakley, Corby,<br />
Northamptonshire,<br />
Velika Britanija<br />
LABORATORI BALDACCI<br />
S.P.A., Via S. Michele degli<br />
Scalzi 73, Pisa, Italija<br />
JOHNSON & JOHNSON S.E.<br />
INC.- PREDSTAVNI[TVO,<br />
Omladinskih brigada 88 b,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
JOHNSON & JOHNSON S.E.<br />
INC.- PREDSTAVNI[TVO,<br />
Omladinskih brigada 88 b,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
JOHNSON & JOHNSON S.E.<br />
INC.- PREDSTAVNI[TVO,<br />
Omladinskih brigada 88 b,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
GLAXOSMITHKLINE<br />
EXPORT LIMITED -<br />
PREDSTAVNI[TVO,<br />
Omladinskih brigada 88,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
MITER INTERNATIONAL<br />
D.O.O., Resavska 16,<br />
Beograd, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Octenisept®<br />
oktenidin, fenoksietanol<br />
Oldon®<br />
acetilsalicilna kiselina,<br />
paracetamol, kofein<br />
Olynth®<br />
ksilometazolin<br />
Olynth® HA<br />
ksilometazolin<br />
Operil®<br />
oksimetazolin<br />
Operil® P<br />
oksimetazolin<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
rastvor <strong>za</strong> ko`u; 1mg/g+20mg/g;<br />
boca plasti~na, 1x250ml<br />
rastvor <strong>za</strong> ko`u; 1mg/g+20mg/g;<br />
boca plasti~na, 1x1000ml<br />
rastvor <strong>za</strong> ko`u; 1mg/g+20mg/g;<br />
boca plasti~na, 1x50ml<br />
tableta; 300mg+200mg+50mg;<br />
strip, 100x10kom<br />
sprej <strong>za</strong> nos, rastvor; 0.1%;<br />
bo~ica sa raspr{iva~em, 1x10ml<br />
kapi <strong>za</strong> nos, rastvor; 0.1%;<br />
bo~ica sa kapaljkom, 1x10ml<br />
kapi <strong>za</strong> nos, rastvor; 0.05%;<br />
bo~ica sa kapaljkom, 1x10ml<br />
sprej <strong>za</strong> nos, rastvor; 0.05%;<br />
bo~ica sa raspr{iva~em, 1x10ml<br />
sprej <strong>za</strong> nos, rastvor; 0.1%;<br />
bo~ica sa pumpom <strong>za</strong> doziranje,<br />
1x10ml<br />
sprej <strong>za</strong> nos, rastvor; 0.05%;<br />
sprej-boca sa dozerom, 1x10ml<br />
kapi <strong>za</strong> nos, rastvor; 0.05%;<br />
bo~ica sa kapaljkom, 1x10ml<br />
sprej <strong>za</strong> nos, rastvor; 0.025%;<br />
sprej-boca sa dozerom, 1x10ml<br />
kapi <strong>za</strong> nos, rastvor; 0.025%;<br />
bo~ica sa kapaljkom, 1x10ml<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
3630/2009/12<br />
3629/2009/12<br />
3631/2009/12<br />
1111/2007/12<br />
644/2008/12<br />
643/2008/12<br />
645/2008/12<br />
646/2008/12<br />
4065/2010/12<br />
1171/2006/12<br />
3806/2009/12<br />
1170/2006/12<br />
3807/2009/12<br />
od 30.06.2009<br />
od 30.06.2009<br />
od 30.06.2009<br />
od 28.05.2007<br />
od 21.02.2008<br />
od 21.02.2008<br />
od 21.02.2008<br />
od 21.02.2008<br />
od 12.08.2010<br />
od 13.07.2006<br />
od 09.07.2009<br />
od 13.07.2006<br />
od<br />
09.07.2009<br />
Str. 29 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
SCHÜLKE & MAYR GmbH,<br />
Robert-Koch Strasse 2,<br />
Norderstedt, Nema~ka<br />
ZDRAVLJE A.D.,<br />
Vlajkova 199,<br />
Leskovac, Srbija<br />
FAMAR ORLEANS, 5,<br />
avenue de Concyr,<br />
Orleans, Francuska<br />
URSAPHARM<br />
ARZNEIMITTEL GMBH &<br />
CO. KG, Industriestraße,<br />
Saarbrücken, Nema~ka<br />
LEK FARMACEVTSKA<br />
DRU@BA D.D.,<br />
Verov{kova 57,<br />
Ljubljana, Slovenija<br />
LEK FARMACEVTSKA<br />
DRU@BA D.D.,<br />
Verov{kova 57,<br />
Ljubljana, Slovenija<br />
MIOFARM D.O.O.,<br />
Stevana Musi}a 17,<br />
Novi Sad, Srbija<br />
ZDRAVLJE A.D.,<br />
Vlajkova 199,<br />
Leskovac, Srbija<br />
JOHNSON & JOHNSON S.E.<br />
INC.- PREDSTAVNI[TVO,<br />
Omladinskih brigada 88 b,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
JOHNSON & JOHNSON S.E.<br />
INC.- PREDSTAVNI[TVO,<br />
Omladinskih brigada 88 b,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
SANDOZ<br />
PHARMACEUTICALS D.D. -<br />
PREDSTAVNI[TVO,<br />
Kneginje Zorke 2,<br />
Beograd, Srbija<br />
SANDOZ<br />
PHARMACEUTICALS D.D. -<br />
PREDSTAVNI[TVO,<br />
Kneginje Zorke 2,<br />
Beograd, Srbija
Panadol®<br />
paracetamol<br />
Panadol®<br />
paracetamol<br />
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Panadol® Baby<br />
paracetamol<br />
Panadol® Extra<br />
paracetamol, kofein<br />
Pankreatin<br />
pankreas pra{ak<br />
Pantenol<br />
kalcijum-pantotenat<br />
Panthenol<br />
kalcijum-pantotenat<br />
Pantogar<br />
medicinski kvasac, tiamin,<br />
kalcijum-pantotenat, cistin,<br />
para-aminobenzoeva<br />
kiselina, keratin<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
film tableta; 500mg;<br />
blister, 1x12kom<br />
film tableta; 500mg;<br />
blister, 2x12kom<br />
film tableta; 500mg;<br />
blister, 1x12kom<br />
film tableta; 500mg;<br />
blister, 2x12kom<br />
oralna suspenzija; 120mg/5ml;<br />
bo~ica staklena, 1x100ml<br />
film tableta; 500mg+65mg;<br />
blister, 2x12kom<br />
film tableta; 500mg+65mg;<br />
blister, 1x12kom<br />
gastrorezistentna tableta;<br />
125mg; blister, 5x10kom<br />
oralna disperzibilna tableta;<br />
120mg; blister, 2x10kom<br />
komprimovana lozenga; 120mg;<br />
blister, 2x10kom<br />
mast; 5%; tuba, 1x30g<br />
kapsula, tvrda; 100mg+60mg+<br />
60mg+20mg+20mg+20mg;<br />
blister, 6x15kom<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
515/2009/12<br />
516/2009/12<br />
4044/2010/12<br />
4045/2010/12<br />
4511/2008/12<br />
517/2009/12<br />
518/2009/12<br />
112/2006/12<br />
6609/2010/12<br />
1237/2008/12<br />
1238/2008/12<br />
1114/2008/12<br />
od 17.02.2009<br />
od 17.02.2009<br />
od 11.08.2010<br />
od 11.08.2010<br />
od 15.12.2008<br />
od 17.02.2009<br />
od 17.02.2009<br />
od 02.02.2006<br />
od 26.11.2010<br />
od 28.03.2008<br />
od 28.03.2008<br />
od<br />
18.03.2008<br />
Str. 30 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
GLAXOSMITHKLINE<br />
DUNGARVAN LTD,<br />
Knockbrack, Dungarvan,<br />
Co.Waterford, Irska<br />
S.C. EUROPHARM S.A.,<br />
2 Panselelor St, Brasov,<br />
County of Brasov, Rumunija<br />
FARMACLAIR, 440, Avenue<br />
de General de Gaulle,<br />
Herouville-Saint-Clair,<br />
Francuska<br />
GLAXOSMITHKLINE<br />
DUNGARVAN LTD,<br />
Knockbrack, Dungarvan,<br />
Co.Waterford, Irska<br />
GALENIKA AD, Batajni~ki<br />
drum b.b., Beograd, Srbija<br />
M.D. NINI D.O.O.,<br />
Vazduhoplovaca b.b.,<br />
Ni{, Srbija<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
Merz Pharma Gmbh & Co.<br />
KGaA, Eckenheimer Landstr.<br />
100, D-60318 Frankfurt,<br />
Nema~ka<br />
GLAXOSMITHKLINE<br />
EXPORT LIMITED -<br />
PREDSTAVNI[TVO,<br />
Omladinskih brigada 88,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
GLAXOSMITHKLINE<br />
EXPORT LIMITED -<br />
PREDSTAVNI[TVO,<br />
Omladinskih brigada 88,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
GLAXOSMITHKLINE<br />
EXPORT LIMITED -<br />
PREDSTAVNI[TVO,<br />
Omladinskih brigada 88,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
GLAXOSMITHKLINE<br />
EXPORT LIMITED -<br />
PREDSTAVNI[TVO,<br />
Omladinskih brigada 88,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
GALENIKA AD, Batajni~ki<br />
drum b.b., Beograd, Srbija<br />
M.D. NINI D.O.O.,<br />
Vazduhoplovaca b.b.,<br />
Ni{, Srbija<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
LIBRA BGD D.O.O.,<br />
Obalskih radnika 4 g,<br />
Beograd, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Paracetamol<br />
paracetamol<br />
Paracetamol<br />
paracetamol<br />
Paracetamol Sopharma<br />
paracetamol<br />
Pepsane<br />
dimetikon, gvajazulen<br />
Perigona®<br />
gluko<strong>za</strong>min<br />
Persen®<br />
valerijana (Valeriana<br />
officinalis), suvi ekstrakt<br />
korena, mati~njak (Melissa<br />
officinalis), suvi ekstrakt<br />
lista, nana (Mentha piperita),<br />
suvi ekstrakt lista<br />
Persen® forte<br />
valerijana (Valeriana<br />
officinalis), suvi ekstrakt<br />
korena, mati~njak (Melissa<br />
officinalis), suvi ekstrakt<br />
lista, nana (Mentha piperita),<br />
suvi ekstrakt lista<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
tableta; 500mg; blister, 3x6kom<br />
sirup; 120mg/5ml;<br />
bo~ica staklena, 1x100ml<br />
tableta; 500mg;<br />
blister, 1x10kom<br />
sirup; 120mg/5ml;<br />
bo~ica staklena, 1x125ml<br />
sirup; 120mg/5ml;<br />
bo~ica plasti~na, 1x125ml<br />
oralni gel; (3g+0.004g)/10g;<br />
kesica, 30x10g<br />
kapsula, meka; 300mg+4mg;<br />
blister, 3x10kom<br />
film tableta; 500mg;<br />
blister, 10x10kom<br />
film tableta; 500mg;<br />
blister, 2x10kom<br />
oblo`ena tableta;<br />
50mg+25mg+25mg;<br />
blister, 4x10kom<br />
kapsula, tvrda;<br />
125mg+25mg+25mg;<br />
blister, 2x10kom<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
3170/2010/12<br />
3466/2010/12<br />
6614/2010/12<br />
237/2009/12<br />
4650/2009/12<br />
2208/2010/12<br />
2209/2010/12<br />
4025/2009/12<br />
4026/2009/12<br />
189/2007/12<br />
1769/2006/12<br />
od 31.05.2010<br />
od 01.07.2010<br />
od 24.11.2010<br />
od 28.01.2009<br />
od 03.09.2009<br />
od 24.03.2010<br />
od 24.03.2010<br />
od 29.07.2009<br />
od 29.07.2009<br />
od 16.01.2007<br />
od<br />
02.10.2006<br />
Str. 31 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
GALENIKA AD, Batajni~ki<br />
drum b.b., Beograd, Srbija<br />
JUGOREMEDIJA A.D.<br />
ZRENJANIN, Pan~eva~ka bb,<br />
Zrenjanin, Srbija<br />
SOPHARMA PLC,<br />
16, Iliensko Shosse Str.,<br />
Sofija, Bugarska<br />
LABORATOIRES ROSA-<br />
PHYTO PHARMA, 2 avenue<br />
du Traité de Rome, Chatou,<br />
Francuska<br />
CHEPHASAAR CHEMISCH -<br />
PHARMAZEUTISCHE<br />
FABRIK GmbH, Muhlstrasse<br />
50, St. Ingbert, Nema~ka<br />
LEK FARMACEVTSKA<br />
DRU@BA D.D.,<br />
Verov{kova 57,<br />
Ljubljana, Slovenija<br />
LEK FARMACEVTSKA<br />
DRU@BA D.D.,<br />
Verov{kova 57,<br />
Ljubljana, Slovenija<br />
GALENIKA AD, Batajni~ki<br />
drum b.b., Beograd, Srbija<br />
JUGOREMEDIJA A.D.<br />
ZRENJANIN, Pan~eva~ka bb,<br />
Zrenjanin, Srbija<br />
SOPHARMA -<br />
PREDSTAVNI[TVO,<br />
Kumodra{ka 235,<br />
Beograd, Srbija<br />
NEW MED D.O.O., Jurija<br />
Gagarina 150/47, Beograd -<br />
Novi Beograd, Srbija<br />
MIP PHARMA D.O.O.,<br />
Ugrinova~ki put 16a,<br />
Beograd, Srbija<br />
SANDOZ<br />
PHARMACEUTICALS D.D. -<br />
PREDSTAVNI[TVO,<br />
Kneginje Zorke 2,<br />
Beograd, Srbija<br />
SANDOZ<br />
PHARMACEUTICALS D.D. -<br />
PREDSTAVNI[TVO,<br />
Kneginje Zorke 2,<br />
Beograd, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Pervivo®<br />
crni jasen (Fraxinus ornus),<br />
etanolni ekstrakt soka,<br />
kamfor, isiot (Curcuma<br />
zedoaria), etanolni ekstrakt<br />
korena, angelika (Angelica<br />
archangelica), etanolni<br />
ekstrakt korena, mira<br />
(Commiphora spp.), etanolni<br />
ekstrakt stabla, grane,<br />
lincura (Gentiana lutea),<br />
etanolni ekstrakt korena,<br />
morski ora{~i} (Myristica<br />
fragrans), etanolni ekstrakt<br />
semena, kravljak (Carlina<br />
acaulis), etanolni ekstrakt<br />
korena, slatki koren<br />
(Glycyrrhi<strong>za</strong> glabra),<br />
etanolni ekstrakt korena,<br />
oman (Inula helenium),<br />
etanolni ekstrakt korena,<br />
ki~ica (Centaurium spp.),<br />
etanolni ekstrakt herbe,<br />
karanfili} (Eugenia<br />
caryophyllus), etanolni<br />
ekstrakt cveta, galanga<br />
(Alpinia officinarum),<br />
etanolni ekstrakt korena,<br />
|umbir (Zingiber officinale),<br />
etanolni ekstrakt rizoma,<br />
bla`eni ~kalj (Cnicus<br />
benedictus), etanolni<br />
ekstrakt herbe, mo{us (Iva<br />
moschata), etanolni ekstrakt<br />
herbe, teriak, etanolni<br />
ekstrakt, perunika (Iris spp.),<br />
etanolni ekstrakt korena,<br />
divizma (Verbasci spp.),<br />
etanolni ekstrakt cveta,<br />
gorka narand`a (Aurantii<br />
amari), etanolni ekstrakt<br />
kore, i|irot (Acorus<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
oralni rastvor; 6 .8mg/5ml+4.75mg/5ml<br />
+6.9mg/5ml+ 6.8mg/5ml+3.5mg/5ml+<br />
2.5mg/5ml+1.4mg/5ml+3.4mg/5ml+<br />
0.85mg/5ml+0.1mg/5ml+0.065mg/<br />
5ml+0.15mg/5ml+0.07mg/5ml+<br />
0.075mg/5ml+0.075mg/5ml+0.03mg/<br />
5ml+4.85mg/5ml+0.025mg/5ml+<br />
0.07mg/5ml+0.155mg/5ml+0.235mg/<br />
5ml+0.175mg/5ml+0.19mg/5ml+<br />
0.085mg/5ml+0.23mg/5ml+0.055mg/<br />
5ml+0.6mg/5ml; boca, 1x500ml<br />
oralni rastvor; 6.8mg/5ml+4.75mg/5ml<br />
+6.9mg/5ml+6.8mg/5ml+3.5mg/5ml+<br />
2.5mg/5ml+1.4mg/5ml+3.4mg/5ml+<br />
0.85mg/5ml+0.1mg/5ml+0.065mg/<br />
5ml+0.15mg/5ml+0.07mg/5ml+<br />
0.075mg/5ml+0.075mg/5ml+0.03mg/<br />
5ml+4.85mg/5ml+0.025mg/5ml+<br />
0.07mg/5ml+0.155mg/5ml+0.235mg/<br />
5ml+0.175mg/5ml+0.19mg/5ml+<br />
0.085mg/5ml+0.23mg/5ml+0.055mg/<br />
5ml+0.6mg/5ml; bo~ica, 1x50ml<br />
oralni rastvor; 6.8mg/5ml+4.75mg/5ml<br />
+6.9mg/5ml+6.8mg/5ml+3.5mg/5ml+<br />
2.5mg/5ml+1.4mg/5ml+3.4mg/5ml+<br />
0.85mg/5ml+0.1mg/5ml+0.065mg/<br />
5ml+0.15mg/5ml+0.07mg/5ml+<br />
0.075mg/5ml+0.075mg/5ml+0.03mg/<br />
5ml+4.85mg/5ml+0.025mg/5ml+<br />
0.07mg/5ml+0.155mg/5ml+0.235mg/<br />
5ml+0.175mg/5ml+0.19mg/5ml+<br />
0.085mg/5ml+0.23mg/5ml+0.055mg/<br />
5ml+0.6mg/5ml; boca, 1x250ml<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
781/2007/12<br />
783/2007/12<br />
782/2007/12<br />
od 04.04.2007<br />
od 04.04.2007<br />
od<br />
04.04.2007<br />
Str. 32 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
RICHARD BITTNER AG.,<br />
Weitensfeld 183,<br />
Weitensfeld, Austrija<br />
FARMALOGIST D.O.O.,<br />
Bulevar Vojvode<br />
Mi{i}a 25- 27,<br />
Beograd, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
calamus), etanolni ekstrakt<br />
korena, beli pelen (Artemisia<br />
absinthium), etanolni<br />
ekstrakt herbe, gorka<br />
narand`a (Citrus aurantium),<br />
etanolni ekstrakt kore, biber<br />
kubebe (Piper cubeba),<br />
etanolni ekstrakt ploda,<br />
gorka narand`a (Aurantii<br />
dulcis), etanolni ekstrakt<br />
kore, zvezdasti anis (Anisi<br />
stellati), etanolni ekstrakt<br />
ploda, gorka narand`a<br />
(Aurantii dulcis), etanolni<br />
ekstrakt kore, gorka detelina<br />
(Menyanthes trifoliata),<br />
etanolni ekstrakt lista<br />
Piascledine®<br />
ulje soje, nesaponifikovano,<br />
ulje avokada,<br />
nesaponifikovano<br />
Pikovit® forte<br />
retinol, holekalciferol,<br />
askorbinska kiselina, tiamin,<br />
riboflavin, piridoksin,<br />
cijanokobalamin,<br />
nikotinamid, folna kiselina,<br />
kalcijum-pantotenat,<br />
tokoferol<br />
Pilfud®<br />
minoksidil<br />
Polinol<br />
Luffa operculata, triturat D4,<br />
Galphimia glauca, triturat<br />
D3, Cardiospermum, triturat<br />
D3<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
kapsula, tvrda; 200mg+100mg;<br />
blister, 1x15kom<br />
oblo`ena tableta; 5000i.j.+400i.j.+<br />
60mg+1.5mg+1.7mg+2mg+6mcg<br />
+20mg+0.4mg+10mg+15mg;<br />
blister, 3x10kom<br />
oblo`ena tableta; 5000i.j.+400i.j.+<br />
60mg+1.5mg+1.7mg+2mg+6mcg<br />
+20mg+0.4mg+10mg+15mg;<br />
blister, 2x15kom<br />
rastvor <strong>za</strong> ko`u; 2%;<br />
bo~ica, 1x60ml<br />
sprej <strong>za</strong> ko`u, rastvor; 5%;<br />
bo~ica sa raspr{iva~em, 1x60ml<br />
tableta; 25mg+25mg+25mg;<br />
blister, 5x20kom<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
3192/2010/12<br />
87/2007/12<br />
3581/2008/12<br />
81/2007/12<br />
82/2007/12<br />
5/2006/1500<br />
od 02.06.2010<br />
od 11.01.2007<br />
od 07.10.2008<br />
od 11.01.2007<br />
od 11.01.2007<br />
od<br />
10.02.2006<br />
Str. 33 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
LABORATOIRES<br />
EXPANSCIENCE,<br />
Rue des quatre filles,<br />
Epernon, Francuska<br />
KRKA, TOVARNA ZDRAVIL,<br />
D.D., [marje{ka cesta 6,<br />
Novo Mesto, Slovenija<br />
BOSNALIJEK D.D.,<br />
Juki}eva 53, Sarajevo,<br />
Bosna i Hercegovina<br />
Deutsche Homöopathie-Union<br />
DHU-Arzneimittel GmbH &<br />
Co. KG., Ottostrasse 24,<br />
76227 Karlsruhe, Nema~ka<br />
BONIFAR D.O.O., Bulevar<br />
Zorana \in|i}a 87/1,<br />
Beograd, Srbija<br />
KRKA FARMA D.O.O.,<br />
Jurija Gagarina 26/V/II,<br />
Beograd, Srbija<br />
BOSNALIJEK D.D. -<br />
PREDSTAVNI[TVO,<br />
Cara Nikolaja II 61/B,<br />
Beograd, Srbija<br />
LIBRA BGD D.O.O.,<br />
Obalskih radnika 4 g,<br />
Beograd, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Polivit B<br />
tiamin, riboflavin, piridoksin,<br />
nikotinamid, kalcijumpantotenat,<br />
cijanokobalamin,<br />
paba<br />
Postinor®-2<br />
levonorgestrel<br />
Povidon jod<br />
povidon-jod<br />
Povidon jod<br />
povidon-jod<br />
Prospan kapi<br />
br{ljan (Hedere helix),<br />
standardizovani suvi<br />
ekstrakt lista<br />
Prospan®<br />
br{ljan (Hedere helix),<br />
standardizovani suvi<br />
ekstrakt lista<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
film tableta; 5mg+5mg+2mg+<br />
25mg+5mg+0.001mg+2mg;<br />
blister, 3x10kom<br />
tableta; 0.75mg; blister,<br />
1x2kom<br />
pena <strong>za</strong> ko`u; 7.5%;<br />
kontejner plasti~ni, 1x500ml<br />
mast; 10%; tuba, 1x40g<br />
rastvor <strong>za</strong> ko`u; 10%;<br />
kontejner plasti~ni, 1x500ml<br />
vagitorija; 200mg;<br />
blister, 2x7kom<br />
rastvor <strong>za</strong> ispiranje usta; 8.5%;<br />
kontejner plasti~ni, 1x50ml<br />
pena <strong>za</strong> ko`u; 7.5%;<br />
kontejner plasti~ni, 1x5000ml<br />
rastvor <strong>za</strong> ko`u; 10%;<br />
kontejner plasti~ni, 1x5000ml<br />
rastvor <strong>za</strong> ko`u; 10%;<br />
kontejner plasti~ni, 1x100ml<br />
oralne kapi, rastvor; 20mg/ml;<br />
bo~ica sa kapaljkom, 1x20ml<br />
sirup; 7mg/ml; bo~ica, 1x100ml<br />
{ume}a tableta; 65mg;<br />
strip, 5x2kom<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
2525/2007/12<br />
757/2009/12<br />
3311/2010/12<br />
4248/2010/12<br />
3315/2010/12<br />
3314/2010/12<br />
3313/2010/12<br />
3312/2010/12<br />
763/2006/12<br />
760/2006/12<br />
258/2006/12<br />
1356/2006/12<br />
od 22.10.2007<br />
od 04.03.2009<br />
od 07.06.2010<br />
od 14.09.2010<br />
od 07.06.2010<br />
od 07.06.2010<br />
od 07.06.2010<br />
od 07.06.2010<br />
od 05.06.2006<br />
od 02.06.2006<br />
od 06.03.2006<br />
od 08.08.2006<br />
1355/2006/12 od 08.08.2006<br />
Str. 34 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
GEDEON RICHTER PLC,<br />
Gyömrõi út. 19-21,<br />
Budimpe{ta, Ma|arska<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
HEMOFARM D.O.O. [ABAC,<br />
Hajduk Veljkova bb,<br />
[abac, Srbija<br />
Engelhard Arzneimittel GmbH<br />
& Co.KG, Herzbergstrasse 3,<br />
Niederdorfelden, Nema~ka<br />
Engelhard Arzneimittel GmbH<br />
& Co.KG, Herzbergstrasse 3,<br />
Niederdorfelden, Nema~ka<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
RICHTER GEDEON NYRT-<br />
PREDSTAVNI[TVO,<br />
Vladimira Popovi}a 6,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
LIBRA BGD D.O.O.,<br />
Obalskih radnika 4 g,<br />
Beograd, Srbija<br />
LIBRA BGD D.O.O.,<br />
Obalskih radnika 4 g,<br />
Beograd, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Prostamol® uno<br />
Serenoa repens, etanolni<br />
ekstrakt 9-11:1<br />
Rapidol®<br />
ibuprofen<br />
Rapten rapid<br />
diklofenak<br />
Remens®<br />
cimicifuga racemosa trit. D1,<br />
sanguinaria canadensis trit.<br />
D6, jaborandi trit. D6, sepia<br />
officinalis trit. D12, lachesis<br />
mutus trit. D12<br />
Rheumon®<br />
etofenamat<br />
Rinasek<br />
pseudoefedrin, triprolidin<br />
Rinofluimucil®<br />
acetilcistein, tuaminoheptan<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
kapsula, meka; 320mg;<br />
blister, 2x15kom<br />
kapsula, meka; 320mg;<br />
blister, 1x15kom<br />
kapsula, meka; 320mg;<br />
blister, 4x15kom<br />
film tableta; 200mg;<br />
blister, 3x10kom<br />
kapsula, meka; 200mg;<br />
blister, 2x10kom<br />
oblo`ena tableta;<br />
12.5mg; blister, 1x10kom<br />
tableta;<br />
17.9mg+37.2mg+37.2mg+37.2mg<br />
+37.2mg; blister, 1x12kom<br />
tableta;<br />
17.9mg+37.2mg+37.2mg+37.2mg<br />
+37.2mg; blister, 3x12kom<br />
tableta;<br />
17.9mg+37.2mg+37.2mg+37.2mg<br />
+37.2mg; blister, 4x12kom<br />
oralne kapi, rastvor;<br />
(5ml+10ml+10ml+10ml+10ml)/100<br />
ml; bo~ica sa kapaljkom, 1x50ml<br />
gel; 5%; tuba, 1x40g<br />
tableta; 60mg+2.5mg;<br />
blister, 1x10kom<br />
sirup; 30mg/5ml+1.25mg/5ml;<br />
bo~ica staklena, 1x100ml<br />
sprej <strong>za</strong> nos, rastvor; 1%+0.5%;<br />
bo~ica, 1x10ml<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
<strong>Agencija</strong> <strong>za</strong> <strong>lekove</strong> i <strong>medicinska</strong> <strong>sredstva</strong> <strong>Srbije</strong> Str. 35 od 42<br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
301/2009/12<br />
300/2009/12<br />
302/2009/12<br />
3934/2008/12<br />
4736/2009/12<br />
286/2010/12<br />
6302/2010/12<br />
6303/2010/12<br />
6304/2010/12<br />
6305/2010/12<br />
571/2007/12<br />
7/2010/12<br />
8/2010/12<br />
2752/2007/12<br />
od 06.02.2009<br />
od 06.02.2009<br />
od 06.02.2009<br />
od 23.10.2008<br />
od 14.09.2009<br />
od 20.01.2010<br />
od 03.11.2010<br />
od 03.11.2010<br />
od 03.11.2010<br />
od 03.11.2010<br />
od 01.03.2007<br />
od 11.01.2010<br />
od 11.01.2010<br />
od<br />
08.11.2007<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
BERLIN-CHEMIE AG<br />
(MENARINI GROUP),<br />
Glienicker Weg 125,<br />
Berlin, Nema~ka<br />
PHARMASWISS D.O.O.,<br />
Milorada Jovanovi}a 9,<br />
Beograd, Srbija<br />
HEMOFARM AD, Beogradski<br />
put bb, Vr{ac, Srbija<br />
RICHARD BITTNER AG.,<br />
Ossiacher Strasse 7,<br />
Feldkirchen, Austrija<br />
MEDA MANUFACTURING<br />
GmbH, Neurather Ring 1,<br />
Keln, Nema~ka<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
ZAMBON S.P.A., Via della<br />
Chimica 9, Vi}enca, Italija<br />
BERLIN-CHEMIE AG<br />
(MENARINI GROUP) -<br />
PREDSTAVNI[TVO,<br />
Prilepska 1,<br />
Beograd, Srbija<br />
PHARMASWISS D.O.O.,<br />
Vojvode Stepe 18,<br />
Beograd, Srbija<br />
HEMOFARM AD, Beogradski<br />
put bb, Vr{ac, Srbija<br />
FARMALOGIST D.O.O.,<br />
Bulevar Vojvode<br />
Mi{i}a 25- 27,<br />
Beograd, Srbija<br />
MEDA PHARMACEUTICALS<br />
SWITZERLAND GMBH -<br />
PREDSTAVNI[TVO,<br />
Vojvode Milenka 52a/2,<br />
Beograd, Srbija<br />
HEMOFARM AD,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
BONIFAR D.O.O., Bulevar<br />
Zorana \in|i}a 87/1,<br />
Beograd, Srbija
Rupurut®<br />
hidrotalcit<br />
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Rupurut® forte<br />
hidrotalcit<br />
Rutacid®<br />
hidrotalcit<br />
Septolete®<br />
nana (Mentha piperita),<br />
etarsko ulje, eukaliptus<br />
(Eucalyptus spp), etarsko<br />
ulje, levomentol, timol,<br />
ben<strong>za</strong>lkonijum-hlorid<br />
Septolete® D<br />
nana (Mentha piperita),<br />
etarsko ulje, eukaliptus<br />
(Eucalyptus spp), etarsko<br />
ulje, levomentol, timol,<br />
ben<strong>za</strong>lkonijum-hlorid<br />
Septolete® divlja tre{nja<br />
cetilpiridinijum<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
tableta <strong>za</strong> `vakanje; 500mg;<br />
blister, 2x10kom<br />
tableta <strong>za</strong> `vakanje; 500mg;<br />
blister, 5x10kom<br />
tableta <strong>za</strong> `vakanje; 1000mg;<br />
blister, 2x10kom<br />
tableta <strong>za</strong> `vakanje; 500mg;<br />
blister, 2x10kom<br />
tableta <strong>za</strong> `vakanje; 500mg;<br />
blister, 6x10kom<br />
komprimovana lozenga;<br />
1mg+0.6mg+1.2mg+0.6mg+1mg;<br />
blister, 3x10kom<br />
komprimovana lozenga;<br />
1mg+0.6mg+1.2mg+0.6mg+1mg;<br />
blister, 2x15kom<br />
komprimovana lozenga;<br />
1mg+0.6mg+1.2mg+0.6mg+1mg;<br />
blister, 3x10kom<br />
komprimovana lozenga;<br />
1mg+0.6mg+1.2mg+0.6mg+1mg;<br />
blister, 2x15kom<br />
komprimovana lozenga; 1.2mg;<br />
blister, 2x9kom<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ re{ewa re{ ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
484/2007/12<br />
2537/2008/12<br />
2594/2008/12<br />
1869/2006/12<br />
1870/2006/12<br />
569/2007/12<br />
3577/2008/12<br />
570/2007/12<br />
3575/2008/12<br />
753/2007/12<br />
od<br />
od<br />
od<br />
od<br />
od<br />
07.02.2007<br />
08.08.2008<br />
13.08.2008<br />
18.10.2006<br />
18.10.2006<br />
od 01.03.2007<br />
od 07.10.2008<br />
od 01.03.2007<br />
od 07.10.2008<br />
od<br />
02.04.2007<br />
Str. 36 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
BAYER BITTERFELD GMBH,<br />
OT Greppin, Salegaster<br />
Chaussee 1, Bitterfeld-<br />
Wolfen, Nema~ka<br />
CATALENT GERMANY<br />
SCHORNDORF GMBH,<br />
Steinbeisstrasse 1 i 2,<br />
Schorndorf, Nema~ka<br />
KRKA, TOVARNA ZDRAVIL,<br />
D.D., [marje{ka cesta 6,<br />
Novo Mesto, Slovenija<br />
KRKA, TOVARNA ZDRAVIL,<br />
D.D., [marje{ka cesta 6,<br />
Novo Mesto, Slovenija<br />
KRKA, TOVARNA ZDRAVIL,<br />
D.D., [marje{ka cesta 6,<br />
Novo Mesto, Slovenija<br />
KRKA, TOVARNA ZDRAVIL,<br />
D.D., [marje{ka cesta 6,<br />
Novo Mesto, Slovenija<br />
BAYER D.O.O.,<br />
Omladinskih brigada 88b,<br />
Beograd - Novi<br />
Beograd, Srbija<br />
BAYER D.O.O.,<br />
Omladinskih brigada 88b,<br />
Beograd - Novi<br />
Beograd, Srbija<br />
KRKA FARMA D.O.O.,<br />
Jurija Gagarina 26/V/II,<br />
Beograd, Srbija<br />
KRKA FARMA D.O.O.,<br />
Jurija Gagarina 26/V/II,<br />
Beograd, Srbija<br />
KRKA FARMA D.O.O.,<br />
Jurija Gagarina 26/V/II,<br />
Beograd, Srbija<br />
KRKA FARMA D.O.O.,<br />
Jurija Gagarina 26/V/II,<br />
Beograd, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Septolete® limun<br />
cetilpiridinijum<br />
Septolete® plus<br />
cetilpiridinijum, benzokain<br />
Septolete® plus med i<br />
limeta<br />
cetilpiridinijum, benzokain<br />
Septolete® zelena jabuka<br />
cetilpiridinijum<br />
Sinedol<br />
heparin-natrijum,<br />
dimetilsulfoksid,<br />
dekspantenol<br />
Sirup br{ljana Bocko®<br />
Standardizovani suvi<br />
ekstrakt lista br{ljana sa<br />
najmanje 9% hederakozida<br />
C<br />
Skinoren®<br />
azelainska kiselina<br />
Smecta®<br />
diosmektit<br />
Solcoseryl®<br />
deproteinizovani<br />
hemodijali<strong>za</strong>t tele}e krvi,<br />
hemijski i biolo{ki<br />
standardizovan, polidokanol<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
komprimovana lozenga; 1.2mg;<br />
blister, 2x9kom<br />
komprimovana lozenga;<br />
1mg+5mg; blister, 2x9kom<br />
komprimovana lozenga;<br />
1mg+5mg; blister, 3x10kom<br />
komprimovana lozenga;<br />
1mg+5mg; blister, 2x9kom<br />
komprimovana lozenga; 1.2mg;<br />
blister, 2x9kom<br />
gel; 500i.j./g+150mg/g+25mg/g;<br />
tuba, 1x40g<br />
sirup; 7mg/ml; bo~ica, 1x125ml<br />
krem; 200mg/g; tuba, 1x30g<br />
pra{ak <strong>za</strong> oralnu suspenziju; 3g;<br />
kesica, 10x1kom<br />
pasta <strong>za</strong> usnu sluznicu;<br />
2.125mg/g+10mg/g; tuba, 1x5g<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
754/2007/12<br />
3579/2008/12<br />
2315/2006/12<br />
02.04.2007<br />
07.10.2008<br />
12.12.2006<br />
6392/2010/12 od 08.11.2010<br />
752/2007/12<br />
4432/2008/12<br />
178/2006/12<br />
186/2006/12<br />
513/2009/12<br />
459/2006/12<br />
od<br />
od<br />
od<br />
od<br />
od<br />
02.04.2007<br />
11.12.2008<br />
od 13.02.2006<br />
od 13.02.2006<br />
od 17.02.2009<br />
od 06.04.2006<br />
Str. 37 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
KRKA, TOVARNA ZDRAVIL,<br />
D.D., [marje{ka cesta 6,<br />
Novo Mesto, Slovenija<br />
KRKA, TOVARNA ZDRAVIL,<br />
D.D., [marje{ka cesta 6,<br />
Novo Mesto, Slovenija<br />
KRKA, TOVARNA ZDRAVIL,<br />
D.D., [marje{ka cesta 6, Novo<br />
Mesto, Slovenija<br />
KRKA, TOVARNA ZDRAVIL,<br />
D.D., [marje{ka cesta 6,<br />
Novo Mesto, Slovenija<br />
HEMOFARM AD, Beogradski<br />
put bb, Vr{ac, Srbija<br />
PHARMANOVA<br />
VELEPRODAJA D.O.O.,<br />
Bore Stankovi}a 2, P.Box 21,<br />
Beograd, Srbija<br />
INTENDIS<br />
MANUFACTURING SPA.,<br />
Via E.Schering, 21, Segrate,<br />
Milano, Italija<br />
BEAUFOUR IPSEN<br />
INDUSTRIE, Rue d'Ethe-<br />
Virthon, Dreux, Francuska<br />
ALKALOID AD, Bul.<br />
Aleksandar Makedonski 12,<br />
Skopje, Makedonija u saradnji<br />
sa VALEANT<br />
PHARMACEUTICALS<br />
SWITZERLAND GMBH,<br />
Ruhrbergstrasse 21,<br />
Birsfelden, [vajcarska<br />
KRKA FARMA D.O.O.,<br />
Jurija Gagarina 26/V/II,<br />
Beograd, Srbija<br />
KRKA FARMA D.O.O.,<br />
Jurija Gagarina 26/V/II,<br />
Beograd, Srbija<br />
KRKA FARMA D.O.O.,<br />
Jurija Gagarina 26/V/II,<br />
Beograd, Srbija<br />
KRKA FARMA D.O.O.,<br />
Jurija Gagarina 26/V/II,<br />
Beograd, Srbija<br />
HEMOFARM AD, Beogradski<br />
put bb, Vr{ac, Srbija<br />
PHARMANOVA D.O.O.,<br />
Industrijska 8,<br />
Obrenovac, Srbija<br />
BAYER D.O.O., Omladinskih<br />
brigada 88b, Beograd - Novi<br />
Beograd, Srbija<br />
PHARMASWISS D.O.O.,<br />
Vojvode Stepe 18,<br />
Beograd, Srbija<br />
MEDA PHARMACEUTICALS<br />
SWITZERLAND GMBH -<br />
PREDSTAVNI[TVO,<br />
Vojvode Milenka 52a/2,<br />
Beograd, Srbija
Spedifen®<br />
ibuprofen<br />
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Spedifen® 200<br />
ibuprofen<br />
Strepsils® Cool<br />
amilmetakrezol, 2,4dihlorbenzil<br />
alkohol<br />
Strepsils® limun bez<br />
{e}era<br />
amilmetakrezol, 2,4-<br />
dihlorbenzil alkohol<br />
Strepsils® med i limun<br />
amilmetakrezol, 2,4dihlorbenzil<br />
alkohol<br />
Strepsils® mentol i<br />
eukaliptus<br />
amilmetakrezol, 2,4-<br />
dihlorbenzil alkohol,<br />
levomentol<br />
Strepsils® plus<br />
amilmetakrezol, 2,4dihlorbenzil<br />
alkohol, lidokain<br />
Strepsils® pomorand`a sa<br />
vitaminom C<br />
amilmetakrezol, 2,4-<br />
dihlorbenzil alkohol,<br />
askorbinska kiselina,<br />
natrijum-askorbat<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
tableta; 200mg;<br />
blister, 2x10kom<br />
granule <strong>za</strong> oralni rastvor; 200mg;<br />
kesica, 20x1kom<br />
pastila; 0.6mg+1.2mg;<br />
blister, 2x8kom<br />
pastila; 0.6mg+1.2mg;<br />
blister, 2x8kom<br />
pastila; 0.6mg+1.2mg;<br />
blister, 2x12kom<br />
pastila; 0.6mg+1.2mg+8mg;<br />
blister, 2x12kom<br />
pastila; 0.6mg+1.2mg+10mg;<br />
blister, 2x12kom<br />
pastila;<br />
0.6mg+1.2mg+33.5mg+74.9mg;<br />
blister, 2x12kom<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
<strong>Agencija</strong> <strong>za</strong> <strong>lekove</strong> i <strong>medicinska</strong> <strong>sredstva</strong> <strong>Srbije</strong> Str. 38 od 42<br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
2468/2008/12<br />
2919/2009/12<br />
8018/2009/12<br />
748/2006/12<br />
227/2006/12<br />
228/2006/12<br />
171/2006/12<br />
229/2006/12<br />
od 04.08.2008<br />
od 01.06.2009<br />
od<br />
11.12.2009<br />
od 02.06.2006<br />
od 28.02.2006<br />
od 28.02.2006<br />
od 13.02.2006<br />
od<br />
28.02.2006<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
ZAMBON S.P.A., Via della<br />
Chimica 9, Vi}enca, Italija<br />
ZAMBON SWITZERLAND<br />
LTD, Via Industria, 13,<br />
Cadempino, [vajcarska<br />
RECKITT BENCKISER<br />
HEALTHCARE<br />
INTERNATIONAL LTD.,<br />
Thane Road, Nottingham,<br />
Velika Britanija<br />
RECKITT BENCKISER<br />
HEALTHCARE<br />
INTERNATIONAL LTD.,<br />
Thane Road, Nottingham,<br />
Velika Britanija<br />
RECKITT BENCKISER<br />
HEALTHCARE<br />
INTERNATIONAL LTD.,<br />
Thane Road, Nottingham,<br />
Velika Britanija<br />
RECKITT BENCKISER<br />
HEALTHCARE<br />
INTERNATIONAL LTD.,<br />
Thane Road, Nottingham,<br />
Velika Britanija<br />
RECKITT BENCKISER<br />
HEALTHCARE<br />
INTERNATIONAL LTD.,<br />
Thane Road, Nottingham,<br />
Velika Britanija<br />
RECKITT BENCKISER<br />
HEALTHCARE<br />
INTERNATIONAL LTD.,<br />
Thane Road, Nottingham,<br />
Velika Britanija<br />
BONIFAR D.O.O., Bulevar<br />
Zorana \in|i}a 87/1,<br />
Beograd, Srbija<br />
BONIFAR D.O.O., Bulevar<br />
Zorana \in|i}a 87/1,<br />
Beograd, Srbija<br />
PHARMASWISS D.O.O.,<br />
Vojvode Stepe 18,<br />
Beograd, Srbija<br />
PHARMASWISS D.O.O.,<br />
Vojvode Stepe 18,<br />
Beograd, Srbija<br />
PHARMASWISS D.O.O.,<br />
Vojvode Stepe 18,<br />
Beograd, Srbija<br />
PHARMASWISS D.O.O.,<br />
Vojvode Stepe 18,<br />
Beograd, Srbija<br />
PHARMASWISS D.O.O.,<br />
Vojvode Stepe 18,<br />
Beograd, Srbija<br />
PHARMASWISS D.O.O.,<br />
Vojvode Stepe 18,<br />
Beograd, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Tanakan®<br />
ginko (Ginkgo biloba), suvi<br />
ekstrakt lista<br />
Tantum® Lemon<br />
benzidamin<br />
Tantum® Verde<br />
benzidamin<br />
Tantum® Verde<br />
benzidamin<br />
Thiogamma® 600 oral<br />
alfa-lipoinska kiselina<br />
Thiogamma® 600 oral<br />
alfa-lipoinska kiselina<br />
Tonsilotren<br />
Atropin-sulfat, triturat D5,<br />
Kalijum-dihromat, triturat D4,<br />
Hepar sulfuris, triturat D3,<br />
@iva(II)-jodid, triturat D8,<br />
Silicijum-dioksid, triturat D2<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
oblo`ena tableta; 40mg;<br />
blister, 6x15kom<br />
oblo`ena tableta; 40mg;<br />
blister, 2x15kom<br />
lozenga; 3mg;<br />
folija, 2x10kom<br />
sprej <strong>za</strong> usnu sluznicu;<br />
1.5mg/ml; bo~ica sa<br />
raspr{iva~em, 1x30ml<br />
lozenga; 3mg; folija, 2x10kom<br />
rastvor <strong>za</strong> ispiranje usta;<br />
1.5mg/ml; bo~ica, 1x150ml<br />
film tableta; 600mg;<br />
blister, 3x10kom<br />
film tableta; 600mg;<br />
blister, 6x10kom<br />
film tableta; 600mg;<br />
blister, 3x10kom<br />
film tableta; 600mg;<br />
blister, 6x10kom<br />
tableta; 12.5mg+50mg+<br />
10mg+25mg+5mg;<br />
blister, 3x20kom<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
1084/2009/12<br />
1085/2009/12<br />
1269/2010/12<br />
1586/2006/12<br />
196/2007/12<br />
195/2007/12<br />
1770/2006/12<br />
1771/2006/12<br />
1731/2010/12<br />
1732/2010/12<br />
2/2006/1500<br />
od 31.03.2009<br />
od 31.03.2009<br />
od 16.02.2010<br />
od 05.09.2006<br />
od 16.01.2007<br />
od 16.01.2007<br />
od 02.10.2006<br />
od 02.10.2006<br />
od 16.03.2010<br />
od 16.03.2010<br />
od<br />
10.02.2006<br />
Str. 39 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
BEAUFOUR IPSEN<br />
INDUSTRIE, Rue d'Ethe-<br />
Virthon, Dreux, Francuska<br />
AZIENDE CHIMICHE<br />
RIUNITE ANGELINI<br />
FRANCESCO (A.C.R.A.F.)<br />
S.P.A., Via Vecchia del<br />
Pinocchio, 22, Ancona, Italija<br />
AZIENDE CHIMICHE<br />
RIUNITE ANGELINI<br />
FRANCESCO (A.C.R.A.F.)<br />
S.P.A., Via Vecchia del<br />
Pinocchio, 22, Ancona, Italija<br />
SANOCHEMIA<br />
PHARMAZEUTIKA AG,<br />
Landeggerstrasse 7, Neufeld,<br />
Leitha, Austrija<br />
ARTESAN PHARMA GMBH<br />
& CO.KG, Wendlandstr. 1,<br />
Lüchow, Nema~ka <strong>za</strong><br />
WÖRWAG PHARMA GMBH<br />
& CO. KG, Calwer Straße 7,<br />
Böblingen, Nema~ka<br />
DRAGENOPHARM<br />
APOTHEKER PÜSCHL<br />
GMBH, Göllstraße 1,<br />
Tittmoning, Nema~ka<br />
Deutsche Homöopathie-Union<br />
DHU-Arzneimittel GmbH &<br />
Co. KG., Ottostrasse 24,<br />
76227 Karlsruhe, Nema~ka<br />
PHARMASWISS D.O.O.,<br />
Vojvode Stepe 18,<br />
Beograd, Srbija<br />
BONIFAR D.O.O., Bulevar<br />
Zorana \in|i}a 87/1,<br />
Beograd, Srbija<br />
BONIFAR D.O.O., Bulevar<br />
Zorana \in|i}a 87/1,<br />
Beograd, Srbija<br />
BONIFAR D.O.O., Bulevar<br />
Zorana \in|i}a 87/1,<br />
Beograd, Srbija<br />
WORWAG PHARMA GMBH<br />
& CO.KG -<br />
PREDSTAVNI[TVO,<br />
Zahumska 23 a/1,<br />
Beograd-Zvezdara, Srbija<br />
WORWAG PHARMA GMBH<br />
& CO.KG -<br />
PREDSTAVNI[TVO,<br />
Zahumska 23 a/1,<br />
Beograd-Zvezdara, Srbija<br />
LIBRA BGD D.O.O.,<br />
Obalskih radnika 4 g,<br />
Beograd, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Tylol® Hot<br />
pseudoefedrin, paracetamol,<br />
hlorfenamin<br />
Tylol® Hot <strong>za</strong> decu<br />
pseudoefedrin, paracetamol,<br />
hlorfenamin<br />
Tylol® Hot-D<br />
pseudoefedrin, paracetamol,<br />
hlorfenamin<br />
Upsarin® sa vitaminom C<br />
acetilsalicilna kiselina,<br />
askorbinska kiselina<br />
Venoruton®<br />
trokserutin<br />
Venoruton®<br />
trokserutin<br />
Venosan®<br />
heparin-natrijum, esencijalni<br />
fosfolipidi, escin<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
oralni pra{ak;<br />
60mg+500mg+4mg;<br />
kesica, 12x20g<br />
oralni pra{ak;<br />
60mg+500mg+4mg;<br />
kesica, 6x20g<br />
oralni pra{ak;<br />
30mg+250mg+2mg;<br />
kesica, 12x10g<br />
oralni pra{ak;<br />
30mg+250mg+2mg;<br />
kesica, 6x10g<br />
oralni pra{ak;<br />
60mg+500mg+4mg;<br />
kesica, 12x6g<br />
oralni pra{ak;<br />
60mg+500mg+4mg;<br />
kesica, 6x6g<br />
{ume}a tableta; 330mg+200mg;<br />
fiola, 1x20kom<br />
{ume}a tableta; 330mg+200mg;<br />
fiola, 1x10kom<br />
gel; 2%; tuba, 1x40g<br />
{ume}a tableta; 500mg;<br />
kontejner plasti~ni, 1x20kom<br />
{ume}a tableta; 1000mg;<br />
kontejner plasti~ni, 1x15kom<br />
kapsula, tvrda; 300mg;<br />
blister, 2x10kom<br />
gel; 100i.j./g+10mg/g+10mg/g;<br />
tuba, 1x40g<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
1181/2006/12<br />
2561/2009/12<br />
1625/2006/12<br />
2560/2009/12<br />
1182/2006/12<br />
2562/2009/12<br />
820/2007/12<br />
819/2007/12<br />
2450/2008/12<br />
2451/2008/12<br />
2452/2008/12<br />
2449/2008/12<br />
648/2009/12<br />
od 13.07.2006<br />
od 06.05.2009<br />
od 12.09.2006<br />
od 06.05.2009<br />
od 13.07.2006<br />
od 06.05.2009<br />
od 13.04.2007<br />
od 13.04.2007<br />
od 04.08.2008<br />
od 04.08.2008<br />
od 04.08.2008<br />
od 04.08.2008<br />
od<br />
23.02.2009<br />
Str. 40 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
NOBELFARMA ILAC<br />
SANAYII VE TICARET A.S.,<br />
Sancaklar, Duzce, Turska<br />
NOBELFARMA ILAC<br />
SANAYII VE TICARET A.S.,<br />
Sancaklar, Duzce, Turska<br />
NOBELFARMA ILAC<br />
SANAYII VE TICARET A.S.,<br />
Sancaklar, Duzce, Turska<br />
BRISTOL-MYERS SQUIBB,<br />
304, avenue Jean Bru, Agen,<br />
Francuska<br />
NOVARTIS CONSUMER<br />
HEALTH SA, Route de<br />
l'Etraz, Nyon, [vajcarska<br />
NOVARTIS FARMACEUTICA<br />
S.A., Ronda de Santa Maria,<br />
158, Barbera del Valles<br />
(Barcelona), [panija<br />
BOSNALIJEK D.D., Juki}eva<br />
53, Sarajevo, Bosna i<br />
Hercegovina<br />
NOBEL ILAC SANAYII VE<br />
TICARET AS -<br />
PREDSTAVNI[TVO,<br />
Molerova 78, Beograd -<br />
Vra~ar, Srbija<br />
NOBEL ILAC SANAYII VE<br />
TICARET AS -<br />
PREDSTAVNI[TVO,<br />
Molerova 78, Beograd -<br />
Vra~ar, Srbija<br />
NOBEL ILAC SANAYII VE<br />
TICARET AS -<br />
PREDSTAVNI[TVO,<br />
Molerova 78, Beograd -<br />
Vra~ar, Srbija<br />
PHARMASWISS D.O.O.,<br />
Vojvode Stepe 18,<br />
Beograd,<br />
MEDIS INTERNATIONAL<br />
D.O.O., Bulevar Mihaila<br />
Pupina 10d/1 pp7, Beograd -<br />
Novi Beograd, Srbija<br />
MEDIS INTERNATIONAL<br />
D.O.O., Bulevar Mihaila<br />
Pupina 10d/1 pp7, Beograd -<br />
Novi Beograd, Srbija<br />
BOSNALIJEK D.D. -<br />
PREDSTAVNI[TVO, Cara<br />
Nikolaja II 61/B, Beograd,<br />
Srbija
Viatromb®<br />
heparin<br />
Viatromb®<br />
heparin<br />
Vidisic®<br />
karbomer<br />
Visine®<br />
tetrizolin<br />
Visine®<br />
tetrizolin<br />
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Vitamin C<br />
askorbinska kiselina<br />
Vitamin C<br />
askorbinska kiselina<br />
Vitamin C<br />
askorbinska kiselina<br />
Vivoton S<br />
Crataegus Ø, Convallaria<br />
majalis Ø, Adonis vernalis<br />
Ø, Valeriana Ø, Lobelia<br />
inflata, Cactus, etar<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
sprej <strong>za</strong> ko`u, gel; 2400i.j./g;<br />
sprej-boca, 1x25g<br />
sprej <strong>za</strong> ko`u, gel; 2400i.j./g;<br />
sprej-boca, 1x25g<br />
gel <strong>za</strong> o~i; 2mg/g;<br />
plasti~na tuba, 1x10g<br />
kapi <strong>za</strong> o~i, rastvor; 0.05%;<br />
bo~ica sa kapaljkom, 1x15ml<br />
kapi <strong>za</strong> o~i, rastvor; 0.05%;<br />
bo~ica sa kapaljkom, 1x15ml<br />
tableta; 500mg; strip, 3x6kom<br />
tableta; 500mg; strip,<br />
25x10kom<br />
tableta; 500mg;<br />
folija, 50x10kom<br />
{ume}a tableta; 1000mg;<br />
fiola, 1x20kom<br />
oralni rastvor; 50g/100g+10g/100g+<br />
1g/100g+10g/100g+5.5g/100g+<br />
3g/100g+3g/100g;<br />
bo~ica, 1x30ml<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
1627/2007/12<br />
21/2006/12<br />
1540/2010/12<br />
1112/2006/12<br />
6584/2010/12<br />
6583/2010/12<br />
646/2006/12<br />
1084/2007/12<br />
3/2006/1500<br />
od<br />
07.08.2007<br />
237/2006/12 od 03.03.2006<br />
od<br />
od<br />
10.01.2006<br />
03.03.2010<br />
od 05.07.2006<br />
od 18.11.2010<br />
od 18.11.2010<br />
od 17.05.2006<br />
od 23.05.2007<br />
od 10.02.2006<br />
MEDICOM INTERNATIONAL<br />
S.R.O., Paterni 7, Brno, Če{ka<br />
Str. 41 od 42<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
PHARBIL WALTROP GMBH,<br />
Im Wirrigen 25, Waltrop,<br />
Nema~ka<br />
DR. GERHARD MANN,<br />
CHEM. - PHARM. FABRIK<br />
GMBH, Brunsbutteler Damm<br />
165 - 173, Berlin, Nema~ka<br />
JANSSEN PHARMACEUTICA<br />
N.V., Turnhoutseweg 30,<br />
Beerse, Belgija<br />
PILLAR5 PHARMA INC., 365<br />
Madawaska Boulevard,<br />
Arnprior, Ontario, Kanada<br />
GALENIKA AD,<br />
Batajni~ki drum b.b.,<br />
Beograd, Srbija<br />
HEMOFARM D.O.O. [ABAC,<br />
Hajduk Veljkova bb,<br />
[abac, Srbija<br />
HEMOFARM AD, Beogradski<br />
put bb, Vr{ac, Srbija<br />
Deutsche Homöopathie-Union<br />
DHU-Arzneimittel GmbH &<br />
Co. KG., Ottostrasse 24,<br />
76227 Karlsruhe, Nema~ka<br />
EUROFARM D.O.O.,<br />
Bo{ka Petrovi}a 3a,<br />
Beograd, Srbija<br />
EUROFARM D.O.O.,<br />
Bo{ka Petrovi}a 3a,<br />
Beograd, Srbija<br />
MEDILENS D.O.O.,<br />
Savski trg 9,<br />
Beograd, Srbija<br />
JOHNSON & JOHNSON S.E.<br />
INC.- PREDSTAVNI[TVO,<br />
Omladinskih brigada 88 b, Beograd<br />
- Novi Beograd, Srbija<br />
JOHNSON & JOHNSON S.E.<br />
INC.- PREDSTAVNI[TVO,<br />
Omladinskih brigada 88 b,<br />
Beograd - Novi Beograd,<br />
Srbija<br />
GALENIKA AD,<br />
Batajni~ki drum b.b.,<br />
Beograd, Srbija<br />
HEMOFARM D.O.O. [ABAC,<br />
Hajduk Veljkova bb,<br />
[abac, Srbija<br />
MULTIVITA D.O.O.,<br />
Beogradski put bb,<br />
Vr{ac, Srbija<br />
LIBRA BGD D.O.O.,<br />
Obalskih radnika 4 g,<br />
Beograd, Srbija
Naziv Naziv leka<br />
leka<br />
INN<br />
INN<br />
Voda <strong>za</strong> injekcije<br />
voda <strong>za</strong> injekcije<br />
Voltaren Emulgel<br />
diklofenak<br />
Zirkulin Anti-Acid<br />
kalcijum-karbonat<br />
Zirkulin Valeriana<br />
suvi vodeni ekstrakt korena<br />
odoljena, suvi vodeni<br />
ekstrakt {i{arica hmelja<br />
Oblik Oblik, Oblik do<strong>za</strong> do<strong>za</strong> ii<br />
i<br />
pakovawe pakovawe pakovawe leka<br />
leka<br />
rastvara~ <strong>za</strong> parenteralnu<br />
upotrebu; 5ml; ampula, 50x5ml<br />
gel; 1%; tuba, 1x50g<br />
gel; 1%; tuba, 1x100g<br />
komprimovana lozenga; 500mg;<br />
blister, 3x10kom<br />
oblo`ena tableta; 160mg+40mg;<br />
blister, 1x20kom<br />
<strong>Agencija</strong> <strong>Agencija</strong> <strong>za</strong> <strong>za</strong> <strong>lekove</strong> <strong>lekove</strong> i i <strong>medicinska</strong> <strong>medicinska</strong> <strong>sredstva</strong> <strong>sredstva</strong> <strong>Srbije</strong><br />
<strong>Srbije</strong><br />
<strong>Agencija</strong> <strong>za</strong> <strong>lekove</strong> i <strong>medicinska</strong> <strong>sredstva</strong> <strong>Srbije</strong> Str. 42 od 42<br />
Broj Broj i i datum datum re{ewa re{ewa o<br />
o<br />
stavqawu stavqawu leka leka u u u promet<br />
promet<br />
2634/2009/12<br />
1235/2008/12<br />
7361/2009/12<br />
3213/2008/12<br />
2268/2008/12<br />
od 13.05.2009<br />
od 28.03.2008<br />
od 19.11.2009<br />
od 15.09.2008<br />
od 15.07.2008<br />
Proizvo|a~ Proizvo|a~<br />
Nosilac Nosilac dozvole<br />
dozvole<br />
GALENIKA AD, Batajni~ki<br />
drum b.b., Beograd, Srbija<br />
NOVARTIS PHARMA<br />
PRODUKTIONS GMBH,<br />
Oflinger Strasse 44, Wehr,<br />
Nema~ka<br />
ROHA ARZNEIMITTEL<br />
GMBH, Rockwinkeler<br />
Heerstrase 100, Bremen,<br />
Nema~ka<br />
ROHA ARZNEIMITTEL<br />
GMBH, Rockwinkeler<br />
Heerstrase 100, Bremen,<br />
Nema~ka<br />
Даном објављивања ове Листе лекова, престаје да важи Листа лекова који се издају без рецепта и који се могу рекламирати у средствима јавног<br />
информисања (Службени гласник РС бр. 49/2008, 4/2009, 35/2009, 82/2009, 94/2009, 7/2010 и 35/2010).<br />
Br.: 515-00-00317-2011-3-001<br />
17. februar 2011. godine<br />
Beograd<br />
GALENIKA AD, Batajni~ki<br />
drum b.b., Beograd, Srbija<br />
MEDIS INTERNATIONAL<br />
D.O.O., Bulevar Mihaila<br />
Pupina 10d/1 pp7, Beograd -<br />
Novi Beograd, Srbija<br />
ACTAVIS D.O.O., \or|a<br />
Stanojevi}a 12, Beograd -<br />
Novi Beograd, Srbija<br />
ACTAVIS D.O.O., \or|a<br />
Stanojevi}a 12, Beograd -<br />
Novi Beograd, Srbija<br />
DIREKTOR<br />
Mr farm. spec. Tatjana [ipeti}, s.r.