1 - Ganatleba
1 - Ganatleba 1 - Ganatleba
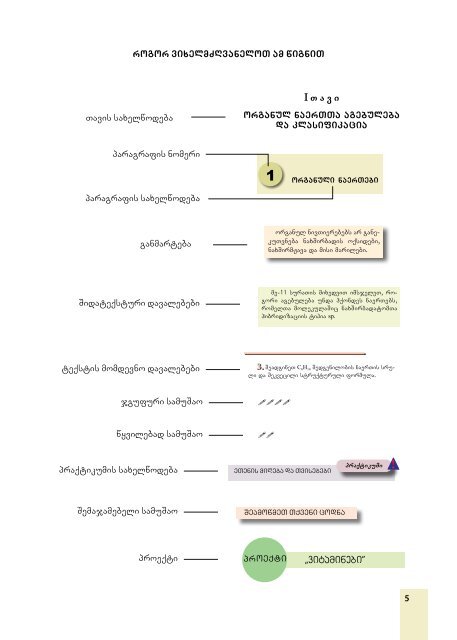
Tavis saxelwodeba paragrafis nomeri paragrafis saxelwodeba ganmarteba Sidateqsturi davalebebi teqstis momdevno davalebebi praqtikumis saxelwodeba Semajamebeli samuSao rogor vixelmZRvaneloT am wigniT jgufuri samuSao wyvilebad samuSao proeqti proeqti I T a v i organul naerTTa agebuleba da klasifikacia 1 organuli naerTebi organul nivTie re bebs ar ganekuTvneba naxSirbadis oqsi debi, naxSirmJava da misi marilebi. me-11 suraTis mixedviT imsjeleT, rogori agebuleba unda hqondes naerTebs, romelTa molekulaSic naxSirbadatomTa hibridizaciis tipia sp. 3. SeadgineT C 6H 14 Sedgenilobis naerTis sruli da Sekvecili struqturuli formula. eTenis miReba da Tvisebebi SeamowmeT Tqveni codna praqtikumi `vitaminebi~ 5
- Page 2 and 3: 78 am TavSi ganvixilavT erTatomian
- Page 4 and 5: 80 spirtis molekulaSi radikali Sesa
- Page 6 and 7: 82 CaatareT 26-e suraTze asaxuli re
- Page 8 and 9: 84 neba formaldehidad da WianWvelmJ
- Page 10 and 11: 86 14 mravalatomiani spirtebi orato
- Page 12 and 13: 88 3 glicerini urTierTqmedebs azotm
- Page 14 and 15: 90 wvrobas, amitom masTan muSaobisa
- Page 16 and 17: 92 fenolformaldehiduri fisisagan am
- Page 18 and 19: 94 saerTaSoriso nomenklaturis mixed
- Page 20 and 21: 96 2 mierTebis reaqcia. mierTebis r
- Page 22 and 23: 98 3. SeadgineT sqemis Sesabamisi
- Page 24 and 25: 100 testosteroni hormonebidan aRsan
- Page 26 and 27: 102 nomenklatura da izomeria SeamCn
- Page 28 and 29: 104 1 R - COOH + NaOH → R - COONa
- Page 30 and 31: 106 grZeljaWviani ujeri karbonmJave
- Page 32 and 33: 108 qimiuri Tvisebebi rogorc araerT
- Page 34 and 35: 110 am naklis aRmofxvra SesaZlebeli
- Page 36 and 37: 112 SeamowmeT Tqveni codna 1 kalciu
- Page 38 and 39: 114 jer kidev XIX saukuneSi mecnier
- Page 40 and 41: 116 am TavSi ganvixilavT naxSirwyl
- Page 42 and 43: 118 qsovilSi moxvedrisas glukoza ne
- Page 44 and 45: 120 glukozis qimiuri Tvisebebi rogo
- Page 46 and 47: 122 glukozasTan vercxlis oqsidis am
- Page 48 and 49: 124 bunebaSi gavrceleba saxamebeli
- Page 50 and 51: 126 iseve, rogorc nitrojgufis Semcv
Tavis saxelwodeba<br />
paragrafis nomeri<br />
paragrafis saxelwodeba<br />
ganmarteba<br />
Sidateqsturi davalebebi<br />
teqstis momdevno davalebebi<br />
praqtikumis saxelwodeba<br />
Semajamebeli samuSao<br />
rogor vixelmZRvaneloT am wigniT<br />
jgufuri samuSao<br />
wyvilebad samuSao<br />
proeqti<br />
proeqti<br />
I T a v i<br />
organul naerTTa agebuleba<br />
da klasifikacia<br />
1<br />
organuli naerTebi<br />
organul nivTie re bebs ar ganekuTvneba<br />
naxSirbadis oqsi debi,<br />
naxSirmJava da misi marilebi.<br />
me-11 suraTis mixedviT imsjeleT, rogori<br />
agebuleba unda hqondes naerTebs,<br />
romelTa molekulaSic naxSirbadatomTa<br />
hibridizaciis tipia sp.<br />
3. SeadgineT C 6H 14 Sedgenilobis naerTis sruli<br />
da Sekvecili struqturuli formula.<br />
<br />
<br />
eTenis miReba da Tvisebebi<br />
SeamowmeT Tqveni codna<br />
praqtikumi<br />
`vitaminebi~<br />
5
78<br />
am TavSi ganvixilavT<br />
erTatomian da mravalatomian spirtebs.<br />
aldehidebs da ketonebs.<br />
karbonmJavebs.<br />
esterebsa da cximebs.<br />
JangbadSecveli organuli naerTebis<br />
miRebas, Tvisebebsa da gamoyenebas.<br />
IV T a v i<br />
JangbadSemcveli organuli naerTebi<br />
am Tavis Seswavlis Semdeg<br />
SeZlebT, formulis mixedviT JangbadSemcveli organuli<br />
naerTi miakuTnoT naerTTa Sesabamis klass.<br />
SeadgenT JangbadSemcveli organuli naerTebis molekulaTa<br />
modelebs.<br />
daakavSirebT JangbadSecveli organuli naerTis Tvisebebs<br />
mis SedgenilobaSi Semaval funqciur jgufTan.<br />
qimiuri reaqciebiT asaxavT JangbadSemcveli organuli<br />
naerTebis Tvisebebs da miRebis xerxebs.<br />
imsjelebT mniSvnelovani JangbadSemcveli organuli<br />
naerTebis praqtikul mniSvnelobaze.
13<br />
SesaZlebelia Tu ara, rom 25-e suraTze gamosaxuli<br />
ori sxvadasxva siTxe erTsa da imave nivTierebas<br />
Seicavdes?<br />
xom ar iciT, ra Tvisebebis gamo ga mo iyenes is<br />
pirvelsa da meore SemTxvevaSi?<br />
am saxelwodebis mqone sxva nivTierebebic xom ar<br />
aris TqvenTvis cnobili?<br />
spirtebi<br />
spirti aris orga -<br />
nu li nivTiereba, rom<br />
lis molekula Sic<br />
naxSirwyalbadis radikalTan<br />
da kav Si rebulia<br />
erTi an ramdenime<br />
hidroqsilis<br />
jgu fi.<br />
spirtis zogadi<br />
fo r mulaa R‑OH.<br />
ro gorc wesi, nax-<br />
Sir wyal badis radikals<br />
R-iT aRniSnaven.<br />
am aRniSvnas gamoviyenebT<br />
Cvenc.<br />
hidroqsilis jgufs yvela spirti Seicavs. Tumca SesaZlebelia, rom sxvadasxva<br />
spirtis molekulaSi hidroqsilis jgufis sxvadasxva raodenoba iyos.<br />
molekulaSi hidroqsilis jgufis raodenobis mixedviT spirti aris:<br />
erTatomiani. magaliTad,<br />
CH 3OH,<br />
meTilis<br />
spirti<br />
meTanoli<br />
oratomiani. magaliTad, samatomiani. magaliTad,<br />
spirtebi erTmaneTisagan gansxvavdeba radikalis bunebiTac. Sesabamisad,<br />
spirti SesaZlebelia iyos:<br />
najeri – aseTi spirtis<br />
molekulaSi Semavali<br />
radikali najeria,<br />
magaliTad,<br />
CH 3 – CH 2 – OH<br />
eTanoli<br />
spirtebi<br />
sur. 25. alkoholiani sasmeli da odekoloni<br />
CH 3 – CH 2 – OH<br />
eTilis spirti<br />
eTanoli<br />
naerTebi, romelTa molekulaSic<br />
benzolis birTvTan uSualodaa dakavSirebuli<br />
OH – jgufi, TvisebebiT<br />
mkveTrad gansxvavdeba spirtebisagan.<br />
amitom isini calke klasadaa gamoyofili<br />
da fenolebi ewodeba.<br />
HO – CH 2 – CH 2 – OH<br />
eTilenglikoli<br />
ujeri – aseTi spirtis<br />
molekulaSi Semaval<br />
radikalSi naxSirbadis<br />
atomebs Soris<br />
jeradi bmebia (ormagi<br />
an sammagi) magaliTad,<br />
CH 2 = CH – CH 2 – OH<br />
fenoli<br />
OH<br />
CH2 – CH – CH2 OH OH OH<br />
glicerini<br />
aromatuli – cxadia<br />
aseTi spirtis molekulaSi<br />
benzolis birTvia,<br />
magram OH – jgufi<br />
masTan dakavSirebulia<br />
ara uSualod, aramed<br />
naxSirbadatomiT.<br />
maga liTad,<br />
CH 2 – OH<br />
fenilmeTanoli<br />
79
80<br />
spirtis molekulaSi radikali SesaZlebelia Seicavdes pirvelad, meoreul<br />
da mesameul naxSirbadatoms. Sesabamisad spirtic SeiZleba iyos:<br />
pirveladi – aseTi<br />
spir tis molekulaSi<br />
hid ro qsilis jgufi<br />
dakavSirebulia pirvelad<br />
naxSirbadatomTan.<br />
CH 3 – CH 2 – CH 2 – OH<br />
izomeria da nomenklatura<br />
spirtis saxelwodebis Sedgenisas Sesabamisi radikalis saxelwodebas<br />
emateba sufiqsi `oli~. sufiqsis Semdeg cifriT miuTiTeben hidroqsilis<br />
jgufis mdebareobas ZiriTad jaWvSi:<br />
H<br />
H – C – OH<br />
H<br />
meTanoli<br />
meTilis spirti<br />
H<br />
H<br />
H – C – C – OH<br />
H H<br />
eTanoli<br />
eTilis spirti<br />
CH 3 – CH 2 – CH 2 OH<br />
propanoli – 1<br />
1 2<br />
HO – CH2 – CH2 – OH<br />
eTandioli-1,2<br />
homologiuri rigis meoTxe da yvela momdevni wevrs ki aqvs ConCxis izomerebi.<br />
magaliTad,<br />
4 3 2 1<br />
CH3 _CH2 – CH2 – CH2 OH da<br />
2 2 1<br />
CH3 – CH – CH2OH butanoli - 1 |<br />
CH3 2 - meTilpropanoli<br />
fizikuri Tvisebebi<br />
m e o r e u l i – a s e T i<br />
spirtis molekulaSi hidroqsilis<br />
jgu fi dakavSirebulia<br />
meoreul nax-<br />
SirbadatomTan.<br />
CH 3 – CH – CH 3<br />
|<br />
OH<br />
mesameuli – am SemTxvevaSi<br />
spirtis molekul<br />
a S i h i d r o q s i l i s<br />
jgufi dakavSirebu lia<br />
mesameul naxSirbadatomTan.<br />
magaliTad.<br />
T a v s a r T e -<br />
bi di-, tri-,<br />
tetra- da a.S.<br />
hidroqsilis<br />
jgufis raodenobasaRniSnavs.<br />
O homologiuri rigis mesame wevridan dawyebuli spirtebSi Tavs iCens funqciuri<br />
jgufis mdebareobis izomeria. magaliTad,<br />
3 2 1<br />
CH3 – CH2 – CH2 OH<br />
propanoli -1<br />
da<br />
3 2 1<br />
CH3 – CH – CH3 |<br />
naxSirbadatomebis<br />
danomvras<br />
iwyeben im bolod<br />
a n , s a i d a n a c<br />
OH<br />
u f r o a x l o s a a<br />
propanoli - 2<br />
fun qciuri jgufi.<br />
nivTierebis Tvisebebi misi Sedgenilobis da agebulebis Sedegia, am mxriv<br />
arc spirtebia gamonaklisi. maTi Tvisebebi ganpirobebulia molekulis SedgenilobiT<br />
da struqturiT.<br />
CH 3<br />
|<br />
CH 3 – C – CH 3<br />
|<br />
OH
Jangbadis atomis eleqtrouaryofiToba gacilebiT<br />
metia, vidre wyalbadisa da naxSirbadis, amis gamo spirtis<br />
molekulaSi C – O da O – H bmebi polarulia. Jangbadis<br />
atomze Tavmoyrilia nawilobriv uaryofiTi muxti, wyalbadis<br />
atomze ki – nawilobriv dadebiTi:<br />
δ + δ − δ +<br />
..<br />
R – CH2 → O<br />
..<br />
← H.<br />
amitom amboben, rom spirtis molekulaSi hidroqsilis<br />
jgufis wyalbadatoms mJavuri Tvisebebi aqvs. amiT<br />
is mkveTrad gansxvavdeba radikalis wyalbadatomisagan.<br />
eleqtronuli simkvrivis aseTi gadanawileba molekula-<br />
Si imis mizezic aris, rom spirtis molekulebs Soris myardeba wyalbaduri bma:<br />
δ + δ – δ + δ – δ + δ –<br />
... H – O ... H – O ... H – O ....<br />
R R R<br />
wyalbaduri bmis arseboba ganapirobebs spirtis fizikur Tvisebebsac.<br />
kerZod, spirtis duRilis temperatura mis molekulur masasTan SedarebiT<br />
Zalian maRalia. ase magaliTad, propani, romlis fardobiTi molekuluri masaa<br />
44, Cveulebriv pirobebSi airia, umartivesi spirti meTanoli ki – siTxea maSin,<br />
rodesac misi fardobiTi molekuluri masaa 32.<br />
erTatomiani najeri spirtebis homologiuri rigis pirveli TerTmeti wevri<br />
siTxea, meTormete wevridan dawyebuli – myari nivTierebebi. dabali molekuluri<br />
masis mqone spirtebi wyalSi kargad ixsneba, maT alkoholis suni da gemo<br />
aqvT. spirtis molekuluri masis gazrdasTan erTad wyalSi xsnadoba mcirdeba.<br />
magaliTad, oqtanoli ukve aRar ixsneba wyalSi.<br />
msgavs reaqciaSi Sedis wyalic:<br />
qimiuri Tvisebebi<br />
spirtebis specifikur Tvisebebs cxadia hidroqsilis<br />
jgufi ganapirobebs.<br />
1 spirtebi urTierTqmedebs tute<br />
da tutemiwa metalebTan. magaliTad,<br />
eTilis spirtis urTierTqmedebiT natriumTan<br />
warmoiqneba natriumis eTilati<br />
da gamoiyofa wyalbadi (sur. 26):<br />
2C 2H 5 – OH + 2Na → 2C 2H 5 – ONa + H 2 ↑<br />
eTanoli natriumis eTilati<br />
vinaidan metali spirtTan gacilebiT<br />
mSvidad urTierTqmedebs vidre wyalTan,<br />
laboratoriaSi tute metalebis narCenebis mosacileblad<br />
da ga sauvnebelyofad swo red spirts<br />
iye neben.<br />
2 Na + 2H 2O → 2NaOH + H 2 ↑<br />
wyalTan natriumis urTierTqmedebisas<br />
reaqcia ufro aqtiurad<br />
mimdinareobs, gamoiyofa<br />
imdenad didi<br />
r a o d e n o b i s<br />
s i T b o , r o m<br />
SeiZleba moxdes<br />
afeTqeba.<br />
msgavsi reaqciis<br />
mixedviT<br />
s p i r t i m J a -<br />
vuri bunebisaa,<br />
magram am<br />
TvisebiT wyal-<br />
Tan SedarebiT<br />
100-jer ufro<br />
sustia, rac ga mowveulia hidroqsilis<br />
jguf ze radikalis<br />
gavleniT. ra dikali amcirebs<br />
O – H bmis po larizacias da<br />
hidroqsilis jgu fis wyalbadatomis<br />
mJavur Tvisebebs:<br />
sur. 26 natriumis ur-<br />
TierTqmedeba spirtTan<br />
H :O: H CH 3 → O : H<br />
81
82<br />
CaatareT 26-e suraTze asaxuli reaqcia, kristalizatorSi,<br />
sadac spirtia TandaTan CaasxiT wyali mcire ulufebad. ras<br />
amCnevT? axseniT es movlena.<br />
2 spirti urTierTqmedebs halo gen wyal bad mJavasTan, magaliTad, bromwyal<br />
badmJavasTan. am SemTxvevaSi spir ti reaqciaSi Sedis ara mxolod hidroqsilis<br />
wyalbadatomiT, aramed mTlianad hidroqsilis jgufiT. am reaqciaSi<br />
spirti fuZea, reaqcia Seqcevadia:<br />
27-e suraTze gamo-<br />
saxulia brom eTanis<br />
misa Rebi laborato-<br />
riuli danadgari.<br />
C 2H 5OH + HBr C 2H 5Br + H 2O.<br />
aRwereT:<br />
C2H5OH a) ra pirobebSi mim-<br />
NaBr<br />
dinareobs reaqcia?<br />
H2SO4 b) ra procesebi mimdinareobsmrgvalsur.<br />
27. bromeTanis miReba<br />
Zira kolbaSi?<br />
g) risTvisaa saWiro wylis macivari?<br />
d) ratom agroveben bromeTans yinuliani wylis gamo yenebiT?<br />
3 spirtebis molekulaTaSorisi dehidratacia. es aris iseTi reaqcia,<br />
rodesac wyalwamrTmevi saSualebis TanxlebiT spirtis ori molekulisagan<br />
gamoiyofa wylis erTi molekula. 100-dan 140 0 C-mde gacxelebiT, gogirdmJavas<br />
Tanaobisas, eTilis spirtisagan am reaqciiT miiReba dieTilis eTeri:<br />
C 2H 5 OH +HO C 2H 5<br />
H2SO 4 (konc.)<br />
C2H5 – O – C<br />
t < 140cC<br />
2H5 +H2O. dieTileTeri aqroladi, advilad aalebadi siTxea. kargi gamxsnelia, medicinaSi<br />
gamoiyeneba narkozisTvis.<br />
4 spirtis Sidamolekuluri dehidratacia. es reaqcia ufro maRal<br />
temperaturaze mimdinareobs, vidre molekulaTSorisi dehidratacia, isev da<br />
isev wyalwamrTmevi saSualebebis TanxlebiT. reaqciis Sedegad miiReba alkeni.<br />
ma galiTad, 170 0 C-ze, gogirdmJavas Tanaobisas, eTilis spirtisagan miiReba<br />
eTileni:<br />
CH 3 – CH 2 – OH H 2SO 4 (konc)<br />
t > 140 0 C CH 2 = CH 2 +H 2O.<br />
wyali da<br />
yinuli<br />
C 2H 5Br<br />
5 spirtis urTierTqmedeba organul mJavasTan. am reaqciis Sedegad miiReba<br />
esteri, amitom reaqcias esterifikaciis reaqcia ewodeba. is mimdinareobs
Zlieri araorganuli mJavas Tanaobisas, romelic katalizatoris funqcias<br />
asrulebs. zogadi saxiT reaqciis toloba ase iwereba:<br />
O<br />
O<br />
kat.<br />
R′ – C + H O – R′′ R′ – C + H2O. mJava O – R′′<br />
OH spirti<br />
esteri<br />
6 spirtis daJangva. Cveulebrivad,<br />
spirti iJangeba damJangavebis moqmedebiT,<br />
magaliTad kaliumis permanganatiT.<br />
rogorc wesi, damJangavi nivTiereba moxleCs<br />
spirtis molekulidan wyalbadis or<br />
atoms, erTs hidroqsilis jgufidan da<br />
meores hidroqsiliani naxSirbadatomidan. pirveladi spirtis daJangvisas<br />
miiReba Sesabamisi aldehidi:<br />
miReba<br />
H<br />
CH 3 – C – O H [O]<br />
H<br />
eTilis spirti<br />
‑H2O CH O<br />
3 – C<br />
H<br />
Zmaraldehidi<br />
1 halogenalkanis hidrolizi. rogorc ukve aRvniSneT, halogenwyalbadTan<br />
spirtis urTierTqmedebis reaqcia Seqcevadia. e. i. reaqciis Sedegad<br />
warmoqmnili halogenalkani urTierTqmedebs wyalTan da kvlav spirti miiReba.<br />
Tu wylis nacvlad tutis wyalxsnars aviRebT, reaqcia Seqcevadi aRar iqneba:<br />
R – Cl+NaOH spirtuli<br />
duRili R – OH + NaCl.<br />
2 alkenis hidratacia. kidev erTxel SegaxsenebT eTilenTan wylis mier-<br />
Tebis reaqcias, is mimdinareobs π-bmis gaxleCis Sedegad da miiReba eTanoli:<br />
CH2 = CH2 + H2O t, H3PO4 C2H5OH. eTileni eTilis spirti<br />
zogierTi spirti gansakuTrebuli xerxiT miiReba. magaliTad, aRsaniSnavia<br />
eTilis spirtis miRebis uZvelesi xerxi – glukozis spirtuli duRili:<br />
C 6H 12O 6 spirtuli<br />
duRili 2C2H 5OH+2CO 2↑.<br />
meoreuli spirtis da-<br />
Jangvisas miiReba ketoni.<br />
[O]<br />
CH3 – C H – CH3 O H<br />
‑H2O CH3 – C – CH3 O<br />
propanoli – 2 acetoni<br />
spirtebidan gansakuTrebuli praqtikuli mniSvneloba aqvs meTanols da<br />
eTanols. amitom swored am spirtebs ganvixilavT dawvrilebiT.<br />
meTanoli CH 3OH – ufero siTxea, romelsac alkoholis susti suni<br />
aqvs, duRs 64,7 0 C-ze, iwvis odnav mocisfro aliT. meTanolis istoriuli<br />
saxelwodebaa – xis spirti. es saxelwodeba ukavSirdeba misi miRebis xerxs<br />
– meTilis spirti tradiciulad xis merqnis mSrali gamoxdiT miiReboda.<br />
meTanoli Zlieri sawamlavia, fermentebis gavleniT organizmSi gardaiqm-<br />
83
84<br />
neba formaldehidad da WianWvelmJavad. es nivTierebebi<br />
ki azianebs Tvalis baduras, iwvevs mxedvelobis nervis<br />
dazianebas da mxedvelobis srul dakargvas.<br />
meTilis spirtis didi raodenoba gamoiyeneba formaldehidis<br />
misaRebad, romelsac Tavis mxriv iyeneben plastmasebis<br />
warmoebaSi. amJamad, araerTi organuli naerTis<br />
miRebis teqnologiuri procesis safuZveli swored meTanolia,<br />
amitom masze moTxovnileba TandaTan izrdeba.<br />
! o r g a n i z m S i<br />
m o x v e d r i l i<br />
50 ml-ze me ti<br />
meTilis spirti<br />
iwvevs sikvdils.<br />
eTanoli C 2H 5OH – aseve ufero, Taviseburi sunis mqone siTxea. duRs<br />
78,3 0 C-ze. kargad iwvis, nebismieri TanafardobiT ixsneba wyalSi. sufTa samedicino<br />
spirtSi 96% (moculobiTi procenti) spirti da 4% (moculobiTi<br />
procenti) wyalia. uwylo absoluturi spirtis misaRebad samedicino spirts<br />
`aTavisufleben~ wylisagan, amisaTvis mas amuSaveben iseTi nivTierebebiT,M<br />
romlebic qimiurad urTierTqmedebs wyalTan. magaliTad, kalciumis oqsidiT<br />
an uwylo spilenZis sulfatiT.<br />
eTanoli farTod gamoiyeneba mrewvelobaSi sinTezuri kauCukis, samkurnalo<br />
preparatebis da sxvaTa dasamzadeblad. is gamoiyeneba gamxsnelad,<br />
Sedis laqebisa da saRebavebis, parfiumeruli saSualebebis SedgenilobaSi.<br />
medicinaSi spirti gamoiyeneba rogorc sadezinfeqcio saSualeba, kvebis<br />
mrewvelobaSi ki misgan alkoholur sasmelebs amzadeben.<br />
1. rogoria spirtis struqtura, Tu Umisi saxelwodebaa:<br />
a) butanoli – 2; b) buten-3-oli-1; g) penten-4-dioli-1,2?<br />
2. ratom ar arsebobs meoTxeuli spirti? pasuxi daasabuTeT.<br />
3. SeadgineT struqturuli formula yvela im spirtisa, romelTa molekuluri<br />
formulaa C 5H 12O. aseTive molekuluri formula SeiZleba hqondes or<br />
sxva nivTierebasac, romelic ar aris spirti, SeadgineT maTi struqturuli<br />
formulebic.<br />
<br />
4. daasaxeleT nivTierebebi, romelTa struqturuli formulebia:<br />
a)<br />
CH 3<br />
CH 3 – C – OH ;<br />
CH 3<br />
b) CH3 – CH – CH – OH ;<br />
CH 3 CH 3<br />
g) CH 3 – CH = CH – CH 2 – OH ; d) CH 3 – CH – CH – CH 3 .<br />
5. SeadgineT 2-propenoli –1 –is:<br />
a) hidrirebis; b) hidrataciis;<br />
g) halogenirebis; d) hidrohalogenebis reaqciaTa tolobebi.<br />
daasaxeleT reaqciis produqtebi.<br />
OH<br />
C 2H 5
6. SeadgineT sqemis Sesabamisi reaqciaTa tolobebi:<br />
a) eTani → bromeTani → eTanoli → eTileni → acetileni → benzoli;<br />
b) kalciumis karbidi → acetileni → benzoli → cikloheqsani → qlorcikloheqsani.<br />
7. ra qimiuri Tvisebebi unda hqondes nivTierebas, Tu misi struqturuli<br />
formulaa H 2C=CH – CH 2OH?<br />
SeadgineT saWiro reaqciaTa tolobebi.<br />
8. gamoiangariSeT wyalbadis moculoba (n.p.), romelic SeiZleba gamoiyos,<br />
Tu sareaqciod aRebulia 1 moli meTilis spirti da 1,5 moli metaluri kaliumi.<br />
<br />
9. gaaanalizeT 28-e suraTze aRwerili procesi. ivaraudeT, C 2H 6O Sedgenilobis<br />
nivTiereba spirtia, Tu eTeri. varaudi daasabuTeT gaangariSebiT.<br />
natriumi<br />
C 2H 6O<br />
wyali<br />
0,05 moli<br />
wyalbadi<br />
wyali<br />
sur. 28<br />
wyali<br />
0,56 litri<br />
85
86<br />
14<br />
mravalatomiani spirtebi<br />
oratomiani spirtia eTilenglikoli, anu eTandioli:<br />
CH 2 _OH<br />
|<br />
CH 2 – OH<br />
samatomiani spirtia glicerini, anu propantrioli:<br />
CH 2 _OH<br />
|<br />
CH _OH<br />
|<br />
CH 2 _OH<br />
cxadia, arsebobs iseTi spirtic, romlis molekulaSic samze meti hidroqsilis<br />
jgufia. aseTia magaliTad, eqvsatomiani spirti – sorbiti, romelic<br />
gamoiyeneba Saqris Semcvlelad imitom, rom tkbilia:<br />
HO – CH2 – CH – CH – CH – CH – CH2 – OH<br />
| | | |<br />
OH OH OH OH<br />
mravalatomian spirtebs didi praqtikuli gamoyeneba aqvs.<br />
eTilenglikoli gamoiyeneba antifrizis dasamzadeblad. antifrizi iseTi<br />
siTxea, romelic Znelad iyineba da am Tvisebis gamo mas avtomobilis radiatorSi<br />
asxamen. eTilenglikoli gamoiyeneba agreTve zogierTi organuli niv-<br />
Tierebebis sinTezisaTvis. magaliTad, maRalmolekuluri sinTezuri boWkos<br />
misaRebad. is gamoiyeneba agreTve feTqebadi nivTierebebis dasamzadeblad.<br />
glicerini gamoiyeneba nitroglicerinis misaRebad. nitroglicerini<br />
ZLli eri asafeTqebeli nivTierebaa. medicinaSi nitroglicerini gamoiyeneba<br />
rogorc sisxlZarRvTa gafarToOebis saSualeba gulis ukmarisobis dros.<br />
glicerini gamoiyeneba parfiumeruli saSualebebis, samedicino preparatebis<br />
dasamzadeblad, tyavis warmoebaSi. is icavs tyavs gamoSrobisagan.<br />
gamoiyeneba agreTve qsovilebis dasamuSaveblad. gliceriniT damuSavebuli<br />
qsovili rbili da elastikuria.<br />
oratomiani spirtebis miReba<br />
oratomiani spirti miiReba:<br />
1 eTilenis daJangvisas kaliumis permanganatis xsnariT:<br />
CH 2 =CH 2 +[O] +H 2O HO – CH 2 – CH 2 – OH<br />
eTileni eTilenglikoli<br />
2 dihalogenalkanis hidroliziT. najeri naxSirwyalbadis dihalogennawarmis<br />
wyalTan urTierTqmedebiT miiReba eTilenglikoli:<br />
CH2 _Cl H _OH<br />
+<br />
CH2 _Cl H _OH<br />
CH 2 _OH<br />
CH 2 – OH<br />
+ 2HCl<br />
mravalatomiani<br />
spi rti ise-<br />
T i s p i r t i a ,<br />
romlis mo lekulaSicramdenimehidroqsilis<br />
jgu fia.<br />
wonasworoba marjvniv gadainacvlebs, Tu reaqcias tutis wyalxsnarSi CavatarebT.
a reaqtivebi iqneba saWEiro analogiuri reaqciiT glicerinis misa Rebad?<br />
SeadgineT reaqciis toloba.<br />
mravalatomiani spirtebis fizikuri Tvisebebi<br />
eTilenglikoli da glicerini ufero, blanti siTxeebia, motkbo gemo aqvT<br />
da kargad ixsnebian wyalsa da eTanolSi. eTilenglikoli duRs 197,6 0 C-ze.<br />
glicerini ki – 290 0 C-ze.<br />
qimiuri Tvisebebi<br />
1 eTilenglikolisa da glicerinis qimiuri<br />
Tvisebebi erTatomiani spirtis qimiuri Tvisebebis<br />
msgavsia, magaliTad, isini urTierTqmedeben<br />
aqtiur metalebTan:<br />
CH 2 – O H + 2 Na →<br />
CH 2 – O H<br />
CH 2 – O – Na<br />
CH 2 – O – Na + H 2↑.<br />
SeadgineT eTilenglikolis halogenwyalbad-<br />
Tan urTierTqmedebis reaqciis toloba.<br />
eTilenglikoli sawamlavia.<br />
amitom frTxilad unda movepyroT<br />
antifrizis xsnars,<br />
romelic eTi lenglikolis<br />
w y a l x s n a r i a . e T i l e n g -<br />
l i k o l i s S e x e b a s a k v e b<br />
produqtebTan dauSvebelia.<br />
2 mravalatomian spirtebs specifikuri, mxolod maTTvis damaxasiaTebeli<br />
Tvisebebic aqvs. aseTia magaliTad reaqcia spilenZ(II)-is hidroqsidTan.<br />
Tu spilen(II)-is hidroqsidze vimoqmedebT gliceriniT (tutis Tanaobisas)<br />
da narevs SevanjRrevT, naleqi gaixsneba da miiReba lurji feris xsnari – es<br />
spilenZis gliceratis xsnaria (sur. 29) .<br />
CH 2 – O – H<br />
CH – O – H<br />
CH 2 – OH<br />
glicerini<br />
spilenZ(II)-is<br />
hidroqsidi<br />
H – OO<br />
+<br />
H – OO<br />
Cu →<br />
CH 2 – O<br />
CH – O<br />
CH 2 – OH<br />
sur.29 glicerinis urTierTqmedeba<br />
spilen(II)-is hidroqsidTan.<br />
spilenZis<br />
glicerati<br />
C u<br />
+2H 2O<br />
am reaqciidan Cans,<br />
rom mravalatomian<br />
spirts susti, magr<br />
a m e r T a t o m i a n z e<br />
uf ro ZlieriA mJava<br />
buneba aqvs. es aixsneba<br />
molekulaSi OH<br />
jgufebis dagrovebiTa<br />
da maTi urTierTgavleniT,<br />
rac wyalbadatomebs<br />
ufro moZravs<br />
xdis.<br />
spilen(II)-is glicerati mocemulia<br />
gamartivebuli saxiT.<br />
faqtobrivad spilenZis glicerats<br />
ufro rTuli Sedgeniloba<br />
aqvs.<br />
87
88<br />
3 glicerini urTierTqmedebs azotmJavasTan. am reaqciis Sedegad miiReba<br />
nitroglicerini – azotmJavasa da glicerinis esteri:<br />
CH 2 – OH<br />
CH – OH<br />
CH 2 – OH<br />
+<br />
H – OO – ONO 2<br />
H – OO – ONO 2<br />
H – OO – ONO 2<br />
H 2 SO 4<br />
CH2 – O – NO2 CH – O – NO2 + 3H2O CH 2 – O – NO 2<br />
trinitroglicerini<br />
saxelwodeba nitrogliceriniistoriulia<br />
da ar<br />
Seesabameba esteris<br />
saxelwodebis<br />
Sedgenis saerTa-<br />
Soriso wesebs.<br />
1. SeadgineT naerTis struqturuli formula, Tu misi saxelwodebaa<br />
butantrioli-1,2,4 .<br />
<br />
2. SeadgineT sqemis Sesabamis reaqciaTa tolobebi:<br />
a) CH 3 – CH 3 → CH 3 – CH 2 – Cl → CH 2 = CH 2→ HO – CH 2 – CH 2 – OH;<br />
b) HO – CH 2 – CH 2 – OH → Cl – CH 2 – CH 2 – Cl → CH ≡ CH →CH 3 – CH 3→<br />
CH 3 – CH 2Cl → CH 3 – CH 2 – OH → CH 2 = CH 2 → HO – CH 2 – CH 2 – OH.<br />
<br />
3. mocemulia organuli naerTTa formulebi.<br />
daadgineT:<br />
– ramdeni sxvadasxva nivTierebaa gamosaxuli?<br />
– ramdeni homologia? SeadgineT TiToeulis saxelwodeba.<br />
– ramdeni izomeria? SeadgineT TiToeulis saxelwodeba.<br />
– aris Tu ara sxvadasxvanairad gamosaxuli erTi da igive nivTiereba.<br />
H H<br />
a)<br />
b) H3C – CH2 – CH – CH<br />
g) H3C – CH – CH3 O O<br />
3<br />
OH<br />
OH<br />
H2C – CH2 d) HO – CH2 HO – CH<br />
HO – CH 2<br />
e) HO – CH 2<br />
HO – CH<br />
HO – CH 2<br />
z) H3C – CH – CH2 – CH – CH3 OH OH<br />
v) H 5C 2 – CH – CH 2<br />
OH OH<br />
T) HO – CH 2 – CH 2 – OH<br />
4. mocemulia Termoqimiuri toloba: C 2H 4 +H 2O → C 2H 5OH +460 kj.<br />
gamoiangariSeT:<br />
a) eTilenis moculoba (n.p.);<br />
b) spirtis raodenoba da masa, Tu cnobilia, rom am procesSi gamoiyo 920 kj<br />
siTbo.
15<br />
TqvenTvis ukve cnobilia, rom hidroqsilis jgufi SesaZlebelia dau-<br />
kavSirdes uSualod benzolis birTvs. naerTTa<br />
im klass, sadac hidroqsilis jgufi uSualod<br />
ukavSirdeba benzolis birTvs, anu fenilis radikals<br />
C 6H 5‑s, fenoli ewodeba.<br />
hidroqsilis jgufis raodenobis mixedviT<br />
fe nolebi SesaZlebelia iyos erTatomiani<br />
da mravalatomiani.<br />
amjerad yuradRebas SevaCerebT fenolebis klasis pirvel warmomadgenelze,<br />
sakuTriv fenolze, romelsac karbolmJavac ewodeba.<br />
mravalatomiani fenolis<br />
molekulaSi er-<br />
Tze meti hidroqsilis<br />
jgufia. magaliTad,<br />
OH<br />
1 2<br />
OH<br />
1,2 – dihidroqsibenzoli<br />
HO<br />
1<br />
OH<br />
2 3<br />
OH<br />
1,2,3 – trihidroqsibenzoli<br />
OH<br />
1<br />
fenoli<br />
5 3<br />
HO OH<br />
1,3,5,-trihidroqsibenzoli<br />
miReba<br />
fenolis misaRebi bunebrivi nedleulia – qvanax-<br />
Siris fisi, magram is Zalian cotaa saimisod, rom<br />
uzrunvelyos fenolze arsebuli moTxovnileba.<br />
SemuSavebulia ramdenime sinTezuri xerxi. erTerTi<br />
maTgani mimdinareobs Semdegi sqemiT:<br />
C 6H 6 → C 6H 5Cl → C 6H 5OH.<br />
aRsaniSnavia is garemoeba, rom halogenis atomi<br />
ufro mtkicedaa dakavSirebuli fenilis radikal-<br />
Tan, vidre alkanis radikalTan. amitom, misi Canacvleba<br />
hidroqsilis jgufiT ufro mkacr pirobebSi<br />
– maRal temperaturasa da wnevaze mimdinareobs.<br />
fenoli gamoiyeneba samkurnalo preparatebis,<br />
feTqebadi nivTierebebis, antiseptikuri saSualebebisa<br />
da sxvaTa dasamzadeblad, bioqimiur laboratoriaSi<br />
ki – dnm-is misaRebad.<br />
fizikuri Tvisebebi<br />
fenoli ufero, kristaluri<br />
nivTierebaa. lRveba 43 0 C-ze.<br />
aqvs mkveTri, Taviseburi (`wamlis~)<br />
suni, momwamlavia.<br />
oTaxis temperaturaze<br />
umniSvnelo raodenobiT<br />
ixsneba wyalSi.<br />
cxel wyal Si ki ixsneba<br />
ga nusazRvreli raode-<br />
nobiT (sur.30). kanze<br />
mox ved risas iwvevs dam-<br />
erTatomiani fenoli<br />
Seicavs hidroqsilis<br />
erT jgufs, ma ga liTad:<br />
OH<br />
fenoli<br />
OH<br />
CH 3<br />
2 – meTilfenoli<br />
fenolis monawileobiT<br />
mimdinare qimiuri<br />
reaqciebi unda Catardes<br />
amwov karadaSi,<br />
damcavi xelTaTmanebis<br />
gamoyenebiT.<br />
civ<br />
wyalSi<br />
cxel<br />
wyalSi<br />
sur. 30. fenolis gaxsna civsa<br />
da cxel wyalSi.<br />
89
90<br />
wvrobas, amitom masTan muSaobisas<br />
saWiroa sifrTxile. organizmSi<br />
moxvedrisas iwvevs nivTierebaTa<br />
cvlis darRvevas.<br />
qimiuri Tvisebebi<br />
radgan fenolis molekulaSi<br />
OH-jgufi uSualodaa dakavSirebuli<br />
fenilis radikalTan, mosalodnelia,<br />
rom mas spirtebisagan<br />
gansxvavebuli Tvisebebi hqondes<br />
(fenilis radikalisa da hidroqsilis<br />
jgufis urTierTgavlenis<br />
gamo). fenolis Tvisebebis ganxilvisas<br />
SesaZlebelia gamoiyos<br />
Tvisebebi, romlebic ganpirobebulia<br />
mis molekulaSi – erTis<br />
mxriv OH jgufis da meores mxriv<br />
– fenilis radikalis arsebobiT.<br />
1 spirtis msgavsad fenoli reaqciaSi<br />
Sedis tute metalebTan:<br />
2C6H5OH + 2Na → 2C6H5ONa + H2 natriumis<br />
fenolati<br />
Ö←H<br />
magram, fenolSi<br />
OH jgufis wyalbada<br />
t o m z e g a v l e n a s<br />
axdens fenilis radikali,<br />
romelsac aqvs unari<br />
Tavisken miizidos hidroqsilis<br />
Jangbadatomis Tavisufali<br />
eleqtronebi. amitom O‑H bma Zalian<br />
polarulia, wyalbadatomi<br />
ki ufro moZravi da reaqciisunariani,<br />
vidre spir tSi. amitom<br />
fenols spirtisagan gansxvavebiT<br />
susti mJava buneba aqvs, rac<br />
gamoixateba imiT, rom fenoli<br />
spirtisagan gansxvavebiT reaqciaSi<br />
Sedis tutesTan:<br />
C 6H 5OH + NaOH → C 6H 5ONa + H 2O.<br />
fenolis molekulaSi fenilis<br />
radikalis arsebobiT ga mowveul<br />
Tvisebebs Soris aRsaniSnavia:<br />
fenolSi mJavuri Tvisebebi Zalian sustadaa<br />
gamoxatuli _ is fers ver ucvlis<br />
indikators, misi gamoZeveba natriumis<br />
fenolatidan SesaZlebelia iseTi susti<br />
mJaviTac ki, rogoric naxSirmJavaa:<br />
C6H5ONa + H2CO3 → C6H5OH + NaHCO3 ,<br />
e. i. fenoli naxSirmJavaze susti mJavaa.<br />
erTatomian spirtebTan SedarebiT fenolSi<br />
mJavuri Tvisebebis gaZliereba hidroqsilis<br />
jgufze benzolis birTvis gavleniT<br />
aixsneba, rac iwvevs Obmis polarobis<br />
gazrdas da O – H hidroqsilis jgufis<br />
wyal badatomis reaqciis unarianobas.<br />
Jangbadatomis gauziarebeli eleqtronuli<br />
wyvili urTierTqmedebs benzolis<br />
bir Tvis π-eleqtronul sistemasTan, ris<br />
gamoc Jangbadis eleqtronuli wyvi li<br />
nawilobriv wainacvlebs C‑O bmis nax Sirbad<br />
atomisaken, Jangbadis atomi miiswrafvis<br />
Tavisi eleqtronuli muxtis kompensaciisaken,<br />
amitom is Tavisken Zlier<br />
waanacvlebs O‑H bmis eleqtronul wyvils,<br />
ris Se degadac wyalbadis atomze dadebiTi<br />
muxti izrdeba da advildeba misi moxleCa<br />
protonis saxiT.<br />
δ<br />
6<br />
–<br />
5<br />
H<br />
:Ο:<br />
δ+<br />
benzolis birTvisaken Jangbadis eleqtronuli<br />
wyvilis wanacvleba iwvevs eleqtronuli<br />
muxtis (simkvrivis) gadanawilebas<br />
benzolis birTvis π – ele q tronul<br />
sis temaSi da mis koncentrirebas 2, 4 da 6<br />
mdgo mareobaSi, am mdgomareobaSi wyalbada<br />
tomebi advilad Canacvldeba sxva atomebiT.<br />
maSasadame, hidroqsilis jgufi, Tavis<br />
mxriv, gavlenas axdens benzolis birTvze,<br />
ris gamoc advildeba Canacvlebis reaqcia.<br />
marTlac, benzolis bromirebisaTvis sa-<br />
Wi roa katalizatori da Tavisufali bromi,<br />
fenolis bromireba ki mimdinareobs katalizatoris<br />
gareSe, bromiani wyliT da<br />
miiReba tribromfenoli – TeTri feris<br />
naleqis saxiT.<br />
1<br />
4<br />
δ –<br />
2<br />
3<br />
δ –
2 bromirebis reaqcia. hidroqsilis jgu fis gavlenis gamo fenolSi benzolis<br />
birTvis bromireba ufro advilad mimdinareobs, vidre benzolisa:<br />
OH<br />
+ 3Br 2<br />
fenoli<br />
Br<br />
2,4,6 – tribromfenoli<br />
3 nitrirebis reaqcia. aseve advilad mimdinareobs nitrirebis reaqcia.<br />
fenolze koncentrirebuli azotmJavas moqmedebiT miiReba 2,4,6 – trinitrofenoli<br />
– feTqebadi nivTiereba, romelsac pikrinis mJava ewodeba.<br />
OH<br />
+ 3HNO 3<br />
Br<br />
O 2N<br />
OH<br />
OH<br />
NO 2<br />
Br<br />
+ 3HBr<br />
NO 2<br />
+ 3H 2O<br />
fenoli 2,4,6 – trinitrofenoli<br />
4 fenolis polikondensacia. fenolisa da formaldehidis urTierTqmedebisas<br />
gamoiyofa wylis molekula, fenolis naSTebi ki erTmaneTs ukav-<br />
Sirdeba. am reaqcias polikondensaciis reaqcia ewodeba.<br />
es reaqcia SeiZleba gamovsaxoT Semdegi sqemiT:<br />
H H<br />
OH<br />
C<br />
OH<br />
OH<br />
OH<br />
H<br />
H<br />
O<br />
+<br />
H H<br />
– H2O H<br />
CH2 H<br />
dimeri<br />
Tu moreagire nivTierebebi sakmarisi raodenobiTaa, reaqcia kidev<br />
SeiZleba gagrZeldes da warmoiqmnas ara mxolod dimeri, aramed makromolekula,<br />
romelsac fenolformaldehiduri fisi ewodeba.<br />
Cveulebriv temperaturaze warmoiqmneba wrfivi agebulebis makromolekulebi,<br />
xolo Tu reaqcias maRal temperaturaze CavatarebT, miRebuli<br />
produqti daitoteba da miiReba badiseburi struqturis polimeri:<br />
OH<br />
OH<br />
OH<br />
CH 2<br />
OH<br />
CH 2<br />
CH 2<br />
CH 2<br />
OH<br />
CH 2<br />
CH 2<br />
CH 2<br />
OH<br />
91
92<br />
fenolformaldehiduri fisisagan amzadeben plastmasebs, romlebic cecxlgamZle<br />
da yinvamedegia. is uZlebs wylis, mJavisa da tutis moqmedebas,<br />
aqvs dieleqtrikuli Tvisebebi. fenolformaldehiduri fisisagan miiReba<br />
agreTve unikaluri Tvisebebis webo, romelic awebebs metalisagan damzadebul<br />
detalebsac ki da rac mTavaria uzrunvelyofs `gadabmis adgilis~<br />
simtkices.<br />
1. SeadgineT sqemis Sesabamisi reaqciaTa tolobebi:<br />
CH4 → C2H2 → C6H6 →C6H5OH →2,4,6 tribromfenoli.<br />
2. ori uetiketo WurWlidan erTSi fenolia, meoreSi ki – eTilis spirti.<br />
rogor ganvasxvavoT isini mxolod fizikuri Tvisebebis mixedviT? daasabuTeT<br />
Tqveni varaudi.<br />
<br />
3. saxelwodebis mixedviT SeadgineT Semdegi nivTierebebis struqturuli<br />
formulebi:<br />
a) 1,2,3 – trihidroqsibenzoli; b) 1,4 – dihidroqsibenzoli.<br />
axseniT, SeiZleba Tu ara es nivTierebebi miviCnioT:<br />
a) izomerebad; b) homologebad?<br />
4. SeadgineT sqemis Sesabamisi reaqciaTa tolobebi:<br />
C6H6 → C6H5 Cl → C6H5OH. 5. SeadareT benzolisa da fenolis qimiuri Tvisebebi, Sedarebis Sedegebi<br />
gamosaxeT diagramiT.<br />
axseniT msgavsebisa da gansxvavebis mizezebi. gansxvaveba msgavseba gansxvaveba
16<br />
gansakuTrebiT gogonebisaTvis kargad unda iyos<br />
nacnobi im siTxis suni, romelsac frCxilebidan lakis<br />
mosacileblad iyeneben, es aqroladi siTxe acetonia. acetoni<br />
ketonebis klass ganekuTvneba da yvela sxva ketonis<br />
msgavsad karbonilis jgufs Seicavs. karbonilis jgufs<br />
Seicavs aldehidic.<br />
wyalbadis atomTan dakavSirebul karbonilis<br />
jgufs aldehidis jgufs uwodeben.<br />
erTi Tavisufali savalento xaziT karbonilis jgufi<br />
SeaerTeT wyalbadis atomTan, meoreTi ki – R-iT aRniSNul<br />
naxSirwyalbadis radikalTan da Tqven aldehidis zogad<br />
formulas miiRebT.<br />
umartivesi aldehidi formaldehidia, sadac karbonilis jgufi dakavSirebulia<br />
mxolod wyalbadatomTan:<br />
organul nivTie<br />
rebas, romlis<br />
m o l e k u l a S i<br />
k a r b o n i l i s<br />
j g u f i d a k a v -<br />
Sirebulia or<br />
naxSir wyal badis<br />
radikalTan, ketoni<br />
ewodeba.<br />
aldehidebi da ketonebi<br />
H<br />
C = O<br />
H<br />
zogjer karbonilis jgufi SeerTebulia or radikalTan.<br />
aseT organul nivTierebas ewodeba ketoni.<br />
umartivesi ketoni praqtikidan TqvenTvis kargad nacnobi<br />
nivTiereba ace tonia. mis molekulaSi karbonilis jgufi dakavSirebulia<br />
meTilis or radikalTan:<br />
izomeria da nomenklatura<br />
aldehidi da ketoni<br />
organuli niv Ti ere<br />
bebia, ro me lTa<br />
mole ku laSic Sedis<br />
nax Sirwyalbadis<br />
ra dikalTan da kav-<br />
Si rebuli<br />
aRniSneT radikalebi R 1-iT da R 2-iT. savalento xazebiT<br />
SeaerTeT karbonilis jgufTan. aseT SemTxvevaSi Tqven<br />
ketonis zogad formulas miiRebT.<br />
O<br />
CH3 – C – CH3 acetoni<br />
SeafaseT aldehidebi naxSirwyalbadis radikalis mixedviT. sqema gadaixa-<br />
zeT rveulSi da rgolis Sig niT CawereT saTanado magaliTebi. es magaliTe-<br />
bi Se arCieT Cvens mier SemOoTavazebuli aldehidebis formulebidan.<br />
romeli maT ga nis Se sa xeb Se giZ li aT TqvaT dabejiTebiT _ `gaau<br />
fe rulebs bromian wyals~. axseniT, ratom fiqrobT ase:<br />
CH 2=CH – C<br />
O<br />
H<br />
CH3 – C O<br />
H<br />
C = O<br />
karbonilis<br />
jgufi.<br />
C<br />
O<br />
H<br />
93
94<br />
saerTaSoriso nomenklaturis mixedviT najeri aldehidis saxelwodeba rom<br />
SevadginoT, naxSirbadatomTa imave ricxvis mqone alkanis saxelwodebas unda<br />
mivumatoT sufiqsi – `ali~, magaliTad,<br />
O<br />
H – C<br />
H<br />
meTanali<br />
saerTaSoriso nomenklaturis paralelurad xSirad iyeneben aldehidis<br />
trivialur saxelwodebas. rogorc wesi, aseTi saxelwodeba momdinareobs<br />
aldehidis Sesabamisi mJavas saxelwodebidan (ix. cxrili).<br />
cxrili: zogierTi mJavas struqturuli formula da saxelwodeba<br />
saerTaSoriso<br />
saxelwodeba<br />
O<br />
CH3 – C<br />
H<br />
eTanali<br />
aldehidebs axasiaTebs mxolod<br />
erTi – naxSirbadovani<br />
ConCxis, izomeria.<br />
formula trivialuri saxelwodeba<br />
meTanali HCHO WianWvelaldehidi (formaldehidi)<br />
eTanali CH 3CHO Zmaraldehidi<br />
propanali CH 3CH 2CHO propionaldehidi<br />
propenali CH 2=CHCHO akroleini<br />
butanali CH 3CH 2CH 2CHO erboaldehidi<br />
pentanali CH 3CH 2CH 2CH 2CHO valerianaldehidi<br />
benzaldehidi C 6H 5CHO benzaldehidi<br />
fizikuri Tvisebebi<br />
aldehidi<br />
najeri ujeri aromatuli<br />
. . . . . . . . .<br />
CH3 – CH2 – C<br />
propanali<br />
ZiriTadi jaWvis naxSirbadatomTa danomvra<br />
iwyeba aldehidis jgufis naxSirbadatomidan.<br />
amitom, aldehidis jgufis<br />
mdebareobis cifriT miTiTeba saWiro ar<br />
aris, is yovelTvis pirvelia.<br />
aldehidebis fizikur TvisebebSi gasarkvevad yuradReba gavamaxviloT<br />
maTi molekulis eleqtronul agebulebaze. imis gamo, rom naxSirbadTan<br />
SedarebiT Jangbadi gacilebiT eleqtrouaryofiTi elementia, C = O bmis<br />
O<br />
H<br />
CH 3 – CH – CH 2 – C<br />
CH 3<br />
3-meTilbutanali<br />
O<br />
H
eleqtronuli simk vrive gadaweulia Jangbadisken, amitom bma Zalian polarulia:<br />
δ<br />
C = O<br />
+<br />
δ –<br />
Sesabamisad, aldehidi polaruli naerTia, rac aisaxeba kidevac mis TvisebebSi:<br />
aldehidebis pirveli warmomadgenlebi – formaldehidi, Zmaraldehidi<br />
wyalSi xsnadia. maTi duRilis temperatura ufro dabalia, vidre Sesabamisi<br />
spirtebis, rac imiT aixsneba, rom spirtebisagan gansxvavebiT aldehidebSi<br />
wyalbaduri bmebi ar arsebobs. moklejaWvian aldehidebs arasasiamovno<br />
suni aqvs. grZeljaWvian aldehidebsa da ketonebs ki yvavilis suni aqvs<br />
da amitomac gamoiyeneba parfiumeriaSi.<br />
qimiuri Tvisebebi<br />
aldehidi ZiriTadad Sedis daJangvisa da mierTebis reaqciaSi.<br />
1 daJangvis reaqcia. aldehidis daJangviT warmoiqmneba Sesabamisi mJava,<br />
sqematurad es procesi ase gamoisaxeba:<br />
R – C O<br />
H<br />
R – C O<br />
OH<br />
TvisebiTi reaqcia<br />
reaqciis Sedegad warmoqm-<br />
aldehidis daJangva advilad mimdinareobs<br />
da SesaZlebelia susti damJangaviT – vercxnili<br />
metaluri vercxli Txlad<br />
efineba WurWlis kedlebs, miiReba<br />
vercxlis sarke. amitomac,<br />
lis oqsidis amiakiani xsnariT daiJangos. am reaqcias `vercxlis sarkis~<br />
ga mar tivebulad es procesi ase gamoisaxeba reaqcia ewodeba. rogorc wesi,<br />
(sur. 31):<br />
aldehidis aRmosaCenad am reaqcias<br />
iyeneben.<br />
CH3– C O<br />
CH3 – C<br />
H<br />
O<br />
+Ag2O →<br />
+2Ag<br />
OH<br />
eTanali eTanmJava<br />
ufro zustad reaqcia ase ga moisaxeba:<br />
R– C O<br />
H +2[Ag(NH3) 2]OH → R– C<br />
ONH4 O +2Ag+3NH3+H2O reaqciis toloba SegiZliaT ar da imaxsovroT.<br />
aldehidis daJangva SesaZlebelia spilenZ(II)is<br />
hidroqsidiTac. is Jangavs aldehidebs,<br />
TviTon ki aRdgeba spilenZ(I)-is hidroqsi dam de<br />
CuOH-is saxiT. es aramdgradi yviTeli feris<br />
naerTi ki maSinve iSleba spilenZ(I)-is oqsidis<br />
warmoqmniT, romelsac wiTeli feri aqvs<br />
(sur. 32):<br />
R – C<br />
O<br />
+ 2Cu(OH) 2<br />
H<br />
lurji<br />
R – C<br />
O<br />
OH<br />
+ Cu 2O↓ + 2H 2O<br />
wiTeli<br />
aldehidisa da vercxlis oqsidis<br />
amikiani xsnaris narevi<br />
aldehidi<br />
sur. 31. vercxlis sarkis<br />
warmoq mnis procesi<br />
vercxlis<br />
sarke<br />
sur. 32. spilen(I)-is hidroqsidi,<br />
rome lic iSleba da warmoqmnis<br />
spi lenZ(I)-is oqsids.<br />
95
96<br />
2 mierTebis reaqcia. mierTebis reaqcias ganekuTvneba aldehidis hid-<br />
rirebis reaqcia, romelic mimdinareobs<br />
karbonilis jgufis ormagi<br />
bmis gaxleCis Sedegad. aldehidis<br />
hidrirebisas miiReba pirveladi<br />
spirti:<br />
CH3 – C<br />
H<br />
O + H2 Ni CH3 – CH2 – OH<br />
miReba<br />
laboratoriaSi aldehidis miRebis xerxSi gasarkvevad CaatareT aseTi cda:<br />
sinjaraSi CaasxiT 4-5 ml eTilis spirti.<br />
spilenZis mavTulisagan daamzadeT spira-<br />
c<br />
li da gaavarvareT spirtquris alze, vidre<br />
d<br />
a<br />
ar gaSavdeba, ris Semdegac CauSviT spirtian<br />
sinjaraSi.<br />
ras amCnevT? _ ra argumentebi gaqvT<br />
imis dasasabuTeblad, rom namdvilad aldehidi miiReba?<br />
zogadi saxiT daJagvis procesi ase gamoisaxeba:<br />
O<br />
R – CH2 – O – H + [O] → R – C<br />
H<br />
+ H2O. mrewvelobaSi aldehids iReben sxvadasxva xerxiT.<br />
1 meTanalis miRebis erT-erTi yvelaze ufro ekonomiuri xerxia meTanis<br />
daJangva specialur reaqtorSi haeris JangbadiT:<br />
H<br />
H – C – H + O2 H<br />
500 0 C, kat.<br />
H – C<br />
O<br />
+ H2O. H<br />
2 meTanali miiReba agreTve meTanolis daJangviT. reaqciis procesSi<br />
meTano lis orTqlis narevs haerTan atareben gavarvarebuli vercxlis an<br />
spilenZis badeze:<br />
2CH 3OH + O 2<br />
Cu an Ag<br />
ketonis hidrirebisas miiReba meoreuli<br />
spirti:<br />
O OH<br />
CH3 – C – CH3 + H2 Ni<br />
CH3 – CH – CH3 2H – C<br />
O<br />
+ 2H2O. H<br />
3 dReisaTvis eTanalis miReba SesaZlebelia eTilenis daJangviT. eTilens<br />
Jangaven haeris JangbadiT paladiumisa da spilenZis qloridebis Tanaobisas:<br />
O<br />
kat<br />
2CH2=CH2+O2 2CH3 – C<br />
H
es xerxi sakmaod ekonomiuria, radgan eTileni iafi nedleulia.<br />
aldehidebidan gansakuTrebuli praqtikuli Rirebuleba aqvs<br />
WianWvelaldehidsa da Zmaraldehids.<br />
WianWvelaldehidi, anu formaldehidi HCHO – ufero,<br />
mZafri sunis mqone momwamlavi airia, wyalSi kargad<br />
ixsneba, formaldehidis wyalxsnars (40%-ians) formalini<br />
ewodeba, mas sadezinfeqciod iyeneben. formaldehidisagan<br />
iReben urotropins, urotropini samkurnali preparatia.<br />
dapresil mdgomareobaSi, briketis saxiT mas sawvavadac<br />
iyeneben (mSrali spirti). yvelaze mniSvnelovani ki is aris,<br />
rom formaldehidi gamoiyeneba fenolformaldehiduri<br />
fisebisa da zogierTi sxva nivTierebis misaRebad.<br />
Zmaraldehidi CH 3CHO, mZafri, arasasiamovno sunis<br />
mqone advilad aqroladi siTxea. wyalSi kargad ixsneba,<br />
mrewvelobaSi Zmaraldehididan iReben plastmasebsa da<br />
acetatur boWkos.<br />
<br />
1. struqturuli formulebis mixedviT SeadgineT Semdegi nivTierebebis<br />
saxelwodebebi:<br />
a)<br />
CH 3 – CH 2 – CH – C<br />
CH 3<br />
O<br />
g) CH2 = CH – CH2 – C<br />
H<br />
2. gadaixazeT rveulSi da SeavseT cxrili:<br />
saxelwodeba najeri naxSir wya lbadis<br />
molekuluri<br />
formula<br />
meTani<br />
eTani<br />
propani<br />
butani<br />
pentani<br />
heqsani<br />
O<br />
H<br />
b)<br />
Br – CH 2 – CH 2 – C<br />
O<br />
H<br />
m o l e k u l u r i f o r m u l a<br />
sur.33. formalini<br />
anadgurebs mikrobebs<br />
ris gamoc masSi inaxaven<br />
cocxal organizmebs.<br />
Sesabamisi Espir tis Sesabamisi aldehidis Sesabamisi mJavas<br />
97
98<br />
<br />
3. SeadgineT sqemis Sesabamisi reaqciaTa tolobebi:<br />
O<br />
a) CH4 → C2H2 → CH3 – C → CH3 – CH2OH → CH3CH2Br → C2H4 →<br />
H<br />
O<br />
→ CH3CH2OH → CH3 – C<br />
H<br />
O<br />
b) CH4 → H – C → CH3OH → CH3Cl → C2H6 → C2H5OH →<br />
H<br />
CH3 – C<br />
4. SeadgineT reaqciis toloba, romelic Seesabameba:<br />
a) propanoli – 1-is daJangvas;<br />
b) propanoli – 2-is daJangvas;<br />
ra gansxvavebaa reaqciis produqtebs Soris?<br />
vercxlis sarkis reaqcia<br />
O<br />
H<br />
praqtikumi<br />
aiReT sinjara, CaasxiT 1 ml formalini (formaldehidis wyalxsnari),<br />
daamateT ramdenime wveTi vercxlis oqsidis amiakiani xsnari. sinjara odnav<br />
SeaTbeT spirtquris alze.<br />
1. ras amCnevT?<br />
2. ratom xdeba sinjaris kedlebi sarkiseburi?<br />
3. SeadgineT reaqciis toloba.
Tu<br />
siyvarulis qimia<br />
TiToeuli cocxali organizmi organoe-<br />
bisa da qsovilebis rTuli, gansakuTreb-<br />
uli da unikaluri sistemaa, romelTagan<br />
TiToeuli Tavis aucile bel, specifikur<br />
funqcias asrulebs. am funqciis Sesrulebas<br />
ki ganapirobebs organizmSi im nivTierebebis<br />
arseboba, romelic gamomuSavdeba Sinagani sekreciis jirkvlebiT. am<br />
nivTierebebs hirmonebi ewodeba.<br />
cocxali samyaro Zalian mravalferovania, magram ZiriTadad is mainc or<br />
nawilad iyofa _ mamrebad da mdedrebad.<br />
or nawilad iyofa kacobriobac _ qalebad da mamakacebad.<br />
qalis sasqeso hormonebs estrogenebi ewodeba, mamakacisas ki _ androgenebi.<br />
estrogenis molekulaSi 18 naxSirbadatomia. estrogenebidan aRsa niSnavia<br />
C18H24O2 _ estradioli:<br />
molekulis aRnagoba cxadyofs, rom estradioli SesaZlebelia mivakuTvnoT<br />
erTdroulad spirtebsac da fenolebsac.<br />
estrogenebs miekuTvneba nivTiereba estronic, is erTdroulad feno licaa<br />
da ketonic:<br />
C 18H 22O 2 _ estroni<br />
androgeni _ mamakacis sasqeso hormonia. aseTi hormonis molekulaSi 19<br />
naxSirbadis atomia.<br />
gainteresebT...<br />
erT-erTi yvelaze mniSvnelovani androgeni testosteronia.<br />
99
100<br />
testosteroni<br />
hormonebidan aRsaniSnavia adrenalini, is Tirkmelzeda jirkvlis hormonia<br />
da rogorc yvela hormoni, adrenalinic organizmSi mudmivad gamomuSavdeba.<br />
Warbi emociebi, dadebiTic da uaryofiTic, iwvevs adre nalinis gaZlierebul<br />
gamoyofas.<br />
uCveulo situaciaSi xarT, guliscema gagixSirdaT, Tvalis gugebi gagifar-<br />
TovdaT, gafiTrdiT an gawiTldiT? _ e. i. Tqvens organizmSi ukve aris adrenalinis<br />
saWiro doza, rac aucileblad mogexmarebaT Sinagani Zalebis mobilizebaSi!<br />
adrenalini
17<br />
karbonmJavebi<br />
alubali, mocxari, tyemali, limoni, for-<br />
Toxali ..., maTi momJavo gemo yvelas gigrZvniaT.<br />
albaT iciT, rom am mJave gemos karbonmJavebi<br />
ganapirobebs.<br />
karbonmJavebi erTmaneTisagan gansxvavdeba<br />
karboqsilis jgufis raodenobiT da naxSir wyalbaduri<br />
radikalis bunebiT.<br />
molekulaSi karboqsilis jgufis Semcvelobis<br />
mixedviT karbonmJava SesaZlebelia iyos:<br />
erTfuZiani. magaliTad,<br />
O<br />
CH3 – C<br />
OH<br />
ZmarmJava<br />
organul nivTierebas, romlis<br />
molekulaSi naxSirwyalbadis<br />
radikalTan erTi<br />
an ramdenime karboqsilis<br />
jgufia dakavSirebuli, karbonmJava<br />
ewodeba.<br />
orfuZiani. magaliTad,<br />
CamoayalibeT najeri da ujeri karbonmJavas ganmarteba. saTanado<br />
magaliTebi Cvens mier SemOoTavazebuli karbonmJavebis formulebidan<br />
SearCieT.<br />
CH 3 ‑ COOH ZmarmJava<br />
O<br />
HO<br />
C – C<br />
C – CH 2 – CH 2 – C<br />
qarvis mJava<br />
CH3 – (CH2) 7 – CH= CH – (CH2) 7 – COOH<br />
oleinmJava<br />
romeli maTganis Sesaxeb SegiZliaT TqvaT dabejiTebiT _ Sedis hidrirebis<br />
reaqciaSi?<br />
axseniT, ratom fiqrobT ase.<br />
O<br />
OH<br />
mJaunmJava<br />
C<br />
benzois mJava.<br />
O<br />
OH<br />
– C O<br />
OH<br />
O<br />
HO<br />
atomTa jgufs kar-<br />
boqsilis jgufi ewodeba.<br />
dagveTanxmebiT, rom naxSirwyalbaduri radikalis bunebis mixedviT karbonmJava<br />
SesaZlebelia iyos: najeri, ujeri, aromatuli.<br />
CH 2=CH – COOH<br />
akrilmJava<br />
aromatuli karbonmJava ki iseTi mJavaa, romlis molekulaSic benzolis<br />
birTvi Sedis. aseTia benzois MmJava<br />
O<br />
OH<br />
101
102<br />
nomenklatura da izomeria<br />
SeamCnevdiT, rom organuli mJavebis klasi sakmaod mravalferovania.<br />
ganvixiloT najeri erTfuZiani karbonmJavebi.<br />
najeri karbonmJavas saxelwodebis Sedgenisas Sesabamisi alkanis saxelwodebis<br />
fuZes emateba sityva `mJava~, naxSirbadatomTa danomvra ki iwyeba<br />
karboqsilis jgufis naxSirbadatomidan. magaliTad,<br />
O<br />
H – C<br />
OH<br />
meTanmJava<br />
CH 3 – CH 2 – CH 2 – C<br />
butanmJava<br />
kidev erTxel aRvniSnavT, rom dRemde gamoiyeneba mJavebis istoriuli, anu<br />
trivialuri saxelwodebebi (ix. cxrili).<br />
cxrili: zogierTi mJavas struqturuli formula da saxelwodeba<br />
saerTaSoriso<br />
saxelwodeba<br />
O<br />
OH<br />
qimiuri formula trivialuri saxelwodeba<br />
meTanmJava HCOOH WianWvelmJava<br />
eTanmJava CH 3COOH ZmarmJava<br />
propanmJava CH 3CH 2COOH propionmJava<br />
butanmJava CH 3CH 2CH 2COOH erbomJava<br />
pentanmJava CH 3CH 2CH 2CH 2COOH valerianmJava<br />
heqsanmJava CH 3 – (CH 2) 4 – COOH kapronmJava<br />
heqsadekanmJava CH 3 (CH 2) 14 – COOH palmitinmJava<br />
oqtadekanmJava CH 3 (CH 2) 16 – COOH stearinmJava<br />
fizikuri Tvisebebi<br />
O<br />
CH3 – C<br />
OH<br />
eTanmJava<br />
O<br />
CH3 – CH2 – CH – C<br />
OH<br />
CH3 2-meTilbutanmJava<br />
SedarebiT mcire molekuluri masis karbonmJavebi<br />
ZmarmJavas msgavsad mkveTri, Taviseburi sunis<br />
mqone siTxeebia. 4-8 atomi naxSirbadis Semcveli<br />
karbonmJavebi arasasiamovno sunis mqone zeTiseburi<br />
siTxeebia.<br />
karbonmJava, romlis molekulaSic naxSirbadatomTa<br />
ricxvi rvas aRemateba, myari, wyalSi uxsnadi<br />
nivTierebaa.<br />
najeri erTatomiani mJavas duRilis temperatura<br />
izrdeba molekulaSi naxSirbadatomis ricxvis<br />
zrdasTan erTad. ase magaliTad, WianWvelm-<br />
miaqcieT yuradReba!<br />
mJavas molekulaSi imdeni nax-<br />
Sir badatomia, ramde nic – Sesabamisi<br />
alkanis molekulaSi.<br />
spirtisa da mJavas duRilis<br />
temperaturebi.
Javas duRilis temperatura 101 0 C-ia,<br />
ZmarmJavasi – 118 0 C, xolo propionmJavasi<br />
– 141 0 C.<br />
sayuradReboa is faqti, rom mcire<br />
molekuluri masis mqone umartivesi<br />
karbonmJava – WianWvelmJavac ki siTxea,<br />
maSin, roca ufro meti molekuluri<br />
masis mqone butani imave pirobebSi airadi<br />
nivTierebaa:<br />
HCOOH C 4H 10<br />
M r=46 M r=58<br />
t duR.=100,8 0 C; t duR.= –0,5 0 C;<br />
wyalbaduri bmis warmoqmnis unari<br />
karbonmJavas wyalSi kargad xsnadobis<br />
mizezicaa – wyalSi gaxsnisas wyalbaduri<br />
bma myardeba karbonmJavasa da wylis<br />
molekulas Soris:<br />
O H<br />
|| |<br />
R – C – O – H ... O<br />
H<br />
kanonzomieria isic, rom naxSirwyalbadur<br />
radikalSi naxSirbadatomTa<br />
ricxvis zrdasTan erTad karbonmJavas<br />
wyalSi xsnadoba mcirdeba.<br />
qimiuri Tvisebebi<br />
karbonmJavebis zogadi qimiuri Tvisebebi araorganuli mJavebis qimiuri<br />
Tvisebebis msgav sia. magaliTad, ZmarmJava wyalSi gaxsnisas disocirdeba wyalbadis<br />
kationad da mJavuri naSTis anionad:<br />
CH3COOH CH3COO –<br />
+ H + .<br />
nivTierebis molekulur masasa<br />
da agregatul mdgomareobas Soris<br />
arsebuli aseTi Seusabamoba ganpirobebulia<br />
imiT, rom karbonmJavas<br />
molekulebs aqvs wyalbaduri<br />
bmebis damyarebis una ri, isini wyvilwyvilad<br />
uerTdeba erTmaneTs da<br />
warmoqmnis dimers.<br />
karbonmJavas molekulebs Soris<br />
wyalbaduri bmis damyarebis unari<br />
ki aixsneba maTi eleqtronuli agebulebiT.<br />
marTlac, karboqsilis<br />
jgufSi eleqtronuli simkvrive isea<br />
gadanawilebuli, rom Jangbadatomi<br />
nawilobriv uaryofiTad, xolo wyalbadatomi<br />
nawilobriv dadebiTad<br />
imuxteba:<br />
karbonmJavebs Soris yvelaze Zlieria WianWvelmJava. is saSualo si<br />
Zlieris mJavaa, danarCeni karbonmJavebi ki sustia. karbonmJavas molekulaSi<br />
naxSirbadatomTa ricxvis zrdasTan erTad disociaciis xarisxi mcirdeba.<br />
Cven ki gTavazobT karbonmJavas monawileobiT mimdinare neitralizaciis<br />
reaqciis zogad tolobas.<br />
δ +<br />
– C<br />
δ –<br />
O<br />
.. δ +<br />
O .. ← H<br />
swored am `boloebiT~ myardeba<br />
wyal baduri bma maT molekulebs<br />
Soris:<br />
R – C<br />
O. . . H – O<br />
O – H . . . O<br />
C – R<br />
gamoikvlieT ZmarmJavas qimiuri Tvisebebi.<br />
ZmarmJava gaanawileT sam sinjaraSi. erTSi CauSviT Mg, meoreSi – MgO,<br />
mesameSi ki – Na 2CO 3 an CaCO 3 (carci).<br />
daakvirdiT reaqciebis mimdinareobas.<br />
SeadgineT reaqciaTa tolobebi. SeadareT ZmarmJavas<br />
qimiuri Tvisebebi araorganuli mJavebis qimiur<br />
Tvisebebs, gamoitaneT daskvna.<br />
ZmarmJavas ma ri -<br />
lebs aceta tebi ewo -<br />
deba. ma ga li Tad,<br />
Mg(CH 3COO) 2 mag ni umis<br />
acetatia.<br />
103
104<br />
1 R – COOH + NaOH → R – COONa + H 2O.<br />
gagacnobT karbonmJavas sxva qimiur Tvisebebsac.<br />
2 karbonmJava reaqciaSi Sedis spirtTan. reaqciis Sedegad ki miiReba esteri<br />
da wyali. amitom, am reaqcias esterifikaciis reaqcia ewodeba.<br />
esterifikaciis reaqcia Seqcevadia da im SemTxvevaSi mimdinareobs bolomde,<br />
Tu sareaqcio ares scildeba warmoqmnili esteri an wyali.<br />
gamoikvlieT esterifikaciis reaqciis mimdinareobis pirobebi:<br />
a) ZmarmJavian sinjaraSi CaasxiT eTilis spirti. amCnevT reaqciis niSnebs?<br />
sinjara SeaTbeT:<br />
amCnevT reaqciis niSnebs?<br />
sinjaraSi mcireodeni gogirdmJava CaasxiT da sinjara kvlav SeaTbeT.<br />
axla Tu amCnevT reaqciis niSnebs?<br />
gamoitaneT daskvna esterifikaciis reaqciis mimdinareobis pirobebis Sesaxeb,<br />
gadaiwereT rveulSi da reaqciis tolobaSi SeitaneT reaqciis mimdinareobis<br />
pirobebis Sesabamisi aRniSvnebi:<br />
O O<br />
|| ||<br />
R – C – OH + R′ – OH E R – C – OR′ +H2O .<br />
karbonmJava spirti esteri<br />
3 karbonmJavebisaTvis damaxasiaTebelia zogierTi specifikuri Tvisebac,<br />
rac ganpirobebulia molekulaSi naxSirwyalbaduri radikalis arsebobiT.<br />
magaliTad, karbonmJava reaqciaSi Sedis qlorTan:<br />
H<br />
Cl – Cl + H – C – C<br />
O<br />
→ Cl – C – C<br />
H<br />
O – H<br />
O<br />
H<br />
+HCl .<br />
H<br />
O – H<br />
ZmarmJava qlorZmarmJava<br />
sagangebod aRvniSnavT WianWvelmJavas Tvisebebs – mo lekulaSi karboqsilis<br />
jgufTan uSualod dakavSirebuli wyalbadis gamo, is aldehidic aris<br />
da mJavac, amitom advilad iJangeba vercxlis oqsidis amiakiani xsnariT da<br />
warmoqmnis naxSirmJavas, romelic iSleba naxSirorJangad da wylad:<br />
H – C O<br />
OH<br />
anu<br />
HO – C O<br />
+ Ag2O H<br />
WianWvelmJava<br />
H – C O<br />
OH<br />
t<br />
t<br />
HO – C O<br />
+ 2Ag↓<br />
OH<br />
H2O + CO2↑ naxSirmJava<br />
koncentrirebul gogirdmJavasTan ga cxelebisas<br />
Wian WvelmJava wylis mo lekulas moixleCs<br />
(Si da mo lekuluri dehidratacia) da<br />
naxSirbad(II)-is oqsids gamoyofs.<br />
es reaqcia SeiZ le ba gamoviyenoT la bo ra toriaSi<br />
nax Sir bad(II)-is oqsidis misaRebad:<br />
H 2O + CO.
miReba<br />
laboratoriaSi karbonmJavas miReba SesaZlebelia araorganuli mJavebis<br />
msgav sad, mis marilze gogirdmJavas moqmedebiT. magaliTad,<br />
2CH3 – C O<br />
O – Na + H2SO4 Na2SO4 + 2CH3 – C O<br />
t<br />
O – H<br />
natriumis acetati ZmarmJava<br />
mrewvelobaSi ki karbonmJavas misaRebad aldehidis daJangvis reaqcias<br />
mimarTaven:<br />
R – CHO +[O] → R – COOH.<br />
aldehidi karbonmJava<br />
vinaidan aldehidi Tavis mxriv spirtis daJangviT miiReba, amitom mJavas<br />
miReba SesaZlebelia uSualod spirtis daJangviT. Sualeduri produqtis,<br />
aldehidis gamoyofis gareSe. am procesis gamosaxva SesaZlebelia Semdegi Semajamebeli<br />
tolobiT:<br />
R – CH 2OH + 2[O] → RCOOH + H 2O.<br />
spirti karbonmJava<br />
najeri erTfuZiani karbonmJavebidan<br />
gansakuTrebuli praqtikuli mniSvneloba<br />
aqvs WianWvelmJavas, ZmarmJavas, agreTve<br />
palmitinis, stearinisa da oleinis mJavebs.<br />
amitom, ufro dawvrilebiT maT Sesaxeb visaubrebT.<br />
WianWvelmJava HCOOH mZafri sunis mqone<br />
siTxea, duRs 100,8 0 C-ze. wyalSi kargad<br />
ixsneba, kanze moxvedrisas iwvevs damwvrobas.<br />
mas aqvs madezinficirebeli Tvisebebi,<br />
ris gamoc gamoiyeneba farmacevtul da<br />
tyavis warmoebaSi, agreTve medicinaSi. is<br />
gamoiyeneba agreTve qsovilisa da qaRaldis<br />
Rebvis procesSi.<br />
sur.34. WianWvelmJava Sedis Wian-<br />
Wvelis organizmSi, WinWarSi da<br />
naZvis wiwvebSi. WinWari imitom<br />
susxavs, rom WianWvelmJava aRizianebs<br />
kans. WianWvel mJava sawamlavia.<br />
ZmarmJava CH3COOH ufero, mZafri sunis mqone siTxea, wyalSi ixsneba nebismieri<br />
raodenobiT. ZmarmJavas 3-5%-iani xsnari farTod gamoiyeneba kvebis mrewvelobaSi<br />
da maRaziaSi iyideba komerciuli saxelwodebiT `Zmari~. ZmarmJavaSi<br />
kargad ixsneba mravali organuli nivTiereba da amitom gamoiyeneba saRebavebisa<br />
da laqebis mosacileblad.<br />
ZmarmJava xmardeba agreTve teqnikurad mniSvnelovani nivTierebebis, magaliTad,<br />
herbicidebis miRebas.<br />
grZeljaWviani najeri erTfuZiani karbonmJavebia – palmitinisa da stearinis<br />
mJavebi.<br />
moklejaWviani najeri, erTfuZiani karbonmJavebisagan<br />
gansxvavebiT myari, wyalSi uxsnadi mJavebia. Tumca, C15H31COOH maTi natriumisa da kaliumis marilebi – stearatebi palmitinis MmJava<br />
da palmitatebi wyalSi kargad ixsneba da gasarecxad<br />
C17H35COOH gamoiyeneba, maTi komerciuli saxelwodebaa `saponi~.<br />
stearinis mJava<br />
105
106<br />
grZeljaWviani ujeri karbonmJavebidan gansakuTrebiT<br />
aRsaniSnavia oleinmJava C 17H 33COOH. is<br />
zeTiseburi, usuno nivTierebaa.<br />
saponi<br />
saponi sxva araferia, Tu ara umaRlesi karbonmJavas<br />
marili. umaRlesi karbonmJavebidan sapnis<br />
misaRebad ZiriTadad gamoiyeneba: palmitinis<br />
sur. 35. sapnebi<br />
mJava C15H31COOH, sterinis mJava C17H35COOH, oleinis mJava C17H33COOH. am mJavebis mier warmoqmnili natriumis marilebi myari sapnebia, kaliumis<br />
marilebi ki _ Txevadi. am marilebis hidrolizisas warmoiqmneba ori niv-<br />
Tiereba, tute da cximovani mJava. tute Slis damaWuWyianebel nivTierebebs,<br />
cximovani mJava zedapirulad-aqtiuri nivTierebaa, romelic xels uwyobs<br />
damaWuWyianebeli nivTierebebis mocilebas gaWuWyianebuli zedapiridan.<br />
1. SeadgineT erbomJavas homologebisa da izomerebis or-ori formula,<br />
daasaxeleT isini.<br />
2. saxelwodebis mixedviT SeadgineT nivTierebis struqturuli formula:<br />
a) meTilacetati; b) mJaunmJava;<br />
g) WianWvelmJava; d) diqlorZmarmJava;<br />
e) magniumis acetati; v) eTilacetati;<br />
z) eTilformiati; T) akrilmJava.<br />
3. daasruleT reaqciaTa tolobebi:<br />
a) C17H35COOH+ NaOH →<br />
b) C17H35COOH +Na2CO3 →<br />
g) C17H33COOH +H2 →<br />
4. sqemis Sesabamisad SeadgineT reaqciaTa tolobebi:<br />
eTani → eTileni → bromeTani → eTanoli → Zmaraldehidi → ZmarmJava.<br />
5. Sedgenilobisa da agebulebis safuZvelze ivaraudeT akrilmJavas<br />
H2C=CH_COOH qimiuri Tvisebebi.<br />
6. SeadgineT araorganuli nivTierebidan, magaliTad CO2-dan an CaC2-dan ZmarmJavas miRebis sqema. Semdeg ki dawereT sqemis Sesabamisi reaqciaTa tolobebi.<br />
7. SeadgineT reaqciis sami toloba, romelTagan erTi – daasabuTebs Wian-<br />
WvelmJavas msgavsebas ZmarmJavasTan, meore – daasabuTebs gansxvavebas WianWvelm-<br />
Javasa da ZmarmJavas Soris, mesame – daasabuTebs WianWvelmJavas da ZmarmJavas<br />
msgavsebas araorganul mJavebTan.<br />
8. gamoiangariSeT airis moculoba (n.p.) romelic SesaZloa gamoiyos 4,6 g<br />
metaluri natriumis urTierTqmedebiT 0,1 mol meTanolTan.
18<br />
Cais gamxmari foTlebis nayeni albaT araerTxel mogimzadebiaT.<br />
Tumca, SesaZloa arc gifiqriaT, rom misi surnelis da nayenis zedapirze<br />
cximovani wveTebis warmoqmnis erT-erTi mizezi esteria.<br />
esteris zogadi formulaa:<br />
nomenklatura<br />
fizikuri Tvisebebi<br />
R – C O<br />
O – R<br />
esteris saxelwodeba Sedgeba Sesabamisi spirtisa da mJavas saxelwodebisagan.<br />
magaliTad,<br />
O<br />
H – C<br />
O – CH3 meTilmeTanati, an<br />
meTilformiati<br />
O<br />
CH3 – C<br />
O – C2H5 eTileTanati, an<br />
eTilacetati<br />
ratom afrqvevs saocar surnels gazafxulze ayvavebuli xexilis baRi, an<br />
ratom igrZnoba larnakSi moTavsebuli sul ramdenime mindvris yvavilis suni<br />
ase mZafrad?<br />
ase imitom xdeba, rom moklejaWviani erTfuZiani mJavasa da moklejaWviani<br />
spirtis esterebi wyal ze msubuqi, aqroladi, sasiamovno sunis mqone siTxeebia.<br />
aseAT esterebs Seicavs yvavilebi, xili da nayofi, ris gamoc maT sasiamovno,<br />
specifikuri suni aqvT. grZeljaWviani erTfuZiani mJavasa da grZeljaWviani<br />
spirtis esteria magaliTad, futkris cvili. is ZiriTadad Sedgeba palmitinis<br />
mJavasa da miricilis spirtisagan C 15H 31COOC 30H 61. veSapis qoni, anu spermaceti<br />
ki – imave palmitinmJavasa da cetilis spirtis esteria C 15H 31COOC 16H 33.<br />
O<br />
||<br />
C5H11 – O – C – C3H7 esterebi, cximebi<br />
O<br />
||<br />
CH3 – O – C – C3H7 sur. 36. esterebis gavrceleba bunebaSi<br />
O<br />
||<br />
C 2H 5 – O – C – C 3H 7<br />
SeadgineT msxlis, vaSlisa da ananasis SemadgenlobaSi Semavali esterebis<br />
saxelwodeba.<br />
107
108<br />
qimiuri Tvisebebi<br />
rogorc araerTxel aRvniSneT esteri urTierTqmedebs wyalTan – mimdinareobs<br />
esterifikaciis sapirispiro reaqcia, ris Sedegadac miiReba mJava da<br />
spirti:<br />
O<br />
CH3 – C<br />
+ HOH E CH3 – C .<br />
O – C2H5 O<br />
+ C2H5 – OH<br />
O – H<br />
miReba<br />
spirtsa da mJavas Soris wylis moxleCiT mimdinare qimiuri reaqciis Sedegad<br />
miiReba organuli nivTiereba – esteri:<br />
R – C O<br />
+HOR R – C<br />
OH<br />
O<br />
E<br />
+ H2O .<br />
O – R<br />
rogorc ukve iciT, es reaqcia Seqcevadia, wonasworobis gadanacvleba<br />
esteris warmoqmnis mxares SesaZlebelia wyalwamrTmevi saSualebebis gamoyenebiT.<br />
esteri gamoiyeneba, rogorc danamati kvebis mrewvelobaSi. meTilakrilati<br />
CH2=CH – COOCH3 da meTilmetakrilati farTod gamoiyeneba<br />
organuli minis warmoebaSi.<br />
esteris klass ganekuTvneba cxoveluri da mcenareuli cximi. isini mniSvnelovnad<br />
gansxvavdeba sxva esterisagan zogierTi TvisebiT, biologiuri<br />
roliTa da mniSvnelobiT, amitom maT calke ganvixilavT.<br />
cximi<br />
cximis Sedgeniloba da agebuleba SeiZleba gamoisaxos zogadi formuliT:<br />
CH 2 – O – C<br />
CH – O – C<br />
O<br />
O – R 1<br />
O<br />
O – R2 cximi grZeljaWviani<br />
karbonmJavas da<br />
glicerinis esteria.<br />
O<br />
CH2 – O – C<br />
O – R3 cximebis SedgenilobaSi ZiriTadad Sedis grZeljaWviani karbonmJavebis<br />
– oleinis, palmitinisa da stearinis mJavaTa radikalebi. maSasadame, cximis<br />
SedgenilobaSi SesaZlebelia Sediodes, rogorc ujeri (oleinmJava), ise najeri<br />
mJavas (palmitinisa da stearinis) naSTi.
Tu esteris SedgenilobaSi<br />
didi raodenobiT Sedis ujeri<br />
mJavas radikali, is Txevadia.<br />
aseTi cximi umetesad mcenareuli<br />
warmoSobisaa da mas<br />
zeTi ewodeba. magaliTad mzesumziras,<br />
soios, zeiTunis da<br />
sxva. iSviaTad, magram, mainc<br />
arsebobs myari mcenareuli<br />
cximi, magaliTad – qoqosisa da<br />
kakaos zeTi.<br />
esterebis msgavsad yvela cximi hidrolizdeba:<br />
R<br />
OH<br />
OH<br />
1<br />
HC – C O<br />
R2 H2C – C<br />
O<br />
R3 + 3HOH H C – OH<br />
R<br />
H2C – OH<br />
2 – C O<br />
R3 – C O<br />
H2C – C<br />
+<br />
glicerini<br />
OH<br />
O<br />
H2C – OH<br />
R1 – C O<br />
karbonmJava<br />
cximebi, romlebic warmoqmnilia um-<br />
Tavresad najeri mJavebisagan, myaria.<br />
ujeri mJavebis Semcvelobis zrdasTan<br />
erTad cximis lRobis temperatura<br />
mcirdeba. cxoveluri<br />
cximis umravle soba<br />
ZiriTadad najer mJavebs<br />
Seicavs. Cveulebriv pirobebSi<br />
isini myaria da<br />
advil lRobadi. magali-<br />
Tad, cxvris an Roris<br />
qoni. Tumca,<br />
gamonaklisi aqac<br />
arsebobs – Tevzis<br />
qoni Cveulebriv<br />
pirobebSi Txevadia.<br />
cxadia, es reaqciac Seqcevadia. wonasworobam rom reaqciis produqtebis<br />
mxares gadainacvlos, mas atareben tute areSi, risTvisac umateben tute<br />
metalis karbonats, magaliTad, sodas (Na 2CO 3) an tutes. aseT pirobebSi<br />
hidrolizi Seuqcevadad mimdinareobs da reaqciis Sedegad miiReba ara karbonmJava,<br />
aramed misi marili.<br />
cximis molekulis Sedgeniloba ganapirobebs mis fizikur da qimiur Tvisebebs.<br />
amitom, ujeri mJavas naSTis Semcvel esters ujeri naerTis Tvisebebi<br />
aqvs – auferulebs bromian wyals, Sedis mierTebis reaqciaSi. Tumca, Cven<br />
yuradRebas gavamaxvilebT didi praqtikuli mniSvnelobis mqone hidrirebis<br />
reaqciaze, radgan am reaqciiT Txevadi cximisagan miiReba myari esteri.<br />
manamde ki mokled mimovixiloT cximi, rogorc sakvebi produqti.<br />
cximi yvelaze ufro kaloriuli sakvebi produqtia. organizmSi moxvedrisas,<br />
fermentebis gavleniT, cximi hidrolizdeba glicerinad da karbonmJavad.<br />
am nivTierebebisagan SemdegSi warmoiqmneba sxva iseTi cximi, romelic<br />
cocxali organizmisaTvis ufro advili SesaTvisebelia.<br />
gemoze rom araferi vTqvaT, organizmisaTvis ufro margebeli ujeri<br />
cximebia, magram molekulaSi ramdenime ujeri bmis arsebobis gamo isini reaqciisunarianebi<br />
arian, iJangebian haeris JangbadiT, ris gamoc maTi didxans<br />
Senaxva SeuZlebelia.<br />
109
110<br />
am naklis aRmofxvra SesaZlebelia cximebis hidrirebiT. am gziT miiReba<br />
garemo pirobebisadmi ufro mdgradi myari cximi, romelsac iyenebenDA<br />
margarinis<br />
warmoebaSi:<br />
CH 2 – O – C<br />
CH – O – C<br />
O<br />
C 17H 33<br />
O<br />
CH2 – O – C O<br />
zeTi<br />
C 17H 33<br />
C 17H 33<br />
+ 3H 2<br />
Cu, Ni<br />
t<br />
CH 2 – O – C<br />
O<br />
C 17H 35<br />
O<br />
CH – O – C<br />
C17H35 CH2 – O – C O<br />
margarini<br />
C17H35 margarinis misaRebad cximis hidrireba mimdinareobs 150-160 0 C-ze, spilenZnikelis<br />
katalizatoris TanxlebiT, 3-4 saaTis ganmavlobaSi.<br />
1. SeadgineT iseTi esteris formula, romelSic naxSirbadatomTa minimaluri<br />
ricxvia.<br />
– daadgineT masSi TiToeuli elementis masuri wili.<br />
– gansazRvreT, ra nivTierebisaganaa SesaZlebeli aseTi esteris miReba.<br />
– SeadgineT Sesabamisi reaqciis toloba.<br />
<br />
2. daasruleT reaqciaTa tolobebi:<br />
H3C – C O<br />
a)<br />
OH<br />
+ HOC2H5 →<br />
H3C – C O<br />
b)<br />
OH<br />
+ HOCH2 – CH – CH3 →<br />
3. ratom wvavs Tvalebs saponi?<br />
CH 3<br />
hidrirebuli<br />
cximi margarini<br />
4. rogor ganasxvavebT or, vardisfrad SemRvreul xsnars, Tu iciT, rom erTerTSi<br />
fenolia, meoreSi ki – saponi?
5. rodesac Tqveni mSoblebi skolaSi swavlobdnen...<br />
80-90-ian wlebSi saqarTveloSi moqmed qimiis saxelmZRvaneloSi amoikiTxavT:<br />
`cximebi rTuli eTerebis klass ekuTvnian. amitom maTi yvelaze damaxasiaTebeli<br />
reaqcia hidrolizia.<br />
cximebis hidrolizis reaqcia, iseve rogorc sxva rTuli eTerebisa,<br />
Seqcevadia, gamovsaxoT es gamartivebuli gantolebiT:<br />
O<br />
||<br />
CH 2 ― O ― C ―R HO ― H CH 2 ― OH<br />
O O<br />
||<br />
CH ― O ― C ― R + HO ― H D CH ― OH + 3R ― C<br />
O<br />
||<br />
CH 2 ― O ― C ―R HO ― H CH 2 ― OH<br />
ra pirobebi iqneba saWiro imisTvis, rom wonasworeobam glicerinisa<br />
da karnbonmJavas warmoqmnis mxares gadainacvlos?”<br />
– upasuxeT cximebis hidrirebis reaqciis Sesaxeb dasmul SekiTxvas. axseniT<br />
Tqveni pasuxi.<br />
– kidev ra SegiZliaT TqveT cximebis qimiuri Tvisebebis Sesaxeb?<br />
OH<br />
111
112<br />
SeamowmeT Tqveni codna<br />
1 kalciumis propionats bunebrivad Seicavs zogierTi saxeobis yveli,<br />
zogierTs ki umateben specialurad, kvebiTi Rirebulebis gasaumjobeseblad:<br />
atomia:<br />
a) saxelwodebis mixedviT SeadgineT am naerTis formula;<br />
b) aRwereT, ra gziTaa SesaZlebeli misi miReba laboratoriaSi.<br />
SeadgineT reaqciis toloba;<br />
g) ivaraudeT da reaqciis tolobiT asaxeT misi Tvisebebi.<br />
2 Txis rZe erTfuZian karbonmJavas Seicavs, romelSic eqvsi naxSir bad-<br />
– SeadgineT am naerTis struqturuli formula;<br />
– ra hqvia am naerTs saerTaSoriso nomenklaturis mixedviT?<br />
– romeli ukeT gaixsneba wyalSi – Txis rZe, Tu Zroxis rZe, romelic Seicavs<br />
rZemJavas?<br />
CH 3CHCOOH<br />
|<br />
OH<br />
– ra msgavsi da gansxvavebuli Tvisebebi aqvs am nivTierebebs?<br />
– CamoayalibeT kriteriumebi, romlebic Sefasebisas gamoiyeneT.
4<br />
suraTze Tqven xedavT qolesterinis struqturul formulas. naerTTa<br />
romel klass miakuTvnebdiT mas? ivaraudeT, unda ixsnebodes Tu ara wyalSi<br />
es naerTi? daasabuTeT Tqveni varaudi. Cven ki dasaxmareblad gTavazobT<br />
spirtebis wyalSi xsnadobis cxrils.<br />
HO<br />
HO<br />
4 sqemaze aRwerilia fenolidan samkurnalo preparatebis, aspirinisa da<br />
dispirinis miReba:<br />
H H<br />
CH 3<br />
CH 3<br />
CH 3<br />
CH 3<br />
spirti wyalSi xsnadoba<br />
250C-ze g/100 g<br />
wyalSi<br />
meTanoli srulad ixsneba<br />
eTanoli srulad ixsneba<br />
1 – propanoli srulad ixsneba<br />
1 – butanoli 9<br />
cikloheqsani 5,6<br />
1-heqsanoli 0,6<br />
a) romeli xerxia ufro ekonomiuri?<br />
b) dispirini ukeT gaixsneba wyalSi Tu aspirini? (varaudis eqsperimentiT<br />
Semowmeba Zneli ar aris. orive preparati afTiaqSi iyideba).<br />
g) erT-erTi maTgani ufro efeqturi tkivilgamayuCebeli saSualebaa,<br />
SesaZlebelia, Tu ara es faqti maT wyalSi xsnadobas ukavSirdebodes? axseniT<br />
amis mizezi.<br />
113
114<br />
jer kidev XIX saukuneSi mecnierebma aRmoaCines, rom arsebobs daavadeba,<br />
romelic azianebs nervul sistemas da romelic gamowveulia sakvebSi romeliRac<br />
gansakuTrebuli nivTierebis ukmarisobiT. 1912 wels polonelma mecnierma<br />
kazimeJ funkma brinjisagan gamoyo es gansakuTrebuli nivTiereba da<br />
mas vitamini uwoda. vitamini warmodgeba laTinuri sityvisagan `vita~ da qar-<br />
Tulad niSnavs `sicocxles~.<br />
dRes es sityva yvelam icis. Tumca ra funqcia akisria vitaminebs organizmSi,<br />
saidan miewodeba vitaminebi organizms, ra saxis vitaminebia dReisaTvis<br />
cnobili _ amis Sesaxeb adamianebma naklebad ician.<br />
proeqti<br />
Temis<br />
aqtualoba<br />
vitaminis roli adamianis organizmisaTvis<br />
gansakuTrebiT mniSvnelovani C da B 1 vitaminebi<br />
sakvebi da vitaminebi<br />
kvlevis sagani:<br />
hipoTeza<br />
kvlevis meTodebi:<br />
`vitaminebi~<br />
zogierTi vitaminis funqcia adamianis organizmSi<br />
adamianis dRiuri norma vitaminebTan mimarTebaSi<br />
wyalSi xsnadi da cximSi xsnadi vitaminebi<br />
vitaminebis Semcveloba sakveb produqtebSi<br />
kvlevis<br />
qimiuri<br />
nawili<br />
zogierTi vitaminis qimiuri Sedgeniloba, qimiuri formula
eqsperimentuli<br />
nawili:<br />
A vitaminis aRmoCena mzesumziris zeTSi (Sesa dareblad<br />
cda CaatareT zeiTunis zeTzec).<br />
sinjaraSi moaTavseT 1 ml mzesumziris zeTi da davamatoT<br />
2-3 ml FeCl 3-is 1%-iani xsnari.<br />
A vitaminis arsebobis SemTxvevaSi warmoiqmneba _ kaSkaSa mwvane Seferiloba.<br />
C vitaminis aRmoCena sam sxvadasxva jiSis vaSlSi. SearCieT sami sxva dasxva<br />
jiSis vaSli. TiToeul SemTxvevaSi eqsperimenti CaatareT Semdegnairad.<br />
sinjaraSi CaasxiT 2 ml vaSlis wveni da daamateT 10 ml wyali. Semdeg daamateT<br />
saxameblis bubko (1 g saxamebeli 100 g mduRare wyalze), wveT-wveTad<br />
daamateT iodis 5%-iani xsnari mdgradi lurji Seferilobis warmoqmnamde,<br />
romelic ar qreba 10-15 wT-is ganmavlobaSi.<br />
es teqnika dafuZnebulia im garemoebaze, rom askorbinis mJavas molekulebi<br />
advilad iJangeba iodiT. rogorc ki iodi mTlianad daJangavs askorbinis<br />
mJavas, momdevno wveTi urTierTqmedebs saxamebelTan da xsnari lurjad<br />
Seifereba. wveTebis raodenobis mixedviT SesaZlebelia miaxloebiT davadginoT<br />
sxvadasxva jiSis vaSlSi C vitaminis raodenoba.<br />
C vitaminis SedarebiT raodenobaze (Zalian miaxloebiT) SeiZleba vimsjeloT<br />
iodis xsnaris wveTebis raodenobis mixedviT, romelic askorbinis<br />
mJavas molekulebis daJangvaze ixarjeba.<br />
eqsperimentis Sedegebi:<br />
A da C vitaminis aRmoCena sakveb produqtebSi.<br />
SerCeulia iseTi<br />
produqtebi, rom<br />
fe ris cvlileba mkafiod<br />
gamoCndes.<br />
115
116<br />
am TavSi ganvixilavT<br />
naxSirwylebs _ monosaqaridebs,<br />
disaqaridebs da polisaqaridebs.<br />
aminebsa da aminomJavebs.<br />
cilebs.<br />
bunebriv da sinTezur polimerebs.<br />
V T a v i<br />
naxSirwylebi, azotSemcveli<br />
organuli naerTebi<br />
polimerebi<br />
am Tavis Seswavlis Semdeg<br />
SeZlebT qimiuri formulis mixedviT erTmaneTisagan<br />
ganasxvavoT mono-, di- da polisaqaridebi.<br />
imsjeloT mono-, di- da polisaqaridebis fizikur da qimiur<br />
Tvisebebze, daakavSiroT es Tvisebebi maT SedgenilobaSi<br />
arsebul funqciur jgufebTan.<br />
SeZlebT qimiuri formulis mixedviT naerTTa Sesabamis<br />
klass miakuTvnoT azotSemcveli organuli naerTi.<br />
SeZlebT qimiuri reaqciebiT asaxoT aminebisa da<br />
aminomJavebis Tvisebebi da miRebis xerxebi.<br />
SeZlebT imsjeloT cilis agebulebaze, Tvisebebsa da<br />
mniSvnelobaze.<br />
SeZlebT ganasxvavoT bunebrivi da sinTezuri polimerebi.
19<br />
naxSirwylebs Soris namdvilad aris tkbili niv-<br />
Tierebebi, magram Turme, rac tkbilia yvelaferi<br />
naxSirwyali ar aris. magaliTad, eqvsatomiani spirti-sorbiti.<br />
yvela naxSirwyalic ar aris tkbili _<br />
magaliTad saxamebeli.<br />
udavo arc naxSirwylis zogadi formulaa<br />
C n(H 2O) m. es formula qimiaSi damkvidrda bunebaSi far-<br />
Tod gavrcelebuli naxSirwylebis analizis Sedegad.<br />
aseTia magaliTad, glukoza da fruqtoza. mogvianebiT<br />
gairkva, rom zogierTi naxSirwylis Sedgeniloba ar<br />
pasuxobs am formulas, magaliTad, dezoqsiroboza<br />
C 6H 10O 4. amis miuxedavad istoriuli saxelwodeba `naxSirwyali~ dRemde SemorCa<br />
qimias.<br />
naxSirwylebs hidrolizis unari aqvs. am unaris mixedviT naxSirwylebi<br />
iyofa sam ZiriTad jgufad: mono, di da polisaqaridebad.<br />
axla ufro dawvrilebiT gavecnoT TiToeuli jgufis mniSvnelovan warmomadgenlebs.<br />
monosaqaridi ar<br />
iSleba wyalSi, e. i.<br />
ar hidrolizdeba.<br />
monosaqaridia magaliTad,<br />
glukoza<br />
da fruqtoza.<br />
monosaqaridi – glukoza, fruqtoza<br />
fizikuri Tvisebebi<br />
glukoza da fruqtoza ufero, myari kristaluri<br />
nivTierebebia. orive kargad ixsneba wyalSi.<br />
glukoza mniSvnelovani sakvebi produqtia. organizmSi<br />
moxvedrisas is nawlavebidan sisxlSi Seiwoveba<br />
da misi saSualebiT miewodeba organizms da qsovilebs,<br />
sadac mimdinareobs glikogenis, cximebisa da aminom-<br />
Javebis sinTezi.<br />
glukoza Sedis<br />
yur Znis wvenSi, amitom<br />
daarqves mas<br />
yurZnis Saqari.<br />
naxSirwylebi<br />
disaqaridi hidrolizdebamonosaqaridis<br />
ori molekulis<br />
warmoqmniT. disaqaridia<br />
magaliTad, saqaroza<br />
da maltoza.<br />
fruqtoza Sedis<br />
xilSi, amitom<br />
daarqves mas xilis<br />
Saqari.<br />
naxSirwyali – iseTi<br />
or ganuli nivTierebaa,<br />
romlis molekula-<br />
Sic Sedis naxSirbadis,<br />
wyal badisa da Jangbadis<br />
atomebi. amave dros<br />
wyalbadisa da Jangbadis<br />
atomTa Tanafardoba<br />
iseTivea, rogorc<br />
wylis molekulaSi 2:1.<br />
polisaqaridi iseTi<br />
naxSirwyalia, romelic<br />
hidrolizdeba monosaqaridis<br />
mravali<br />
molekulis warmoqmniT<br />
– ZiriTadad, es glukozis<br />
molekulebia.<br />
glukozis Sem cveloba<br />
adamianisa da<br />
cxovelis sisxlSi<br />
0,1%-is far glebSia.<br />
Tu 100 ml sis xlSi<br />
glu kozis Semcvelobam<br />
180 mg-s gadaaWarba,<br />
niv TierebaTa cvla<br />
irRveva da viTardeba<br />
daavadeba – Saqriani<br />
diabeti.<br />
glukoza cocxali<br />
organizmis ujredis<br />
energiis wyaroa,<br />
amitom is farTod<br />
gamoiyeneba medicina-<br />
Si sxvadasxva daavadebebis,<br />
gansakuTrebiT<br />
ki saerTo sisustis<br />
samkurnalod.<br />
117
118<br />
qsovilSi moxvedrisas glukoza nela iJangeba da gardaiqmneba wylad da<br />
naxSirorJangad. am procesSi gamoiyofa energia:<br />
C 6H 12O 6 + 6O 2 → 6CO 2 + 6H 2O + @920 kj.<br />
es energia mniSvnelovnad uzrunvelyofs cocxali organizmebis energetikul<br />
moTxovnebs. am energiis xarjze organizmi asrulebs sxvadasxva funqcias<br />
_ nivTierebaTa sinTezi, kunTebis muSaoba da sxva.<br />
glukozis molekulis agebuleba<br />
glukoza erTdroulad mravalatomiani spirtic aris da aldehidic. amitom<br />
amboben, rom – is aldehidspirtia.<br />
glukozis molekuluri formulaa C 6H 12O 6.<br />
SeecadeT glukozis struqturuli formulis Sedgenas. gaiTvaliswineT,<br />
rom naxSirbadis eqvsi atomidan erTi Sedis aldehidis jgufSi, darCenil<br />
xuT atomTan ki TiTo spirtuli jgufia SeerTebuli.<br />
Sekvecili struqturuli formula ki iqneba:<br />
O<br />
CH2OH – (CHOH) 4 – C<br />
H<br />
glukozis izomeri fruqtoza<br />
ke ton spirtia:<br />
cdis Sedegad dadgenilia, rom kris ta luri<br />
glukoza Sedgeba cik luri agebulebis molekulebisagan.<br />
cikli ikvreba C‑5-is hidroqsilis<br />
wyal badis gadasvliT aldehidis jgufis<br />
JangbadatomTan.<br />
CH 2OH – (CHOH) 3 – C – CH 2OH<br />
||<br />
O<br />
glukozis struqturul formulaSi spirtuli hidroqsilebi ganlagebulia<br />
ZiriTadi jaWvis marjvniv, an marcxniv.<br />
Tu glukozis molekulaSi, OH jgufi me-5 naxSirbadatomis marjvnivaa, maSin<br />
glukoza D-rigs miekuTvneba. Tu marcxnivaa – L-rigs. bunebaSi gavrcelebuli<br />
naxSirwylebi, rogorc wesi, D-rigis izomerebia. L-rigis izomerebi ki bunebaSi<br />
ar arsebobs. C-2, C-3 da C-4 naxSirbadatomTan OH-is ganla-<br />
fruqtoza proeqciuli<br />
for muliT ase gamo isaxeba:<br />
CH 2OH<br />
|<br />
C = O<br />
|<br />
HO – C – H<br />
|<br />
H – C – OH<br />
|<br />
H – C – OH<br />
|<br />
CH 2OH<br />
D-fruqtoza.<br />
gebis mixedviT arsebobs D-rigis<br />
monosaqaridebis sxvadasxva izomeri.<br />
D-glukozis struqturul<br />
formulaSi C‑3 Tan hidroqsili<br />
ganlagebulia marcxniv, C‑2 da<br />
C‑4-Tan ki – marjvniv.<br />
iseT struqturul formulas,<br />
sadac C‑2, C‑3, C‑4 da C‑5 naxSirbadatomebTan<br />
hidroqsilis<br />
orientacia cnobilia, proeqciuli<br />
formula ewodeba.<br />
CHO<br />
2|<br />
H – C – OH<br />
3|<br />
HO – C – H<br />
4|<br />
H – C – OH<br />
5|<br />
H – C – OH<br />
6|<br />
CH 2OH<br />
D-glukoza.
am faqtis asaxsnelad gavixsenoT is, rom naxSirbadatomebs SeuZlia Tavisufali<br />
brunva δ-bmis irgvliv. brunvis procesSi aldehidis jgufi maqsimalurad<br />
uaxlovdeba mexuTe naxSirbadatomis spirtul jgufs.<br />
aseT geometriul formaSi mexuTe naxSirbadis (C‑5) hidroqsilis wyalbadi<br />
gadadis karbonilis JangbadTan, xolo hidroqsilis Jangbadatomi ukav Sirdeba<br />
naxSirbadis pirvel atoms da warmoq mnis cikls, romelsac eqvskuTxedis forma<br />
aqvs.<br />
6CH2OH<br />
6CH<br />
2OH<br />
5 C OH<br />
5C<br />
O<br />
H<br />
C 4<br />
H<br />
1<br />
C<br />
O<br />
H<br />
C 4<br />
H<br />
OH<br />
1<br />
C<br />
HO OH<br />
3<br />
C<br />
H<br />
2<br />
C<br />
H HO<br />
OH<br />
3<br />
C<br />
H<br />
2<br />
C<br />
H<br />
H OH<br />
H OH<br />
glukozis ciklis SedgenilobaSi<br />
Sedis erTi Jangbadisa da xuTi<br />
naxSirbadis atomi, romlebic<br />
cikluri formula gviCvenebs ara mxolod<br />
atomTa SeerTebis Tanmimdevrobas,<br />
aramed maT urTierTganlagebasac sivrcesavalento<br />
xazebis gadakveTaSia. Si.Seam CnevdiT, rom glukozis molekulis<br />
ciklSi Semavali TiToeuli nax- pirveli da mexuTe naxSirbadatomis urT-<br />
Sirbadatomi or valentobas xarierTqmedebis Sedegad, pirvel naxSirbadjavs<br />
mezobel naxSirbadatomebTan atomTan warmoiqmna axali hid roqsilis<br />
dasakavSireblad. danarCeni ori jgufi. am hidroqsils SeuZlia daikavos<br />
valentoba perpendikularuli<br />
xazebiT aris gamosaxuli da nax-<br />
Sirbadatomebs OH-Tan da OH-Tan<br />
aerTebs.<br />
orgvari mdebareoba – erTi, rodesac<br />
pirveli da meore naxSirbadatomis hidroqsili<br />
ciklis sibrtyis erT mxaresaa da<br />
meore – rodesac pirveli da meore naxSir-<br />
CHO<br />
H – C – OH<br />
HO – C – H<br />
H – C – OH<br />
CH2OH H O OH<br />
H<br />
OH H<br />
HO H<br />
badatomis hidroqsili ciklis sibrtyis<br />
sxvadasxva mxaresaa.<br />
Sesabamisad, glukozas cikluri α da<br />
β forma aqvs:<br />
H – C – OH<br />
H<br />
OH<br />
6<br />
CH2OH 6<br />
CH2OH CH2OH D-glukozis<br />
wrfivi forma.<br />
D-glukozis<br />
cikluri forma.<br />
5 C<br />
H<br />
H<br />
C 4<br />
OH<br />
HO 3<br />
C<br />
O<br />
H<br />
2<br />
C<br />
H<br />
1C<br />
OH<br />
5 C<br />
H<br />
H<br />
C 4<br />
OH<br />
HO 3<br />
C<br />
O<br />
H<br />
2<br />
C<br />
OH<br />
1C<br />
H<br />
H<br />
OH<br />
α‑D-glukoza<br />
glukozis α-forma.<br />
H<br />
OH<br />
β‑D-glukoza<br />
glukozis β-forma.<br />
wyalxsnarSi glukoza arsebobs samive formiT – cikluri α forma, wrfivi<br />
forma da cikluri β forma, maT So ris dinamikuri wonasworoba myar deba:<br />
cikluri wrfivi cikluri forma;<br />
α-forma aldehiduri forma β-forma.<br />
119
120<br />
glukozis qimiuri Tvisebebi<br />
rogorc ukve aRvniSneT, glukoza aldehidspirtia, amitom is Sedis aldehidisa<br />
da spirtisaTvis damaxasiaTebel qimiur reaqciebSi.<br />
glukoza Sedis reaqciaSi spilenZ(II)-is hidroqsidTan da glicerinis msgavsad<br />
kaSkaSa – lurji feris xsnars warmoqmnis.<br />
glukoza warmoqmnis esterebs.<br />
daasabuTeT am ori mosazrebis marTebuloba Teoriuli mtkicebulebis safuZvelze.<br />
erTi maTgani mainc daasabuTeT eqsperimentulad.<br />
rogorc aldehidi glukoza iJangeba da warmoqmnis Sesabamis mJavas. magaliTad,<br />
is reaqciaSi Sedis vercxlis oqsidis amiakian xsnarTan:<br />
CH2OH – (CHOH) 4 – C O<br />
H + Ag CH<br />
2O 2OH – (CHOH) 4 – C O<br />
+ 2Ag↓<br />
OH<br />
SesaZlebelia agreTve, glukozis molekulaSi aldehidis<br />
jgufis aRdgena Sesabamis spirtamde.<br />
SeadgineT glukozis aRdgenis reaqciis<br />
toloba, Tu aRmdgenad gamoiyeneba wyalbadi,<br />
reaqciis Sedegad ki miiReba eqvsatomiani<br />
spirti. reaqciis katalizatoria Ni.<br />
glukozis aRdgeniT miRebul spirts (sorbits), Saqris<br />
Semcvlelad uniSnaven Saqriani diabetiT daavadebul avadmyofebs.<br />
glukoza Sedis e. w. duRilis reaqciaSi. es reaqcia mimdinareobs<br />
gansakuTrebuli biologiuri katalizatorebis<br />
– fermentebis monawileobiT. duRilis reaqciebs Soris mniSvnelovania<br />
spirtuli da rZemJavuri duRili.<br />
spirtuli duRilis reaqcias odiTgan iyenebda adamiani alkoholuri sasmelebis<br />
misaRebad:<br />
C 6H 12O 6<br />
spirtuli<br />
duRilis<br />
baqteriebi<br />
2C 2H 5OH + 2CO 2@↑.<br />
mrewvelobaSi glukozis spirtuli<br />
duRiliT miiReba eTilis spirti.<br />
glukozas iyeneben sakonditro warmoebaSi<br />
marmeladis, karamelisa da<br />
sxva tkbileulis dasamzadeblad.<br />
rZemJavuri duRili mimdinareobs<br />
rZemJavuri duRilis baqteriebis<br />
monawileobiT:<br />
C6H12O6 2CH3 – CH – COOH<br />
rZemJavuri<br />
duRilis<br />
baqteriebi<br />
e s r e a q c i a<br />
iwvevs rZis am-<br />
Javebas.<br />
duRilis baqte<br />
riebi iwvevs<br />
b o s t n e u l i s<br />
dam Javeba sac.<br />
`vercxlis sarke~,<br />
romelic glukozis<br />
vercxlis oqsidis<br />
amiakian xsnarTan<br />
urTierTqmedebiT<br />
warmoiqmneba.<br />
OH rZemJava
1. SeiZleba Tu ara glukozis wrfivi forma CaiTvalos cikluri formis<br />
izomerad? pasuxi daasabuTeT.<br />
<br />
2. SeadareT glukozisa da glicerinis Tvisebebi,<br />
Sedarebis Sedegi gamosaxeT diagramiT.<br />
gansx.<br />
msgavsi gansx.<br />
glukoza glicerini<br />
3. sorbiti eqvsatomiani spirtia, misi miReba SesaZlebelia glukozisagan.<br />
SeadgineT am reaqciis toloba. miuTiTeT,M rogor SeiZleba sorbitSi spirtis<br />
funqciu ri jgufebis aRmoCena?<br />
4. 59 g glukozis sruli daJangvisas gamoiyo 146,3 kj siTbo. gamoTvaleT am<br />
reaqciis siTburi efeqti.<br />
121
122<br />
glukozasTan vercxlis oqsidis amiakiani<br />
xsnaris urTierTqmedeba<br />
praqtikumi<br />
1 sinjaraSi CaasxiT 1-2 ml glukozis wyalxsnari, daamateT 1-2 ml<br />
vercxlis oqsidis amiakiani xsnari. sinjara gaacxeleT mduRare wylis<br />
abazanaze.<br />
ras amCnevT? glukozis Ria Tu ciklur formas axasiaTebs vercxlis sarkis<br />
reaqcia?<br />
SeadgineT reaqciis toloba.<br />
2 spilenZ (II)-is hidroqsidis urTierTqmedeba glukozasTan<br />
a) sinjaraSi CaasxiT 0,5 ml glukozis wyalxsnari da 2 ml natriumis<br />
hidroqsidis wyalxsnari. miRebul narevs daamateT 1 ml Sabiamnis wyalxsnari.<br />
ras amCnevT?<br />
b) miRebul wyalxsnars frTxilad daamateT 1 ml wyali. sinjara ise<br />
daamagreT Stativze, rom spirtquris aliT mxolod xsnaris zeda nawili<br />
Tbebodes. rogorc ki feris cvlileba daiwyeba, SewyviteT gaTboba.<br />
g) warmoqmnili spilenZ (II)-is hidroqsidis naleqi ratom gardaiqmneba<br />
lurji feris gamWvirvale xsnarad? romeli funqciuri jgufi iwvevs am<br />
cvlilebas?<br />
d) ratom icvleba gacxelebisas sareaqcio narevis feri lurjidan<br />
wiTlamde? ra aris wiTeli naleqi?<br />
romeli funqciuri jgufi iwvevs am reaqcias?<br />
SeadgineT reaqciis toloba.
20<br />
disaqaridebi<br />
disaqaridebi farTodaa gavrcelebuli bunebaSi. saqaroza, anu Cveulebrivi<br />
Saqari, rZis Saqari laqtoza da alaos Saqari maltoza disaqaridebia.<br />
disaqaridebidan yvelaze mniSvnelovani saqarozaa.<br />
disaqaridis molekula ori monosaqari<br />
saqarozas kubaSi iReben disagan Sedgeba. ori<br />
Saqris lerwmisagan, evro-<br />
monosaqaridis molekupaSi<br />
– Saqris Warxlisagan;<br />
agaris Saqars Saqris lis naSTebi ki erTmaneTs<br />
Warxlisagan iReben. uerTdeba monosaqaridis<br />
ori moleku lidan erTi<br />
molekula wylis moxle-<br />
Cis Sedegad.<br />
magaliTad, saqarozas molekula warmoiqmneba glukozisa da fruqtozis<br />
mo le kulebidan erTi molekula wylis moxleCiT:<br />
C 6H 12O 6 + C 6H 12O 6 → C 12H 22O 11 + H 2O<br />
glukoza fruqtoza saqaroza<br />
polisaqaridi – saxamebeli da celuloza<br />
fizikuri Tvisebebi<br />
saxamebeli TeTri, amorfuli fxvni lia, civ wyalSi ar ixsneba, cxel wyalSi<br />
ki ijirjveba da warmoqmnis koloidur xsnars (sur. 37)<br />
celuloza myari, boW kovani nivTierebaa, wyalSi<br />
ar ixsneba. celulozaa bamba da filtris qaRaldi<br />
(sur. 38).<br />
sur.37 saxamebeli.<br />
disaqaridi fermentis<br />
moqmedebiT hidrolizdeba<br />
da iSleba isev glukozad<br />
da fruqtozad.<br />
saqarozas nawi lob rivi<br />
hidrolizi mimdinareobs<br />
murabis momzadebis procesSic.<br />
celuloza mom di nareobs<br />
la Tinuri sityvidan<br />
`celula~, rac<br />
qarTulad `uj reds~<br />
niSnavs.<br />
sur.38 bambis buCqi.<br />
123
124<br />
bunebaSi gavrceleba<br />
saxamebeli Sedis mce nareuli ujredis cito plazmaSi, sakvebi nivTierebis<br />
maragis Semcveli marcvlebis saxiT.<br />
kartofilis bolqvSi<br />
20%-mde saxamebelia<br />
celuloza mcenareuli ujredis garsis Semadgeneli aucilebeli komponentia.<br />
SeiZleba iTqvas, rom mcenarisaTvis celuloza `saaRmSeneblo~,<br />
`makonstruirebel~ funqcias asrulebs. bambis boWkoSi 95% celulozaa,<br />
selis boWkoSi celulozas Semcveloba 80% aRwevs, xis merqanSi ki – 59%-s<br />
uaxlovdeba.<br />
saxameblisa da celulozis agebuleba<br />
saxameblisa da celulozas<br />
monomeruli er Teulebi<br />
sxvada sxva nairadaa<br />
agebuli. saxa meblis monomeruli<br />
erTeuli α<br />
glukozis naSTia, celulozas<br />
mono meruli er-<br />
Teuli ki – β glukozisa.<br />
saxameblis<br />
monome ruli<br />
erTeuli<br />
celulozas<br />
monomeruli<br />
erTeuli<br />
xorblisa da simindis<br />
marcvlebSi 70%-mdea.<br />
makromolekulebi, romelsac amilozas<br />
uwodeben, xolo ganStoebuli struqtura<br />
aqvs makromolekulebs, romelsac<br />
amilopeqtini ewodeba (sur. 39). saxamebelSi<br />
daaxloebiT 10-20% amiloza da<br />
80-90% amilopeqtinia. amiloza ixsneba<br />
cxel wyalSi, amilopeqtini ki – ijirjveba,<br />
amitom warmoiqmneba bubko, romelic<br />
gamoiyeneba qsovilebis gasaxameblad.<br />
brinjSi ki saxameblis<br />
Semcveloba<br />
80%-s aRwevs.<br />
saxameblisa da celulozis Sedgeniloba<br />
ga moisa xeba zogadi formuliT (C 6H 10O 5) n. ganmeorebadi<br />
mono meruli erTeulebis raodenoba<br />
saxamebelSi ramdenime aTass aRwevs. saxameblisagan<br />
gansxvavebiT celulo za Si monomeruli<br />
erTeulebis raodenoba mniSvnelovnad metia, e.<br />
i. metia misi mo lekuluri masac, romelic xSir<br />
SemTxvevaSi ramdenime milions aRwevs.<br />
celuloza da saxamebeli gansxvavdeba ara<br />
mxo lod molekuluri masiT, aramed agebulebiTac.<br />
kerZod, saxameblis makromolekulas<br />
SesaZloa orgvari struqtura hqondes<br />
– wrfivi da ganStoebuli. wrfivia saxameblis<br />
(a)<br />
(b)<br />
sur. 39 a) amiloza; b) amilopeqtini.
celulozas makromolekulebs mxolod wrfivi<br />
struqtura aqvs (sur. 40).<br />
qimiuri Tvisebebi<br />
celuloza da saxamebeli hidrolizdeba da<br />
warmoqmnis glukozas. es procesi mimdinareobs<br />
zogierTi cxovelis saWmlis<br />
momnelebel sistemaSi<br />
mravldebian mikroorganizmebi,<br />
romlebic gamoimuSaveben<br />
celulozis gadasamuSaveblad<br />
saWiro<br />
fermentebs, am fermentebis<br />
monawileobiT celulozas<br />
makromolekulaSi β glukozis<br />
naSTebs Soris arsebuli<br />
bmebi wydeba da celuloza<br />
iwyebs daSlas.<br />
aseT fermentebs adamianis<br />
organizmi ar gamoimuSavebs,<br />
amitom celuloza<br />
adamianisaTvis sakvebad ar<br />
gamodgeba.<br />
mineraluri mJavas,<br />
magaliTad gogirdmJavas<br />
Tan xlebiT:<br />
(C6H10O5) n + (n‑1)H2O → nC6H12O6 .<br />
polisaqaridi glukoza<br />
rac Seexeba saxamebels, is cocxal organizmSi<br />
hidrolizdeba safexurebad:<br />
saxamebeli → deqstrinebi →<br />
glukoza:<br />
maltoza →<br />
(C6H10O5) → (C6H10O5) → nC12H22O11 → nC6H12O6 .<br />
saxamebeli deqstrinebi maltoza glukoza<br />
Tu celulozas monomeruli erTeulis<br />
struqturas daakvirdebiT, Se am CnevT, rom masSi<br />
sami Tavisufali spirtuli jgufia, amis gamo<br />
celulozas molekuluri formula SeiZleba<br />
asec gamoisaxos:<br />
[C 6H7O2(OH) 3] n .<br />
TvisebiTi reaqcia<br />
am spirtuli jgu -<br />
fe bis xarjze celuloza<br />
warmoqmnis<br />
esters. magaliTad,<br />
c e l u l o z a s d a -<br />
muSavebisas azot mJa vasa da gogirdmJa vas nareviT,<br />
pirobe bis mixedviT miiReba mono, di da trinitroceluloza:<br />
OH<br />
OH<br />
C6H7O2 + nHNO3 → C6H7O2 OH + nH [<br />
2O<br />
OH n<br />
ONO n<br />
2<br />
mononitroceluloza<br />
C 6H 7O 2<br />
OH[<br />
[[<br />
OH<br />
OH<br />
OH<br />
[[<br />
C 6H 7O 2<br />
OH<br />
OH<br />
OH<br />
[[<br />
[[<br />
+ 2nHNO3 → C6H7O2 n<br />
+ 3nHNO3 → C6H7O2 n<br />
OH<br />
ONO2 ONO2 n<br />
dinitroceluloza<br />
[[<br />
ONO2 ONO2 ONO2 trinitroceluloza<br />
n<br />
+ 2nH 2O<br />
+ 3nH 2O<br />
sur. 40. celulozas makromoleku<br />
lebis orientacia<br />
celulozasagan gansxvavebiT<br />
saxamebeli iod-<br />
Tan urTierTqmedebisas<br />
warmoqmnis lurji<br />
feris nivTierebas. es<br />
aris Tvisebi-<br />
Ti reaqcia,<br />
rogorc iodis,<br />
ise saxameblisaRmosaCenad.<br />
esteris warmoqmnis<br />
unari aqvs saxamebelsac<br />
da celulozasac.<br />
magram, saxa meblis<br />
mier warmoqmnili esteri<br />
jer-jerobiT<br />
praqtikaSi naklebad<br />
gamoiyeneba. celulozas<br />
mier warmoqmnil<br />
esters ki farTo<br />
praqtikuli gamo yeneba<br />
aqvs.<br />
125
126<br />
iseve, rogorc nitrojgufis Semcveli sxva nivTierebebi, nitrocelulozas<br />
yvela saxeoba cecxlsaSiSia. gansakuTrebiT ki trinitroceluloza, romelic<br />
feTqebadi nivTierebaa. `piroqsilinis~ saxelwodebiT is farTod gamoiyeneba<br />
ukvamlo denTis dasamzadeblad.<br />
celulozas urTierTqmedebisas ZmarmJavasTan aseve SesaZlebelia nitrocelulozas<br />
analogiuri produqtebis, diacetilcelulozasa da triacetilcelulozas<br />
miReba:<br />
[C6H7O2(OH) 3]n + 3nCH3COOH → [C6H7O2(OCOCH3) 3]n + 3nH2O. triacetilceluloza<br />
acetilceluloza gamoiyeneba laqebis da saRebavebis<br />
warmoebaSi.<br />
triacetilcelulozasagan amzadeben agreTve rentgenis<br />
firsa da kinofirs, romelic cecxlgamZlea.<br />
cxeli siTxe<br />
cxeli haeri<br />
filieri, anu<br />
Tvalaki<br />
filieri<br />
Saxta<br />
boWko<br />
acetaturi boWkos formirebis sqema<br />
aceti lceluloza gamoiyeneba xelovnuri abreSumis<br />
misaRebad. saamisod acetilcelulozas xsnian acetonSi.<br />
Semdeg am xsnars gaatareben wvrilnaxvretebian<br />
filierSi. filieridan gamosuli xsnari wvrili<br />
Wavlis saxiT niavdeba Tbili haeriT. am dros acetoni<br />
swrafad orTqldeba, xolo gamSrali acetilceluloza<br />
warmoqmnis wvril, mbzinav Zafebs. mas axveven<br />
da misgan qsovils _ acetatur abreSums qsoven.<br />
acetonSi gaxsnili acetilcelulozas<br />
xsnari.<br />
acetaturi abreSumi
1. ori uetiketo WurWlidan erT-erTSi myari sapnis fxvnilia, meoreSi ki –<br />
saxamebeli. rogor ganasxvavebT maT erTmaneTisagan?<br />
2. rogor daamtkicebT, rom mwife vaSlSi aris glukoza, mkvaxe vaSlSi ki ar<br />
aris? daasabuTeT Tqveni varaudi.<br />
3. sityvebis nacvlad CawereT im nivTierebaTa formulebi, romelTa saSualebiTac<br />
SesaZlebelia aRniSnuli gardaqmnebis ganxorcieleba.<br />
ricxvebis nacvlad ki miuTiTeT saWiro reagentebi:<br />
saxamebeli glukoza eTanoli Zmris aldehidi ZmarmJava<br />
magniumis acetati.<br />
4. ratomaa moxarSuli kartofili webovani? daasabuTeT Tqveni mosazreba.<br />
5. mxolod fizikuri Tvisebebis safuZvelze rogor ganvasxvavoT xorblis<br />
fqvili saxameblisagan?<br />
<br />
6. ra garegnuli niSnebi eqneba qvemoT aRweril reaqciebs:<br />
a) kaliumis iodidis ufero xsnars daamates saxameblis bubko;<br />
b) iodis spirtxsnars daamates saxameblis bubko;<br />
g) natriumis iodidis ufero xsnars daamates qloriani wyali, Semdeg ki saxameblis<br />
bubko.<br />
7. ra masis saqaroza SeiZleba miviRoT1,5 t Saqris Warxlidan, Tu masSi saqarozas<br />
masuri wili 20 % -ia?<br />
127
128<br />
21<br />
aminebi<br />
azotSemcveli naerTebi<br />
arsebobs uamravi organuli naerTi, romelic<br />
azots Seicavs, maT ASorisaa aminebi. aminebis SedgenilobaSi<br />
azoti Sedis aminojgufis saxiT NH 2.<br />
bunebaSi aminebi warmoiqmneba cilovani nivTierebebis<br />
xrwnis Sedegad _ am dros warmoqmnili arasasiamovno<br />
suni aminebis sunia. magaliTad, gafuWebuli<br />
Tevzis suni trimeTilaminis warmoqmniTaa ganpirobebuli.<br />
arsebobs aminebis sami tipi:<br />
pirveladi amini – rodesac amiakis molekulaSi<br />
wyalbadis erTi atomia Canacvlebuli radikaliT:<br />
CH 3 – NH 2<br />
meTilamini<br />
pirvelad amins ganixilaven agreTve, rogorc<br />
naxSirwyalbadis nawarms, sadac wyalbadis atomi<br />
Canacvlebulia NH 2 jgufiT (aminojgufiT).<br />
NH 2 jgufi aminebis funqciuri jgufia.<br />
amini SesaZlebelia<br />
ganvixiloT rogorc<br />
ami akis nawarmi, ro mlis<br />
molekulaSic erTi, ori<br />
an samive wyalbadato mi<br />
Cana cvlebulia naxSirwyalbadis<br />
radi kaliT:<br />
meoreuli amini – rodesac<br />
amiakis molekulaSi<br />
ori wyalbadis<br />
atomia Canacvlebuli<br />
naxSirwyalbaduri radikaliT:<br />
CH 3CH 2<br />
CH 3CH 2<br />
dieTilamini<br />
axla, amiakis molekulaSi samive wyalbadis atomi CaanacvleT eTilis radikaliT<br />
da Tqven mesameul amins miiRebT. misi saxelwodebaa trieTilamini.<br />
naerTs, romlis molekulaSic aminojgufi uSualodaa<br />
dakavSirebuli aromatul birTvTan, aromatuli amini<br />
ewodeba. umartivesi aromatuli aminia aminobenzoli, anu<br />
anilini.<br />
izomeria da nomenklatura<br />
aminebisaTvis damaxasiaTebelia:<br />
1) naxSirbadovani ConCxis izomeria<br />
CH 3 – CH 2 – CH 2 – CH 2 – NH 2<br />
butilamini<br />
CH 3 – CH –CH 2 – NH 2<br />
CH 3<br />
izobutilamini<br />
NH 2<br />
NH<br />
anilini<br />
2) funqciuri jgufis mdebareobis<br />
izomeria<br />
CH3 – CH2 – CH2 – NH2 propilamini<br />
CH 3 – CH – CH 3<br />
NH 2<br />
izopropilamini
uwodebdiT Tu ara pirvelad, meoreul<br />
da mesameul amins erTmaneTis izomerebs?<br />
daasabuTeT Tqveni varaudi SemoTavazebuli<br />
magaliTebis ganxilvis safuZvelze.<br />
fizikuri Tvisebebi<br />
iseTi aminebi, rogoricaa meTilamini, dimeTilamini da<br />
trimeTilamini airadi nivTierebebia. sxva grZeljaWviani aminebi wyalSi<br />
xsnadi siTxeebia. aqvs amiakis msgavsi, Taviseburi suni, romelic Tevzis<br />
sunsac mogvagonebs.<br />
rogorc pirveladi, ise meoreuli aminis molekulebi erTmaneTTan amyarebs<br />
molekulaTaSoris wyalbadur bmebs. amiT aixsneba is faqti, rom maTi lRobisa<br />
da duRilis temperatura mniSvnelovnad maRalia im naerTebis lRobisa<br />
da duRilis temperaturasTan SedarebiT, romelTac daaxloebiT igive<br />
molekuluri masa aqvs, magram ar gaaCnia wyalbaduri bmebis damyarebis unari.<br />
mesameul aminebSi wyalbaduri bma ar arsebobs.<br />
qimiuri Tvisebebi<br />
aminis molekulaSi, amiakis msgavsad, azotis atoms aqvs Tavisufali eleqtronuli<br />
wyvili. am eleqtronuli wyvilis saSualebiT is amyarebs kovalentur bmas<br />
donorul – aqceptoruli meqanizmiT, sadac donoris rolSi azotis atomia:<br />
amis gamo amins SeuZlia miierTos wyalbadis kationi da gamoamJRavnos tutis<br />
Tvisebebi.<br />
1 CH3 – CH 2 – NH 2 + → [CH 3 – CH 2 – NH 3] + + OH –<br />
eTilamoniumis ioni<br />
CH 3 – CH 2 – CH 2 – NH 2 propilamini<br />
CH 3 – CH 2 – NH – CH 3 meTileTilamini<br />
CH 3 – N – CH 3 trimeTilamini<br />
│<br />
CH 3<br />
wyalSi gaxsnisas eTilamini, amiakis msgavsad, warmoqmnis eTilamoniumis<br />
dadebiTad damuxtul ions da uaryofiTad damuxtul<br />
hidroqsidis ions, amitom aminis wyalxsnars tute reaqcia aqvs:<br />
2 amiakis msgavsad, amini urTierT qme debs mJavasTan:<br />
2CH3 – CH2 – NH2 + H2SO4 → [CH3– CH2– NH3] 2SO4. eTilamoniumis sulfati<br />
2NH3 + H2SO4 → ( NH4) 2SO4 amoniumis sulfati<br />
aminis saxelwode bis<br />
Sedgenisas Camo Tvlian<br />
Camnacvleblebs da umateben<br />
sufiqss – `amini~.<br />
129
130<br />
aRsaniSnavia, rom aminis fuZe<br />
Tviseba Warbobs amiakis fuZe Tvisebas,<br />
aq Tavs iCens alkilis radikalis<br />
gavlena aminojgufze. es jgufi bmis<br />
eleqtronul simkvrives gadaswevs<br />
aminojgufis azotis mxa res, amis gamo<br />
azotis atomze Tavisu fali eleqtronuli<br />
wyvilis simkvri ve ufro maRalia,<br />
vidre amiakSi, rac aZlierebs<br />
wyal badis kationis mierTebis unars,<br />
e. i. fuZe Tvisebebs:<br />
CH 3CH 2NO 2<br />
3 amini iwvis da wvis Sedegad warmoqmnis<br />
wyals, naxSirorJangsa da Tavisufal azots:<br />
miReba<br />
4CH 3NH 2 + 9O 2 → 4CO 2+ 10H 2O+ 2N 2 .<br />
aminebis miReba SesaZlebelia nitrona er-<br />
Tebis aRdgeniT. aRmdgenlad gamo iyeneba<br />
wyalbadi gamoyofis momentSi (ato muri<br />
wyalbadi, romelic sareaqcio areSi warmoiqmneba.<br />
magaliTad, rkinis an TuTiis<br />
urTierTqme debiT marilmJavasTan)L:<br />
6 [ H ]<br />
Zn + HCl<br />
CH 3CH 2NH 2 + 2H 2O.<br />
eTilamini<br />
amave gziT SesaZlebelia aromatuli aminis miRebac. ase magaliTad, mre wvelobaSi<br />
anilins iReben nitrobenzolis aRdgeniT, risTvisac am ukanasknels<br />
acxeleben rkinasTan da wylis orTqlTan erTad:<br />
6 [ H ]<br />
Fe + H2 O , t<br />
C6H5NO2 C6H5NH2 + 2H2O. nitrobenzoli anilini<br />
aminebi farTod gamoiyeneba samedicino<br />
preparatebis, polimerebisa da sxvaTa misa<br />
Rebad, gansakuTrebiT didi praqtikuli<br />
mniSvneloba aqvs anilins. is gamo iyeneba<br />
saRebavebis, medikamentebis – sulfanilamiduri<br />
preparatebis, polimerebis misa-<br />
Rebad.<br />
anilinis miRebis es xerxi SeimuSava<br />
rusma mecnierma n. zininma,<br />
amitom am reaqcias zininis<br />
reaqcia ewodeba.<br />
1. SeadgineT propileTilaminis izomerebis struqturuli formulebi.<br />
2. fuZe Tvisebebis zrdis mixedviT ganalageT nivTierebebi:<br />
CH 3NH 2 , NH 3 , (CH 3) 2NH, C 6H 5NH 2 . daasabuTeT Tqveni varaudi.<br />
<br />
3. SeadgineT meTanidan anilinis miRebis gardaqmnaTa sqema yvela Sualeduri<br />
produqtis miTiTebiT, Semdeg ki dawereT reaqciaTa tolobebi.
4. ramdeni grami anilinis miReba SeiZleba 210 g nitrobenzolisagan, Tu am<br />
reaqciis praqtikuli gamosavlianoba Teoriulis 75%-ia?<br />
5. rodesac Tqveni mSoblebi skolaSi swavlobdnen<br />
80-90-ian wlebSi saqarTveloSi moqmed qimiis saxelmZRvaneloSi aminebis Sesaxeb<br />
weria:<br />
`azoti organul naerTebSi SeiZleba Sediodes ara mxolod<br />
azotmJavas naSTis saxiT (gaixseneT nitrobenzoli C 6H 5NO 2 da<br />
celoluzis rTuli eTeri _ trinitrati [C 6H 7O 2ONO 2] 3) 11, aramed<br />
sxva formiTac. cnobilia mravali organuli naerTi, romelSic<br />
azoti amiakis saxiT Sedis, magaliTad,<br />
H<br />
CH3 – N<br />
H<br />
CH3 CH3 N – H<br />
CH3 CH3 N<br />
meTilamini dimeTilamini trimeTilamini<br />
sityvierad CamoayalibeT, ra iciT ufro meti aminebis agebulebis, izomeriisa<br />
da nomenklaturis Sesaxeb.<br />
CH 3<br />
CH3 C2H5 N – H<br />
H<br />
C6H5 – N<br />
H<br />
C6H5 C6H5 N – H<br />
meTileTilamini fenilamini (anilini) difenilamini<br />
yvela es naerTi aminebis klass ekuTvnis.<br />
aminebi SeiZleba ganvixiloT rogorc amiakis nawarmebi,<br />
romelTa molekulaSi wyalbadis erTi an ramdenime atomi Canacvlebulia<br />
naxSirwyalbadebis radikalebiT.~<br />
131
132<br />
22<br />
aminomJavebi<br />
yvelaze mniSvnelovani, rac aminomJavas Sesaxeb Sei-<br />
Zleba iTqvas, aris is, rom Sedis cilis SedgenilobaSi.<br />
Tumca, arsebobs Cveni janmrTelobisa da komfortisaTvis<br />
saWiro araerTi nivTiereba, romelic aminomJavasagan<br />
miiReba, aminomJavas mravalferovan Tvisebebs ki misi<br />
Sedgenilobis Tavisebureba ganapirobebs.<br />
saxelwodebidanac gamomdinareobs, rom aminomJavas<br />
mo lekulaSi aucileblad Sedis ori funqciuri jgufi<br />
– NH2 aminojgufi da – C O karboqsilis jgufi.<br />
OH<br />
amitom aminomJavas zogadi formula SesaZloa iyos mxolod<br />
aseTi:<br />
H2N – CH – COOH<br />
R<br />
izomeria da nomenklatura<br />
aminomJavebis izomeria ganpirobebulia:<br />
1<br />
naxSirbadovani ConCxis agebulebiT:<br />
4 3 2 1<br />
CH3 – CH2 – CH – C<br />
NH 2<br />
2– aminobutanmJava<br />
CH 3<br />
3 2 1<br />
CH3 – C – C<br />
NH 2<br />
O<br />
OH<br />
O<br />
OH<br />
2-amino-2-meTilpropanmJava<br />
2<br />
aminomJava karbon<br />
mJa vas iseTi<br />
na war mia, rome lic<br />
Se icavs naxSirwyal<br />
badis radikalTandakavSirebul<br />
ami no jgufs<br />
–NH 2 da karboqsilis<br />
jgufs –<br />
–COOH.<br />
aminojgufis mdebareobiT:<br />
aminomJavas sa xel wodebis Sedge<br />
nisas danomvra iwyeba im naxSirbadatomidan,<br />
romelic Sedis karboqsilis<br />
jgufSi.<br />
Tumca, ufro xSirad aminomJavas saxelwodebis Sedgenisas<br />
amino jgufis mdebareobas aRniSnaven berZnuli<br />
asoebiT: α, β, γ da a.S. ase<br />
magaliTad 2 –amonobutan- γ β α O<br />
mJavas saxelwodeba iqneba<br />
α-aminobutanmJava.<br />
CH3 – CH2 – CH – C<br />
OH<br />
cilis sinTezSi monawileobs daaxloebiT 20 sxvadasxva aminomJava. zogier-<br />
Tis tri vi aluri saxelwodeba mocemulia cxril Si:<br />
NH 2
cxrili: zogierTi aminomJavas saxelwodeba da struqturuli formula<br />
mJavas saxelwodeba formula<br />
glicini<br />
alanini<br />
cisteini<br />
serini<br />
glutaminmJava<br />
lizini<br />
fenilalanini<br />
H 2N – CH 2 – COOH<br />
CH 3–CH–COOH<br />
|<br />
NH 2<br />
HS – CH2 – CH – COOH<br />
|<br />
NH2 HO – CH2 – CH – COOH<br />
|<br />
NH 2<br />
HOOC – CH 2 – CH 2 – CH – COOH<br />
|<br />
NH 2<br />
H 2N – (CH 2) 4 – CH – COOH<br />
|<br />
NH 2<br />
– CH 2– CH – COOH<br />
|<br />
NH 2<br />
tirozini HO – – CH 2 – CH – COOH<br />
|<br />
NH 2<br />
fizikuri Tvisebebi<br />
aminomJavebi ufero, kristaluri nivTierebebia. rogorc wesi ixsnebian<br />
wyalSi da ar ixsnebian eTerSi. aminomJava SeiZleba iyos tkbili, mware, an<br />
saerTod ar hqondes gemo.<br />
bunebrivi 20 α-aminomJavadan, naxevari e.w. Seucvleli aminomJavaa, radgan<br />
isini organizmSi ar sinTezirdeba da miewodeba sakvebis saSualebiT. Tu<br />
sakvebSi aseTi aminomJavebis raodenoba arasakmarisia, organizmis normaluri<br />
ganviTareba da funqcionireba ferxdeba.<br />
qimiuri Tvisebebi<br />
nivTierebis Sedgeniloba mis Tvisebebs ganapirobebs. amitom, dabejiTebiT<br />
SeiZleba vTqvaT, rom aminomJavas aqvs rogorc aminebis, ise karbonmJavebis<br />
Tvisebebi. Tumca, axla Cven ganvixilavT aminomJavebis iseT mniSvnelovan qimiur<br />
Tvisebebs, romelic am ori funqciuri jgufis erTdrouli arsebobiTaa<br />
gamowveuli. es aris amfoteroba da peptidebis warmoqmna. aminomJava amfoteruli<br />
organuli nivTierebaa, tutesTan urTierTqmedebs ro gorc mJava,<br />
xolo mJavasTan – rogorc fuZe:<br />
O<br />
H2N – CH – C<br />
OH<br />
R<br />
+ NaOH → H2N – CH – C O<br />
O – Na + H2O R<br />
mJava<br />
HOOC – CH – NH 2 + HCl → [ HOOC – CH – NH 3[<br />
R<br />
R<br />
fuZe<br />
+<br />
Cl –<br />
133
134<br />
aminomJavebis umniSvnelovanesi Tviseba erTmaneTTan urTierTqmedebaa<br />
– mo lekulaTSorisi kondensacia, romlis drosac warmoiqmneba amiduri<br />
dajgufeba – NH – CO da gamoiyofa wyali.<br />
mJava urTierTqmedebs fuZesTan, fuZe ki mJavasTan. amitom cxadia, rom<br />
erTi aminomJava karboqsilis jgufiT reaqciaSi Sedis meore aminomJavas aminojgufTan:<br />
a-aminomJavebisagan<br />
war moq mnil poliamids<br />
polipeptidi ewodeba.<br />
dimeri<br />
rogorc xedavT, dimeric Seicavs orive funqciur<br />
jgufs, amitom SesaZle belia reaqcia kvlav<br />
gagrZeldes maRalmolekuluri naerTis – polimeris<br />
warmoqmnamde. magaliTad,<br />
nNH 2 – (CH 2) 5 – COOH → (... – NH – (CH 2) 5) – CO – ...)n + +(n‑1)H 2O.<br />
aseTi reaqciis Sedegad miRebuli polimerebi Seicavs amiduri fragmentebis<br />
did raodenobas da amitom ewodeba poliamidi.<br />
aminomJavebi farTod gamoiyeneba medicinaSi. magaliTad, nervuli sistemis<br />
daavadebisas rekomendebulia glutaminis mJavas gamoyeneba:<br />
HOOC – CH – CH 2 – CH 2 – COOH<br />
|<br />
NH 2<br />
d a j g u f e b a s<br />
polipeptidis mo lekulaSi<br />
amiduri an peptiduri<br />
dajgufeba ewodeba.<br />
amave mJavas, an mis marils umateben konservebsa da sxva sakveb koncentratebs<br />
gemos gasaumjobeseblad. aminomJavebs uniSnaven avadmyofs mZime operaciis<br />
Semdeg, organizmis gamofitvisas da a.S.<br />
normaluri, anu wrfivi agebulebis aminomJavebi, aminokapronmJava<br />
H 2N – (CH 2) 5 – COOH da aminoenantis mJava H 2N – (CH 2) 6 – COOH gamoiyeneba sinTezuri<br />
boWkos, kapronis da enantis misaRebad.<br />
<br />
1. SeadgineT reaqciaTa tolobebi, romlebic asaxavs aminopropanmJavas<br />
urTierT qmedebas:<br />
a) gogirdmJavasTan;<br />
b) meTilis spirtTan;<br />
g) natriumis hidroqsidTan.
2. SeadgineT ε-aminokapronmJavas polikondensaciis reaqciis toloba, Tu<br />
misi formulaa:<br />
H 2N – (CH 2) 5 – C<br />
O<br />
OH<br />
3. SeadgineT dipeptidis warmoqmnis reaqciis toloba aminoZmarmJavasa da<br />
α-aminopropanmJavasgan.<br />
4. SeadareT polimerizaciis da polikondensaciis reaqciebi, Sedarebis<br />
Sedegebi asaxeT diagramiT.<br />
135
136<br />
23<br />
sinjaraSi moTavsebuli erTi α− aminomJava Cveulebrivi organuli niv-<br />
Tierebaa, is umniSvnelovanesi maSin xdeba, rodesac isev α− aminomJavebTan<br />
urTierTqmedebiT peptids warmoqmnis, radgan peptidis warmoqmna ukavSirdeba<br />
cilis warmoqmnas, cila da sicocxle ki ganuyofeli cnebebia, sadac cila ar<br />
aris, iq arc sicocxlea.<br />
qimiuri TvalsazrisiT cila biologiuri polimeria, romelic<br />
α-aminomJavebis naSTebisgan Sedgeba. misi molekuluri masa ramdenime aTasidan<br />
ramdenime milionamde meryeobs.<br />
ganvixiloT peptidis miRebis reaqcia umartivesi α-aminomJavebisagan.<br />
ori aminomJavasagan miRebul peptids dipeptidi ewodeba.<br />
glicini<br />
cilebi<br />
O H<br />
O H<br />
H 2NCH 2C OH + H N – CH – COOH → H 2O + H 2N – CH 2 – C – N – CH – COOH<br />
CH 3<br />
alanini<br />
dipeptidi isev urTierTqmedebs α-aminomJavasTan<br />
da axla tripeptidi warmoiqmneba:<br />
O H O H<br />
H2N – CH2 – C – N – CH – C – OH + H N – CH2 – COOH → H2O +<br />
glicini<br />
CH 3<br />
O H O H<br />
+ H2N – CH2 – C – N – CH – C – N – CH2 –COOH<br />
CH 3<br />
tripeptidi – glicilalanilglicini<br />
mravali α-aminomJavebisagan warmoqmnil peptids<br />
polipeptidi ewodeba, rac zogadad ase Caiwereba:<br />
H O H O H O<br />
. . . – N – CH – C – N – CH – C – N – CH – C – . . .<br />
CH 3<br />
dipeptidi<br />
m i R e b u l t r i -<br />
p e p t i d s , m o l e k u -<br />
laSi Semavali aminomJavebis<br />
naSTebis<br />
m i x e d v i T e w o d e b a<br />
glicilalanilglicini<br />
(Gly‑Ala‑Gly) (gliala-gli).<br />
zogadad<br />
peptids kiTxuloben<br />
bolo α-aminomJavaSi<br />
Canacvlebuli aminom-<br />
Javebis radikalebis<br />
mixedviT.<br />
R 1 R 2 R 3<br />
sadac R 1 , R 2 , R 3 – α-aminomJavas radikalebia. 50ze meti αaminomJavasagan Sedgenili<br />
polipeptidi ukve cilaa.<br />
cila erTi an ramdenime polipeptidis jaWvisagan Sedgeba, sadac<br />
α-aminomJavebis naSTebi erTmaneTs peptiduri jgufiT uerTdeba.<br />
peptiduri bmis garSemo atomebis brunva SeuZlebelia, amitom peptiduri<br />
bmiT dakavSirebuli atomebi erT sibrtyeSia moTavsebuli. cilis SedgenilobaSi<br />
monawileobs 20 sxvadasxva α-aminomJava. sami α-aminomJavas erTmaneTTan<br />
SeerTebis 6 varianti arsebobs, e. i. maTi SeerTebiT 6 tripeptidi warmoiqmneba,<br />
xolo 20 α-aminomJavas SeerTebis varianti TiTqmis ganusazRvrelia, radgan<br />
erTi da igive aminomJava polipeptidis SedgenilobaSi SesaZlebelia ramdenjerme<br />
monawileobdes.
ama Tu im individualuri cilis makromolekulaSi<br />
aminomJavis naSTebis ganlagebis sakUu-<br />
Tari, mkacri Tanmimdevroba arsebobs, rac imiT<br />
gamoixateba, rom polimeris jaWvis gaswvriv<br />
ganlagebuli aminomJavas naSTSi Semavali radikalebi<br />
sxvadasxvaa. sqematurad aminomJavaTa<br />
naSTebis Tanmimdevroba SesaZloa gamoi saxos<br />
ase:<br />
..NH – CH – CO – NH – CH – CO – NH – CH – CO – ...<br />
| | |<br />
R 1 R 2 R 3<br />
aminomJavaTa naSTebis SeerTebis gansaz-<br />
Rvrul Tanmimdevrobas cilis pirveladi<br />
struqtura ewodeba.<br />
SesaZloa vifiq roT, rom polipeptiduri<br />
jaWvi wrfivi unda iyos. sinamdvi leSi mkacrad<br />
wrfivi struqtura cilebis arc ise did raodenobas<br />
aqvs. gamokvle vebma cxadyo, rom polipeptiduri<br />
jaWvis zogierTi ubnebi cilis molekulaSi<br />
spi raluradaa daxveuli. cilis spiraluri<br />
struqturis formirebas ganapirobebs C = O<br />
da N – H jgufebs Soris damyarebuli wyalbaduri<br />
bmebi.<br />
cilis pirveladi struqtura<br />
ZiriTadad gansazRvravs mis<br />
specifikur Tvisebebs. Tu Se icva<br />
la cilis pirveladi struqtura,<br />
icvleba misi Tvisebebic.<br />
magaliTad, adamianis hemoglobini<br />
Sedgeba 574 aminomJavas<br />
naSTisagan. Tu erT-erT jaWvSi<br />
glutaminmJavas naSTis nacvlad<br />
meore aminomJavas valinis naSTi<br />
Caenacvleba, maSin sisxlis wiTeli<br />
ujredebi iRebs namglisebur<br />
formas. aseT SemTxvevaSi adamiani<br />
sneuldeba namgliseburi<br />
anemiiT, romelic adreul asakSi<br />
iwvevs sikvdils.<br />
polipeptidebSi Semavali<br />
20 α-aminomJavidan 10 Sesa-<br />
Zlebelia adamianis organizmSi<br />
warmoiqmnas sxvadasxva<br />
nivTierebisgan. magram, danarCeni<br />
10 aminomJavas sin-<br />
Tezi adamianis organizmSi<br />
ar mimdinareobs. aseT aminomJavebs<br />
ZiriTadi aminom<br />
Javebi, anu Seucvleli aminomJavebi<br />
ewodeba. Seucvleli<br />
α-aminomJavebi adamianis<br />
organizmma sxvadasxva<br />
cilovani sakvebidan unda<br />
miiRos. cilas, romelic 10<br />
Seucvlel aminomJavas Seicavs<br />
srulfasovani cila<br />
ewodeba, cxoveluri cila<br />
da rZe srulfasovania, xolo<br />
zogierT mcenareul cilaSi<br />
erTi an ramdenime Seucvleli<br />
aminomJava ar Sedis. aseTi Ucila<br />
arasrulfasovani cilaa.<br />
soios cila Tavisi SedgenilobiT<br />
srul fasovania, magram<br />
simindis cila arasrulfasovania,<br />
radgan TiTqmis ar Seicavs<br />
or Seucvlel aminomJavas<br />
– lizinsa da triptofans.<br />
brinjis cilaSi cotaa lizini<br />
da treonini, xorbalSi ki –<br />
li zini. amitom srulfasovani<br />
kvebisaTvis racionSi unda<br />
Sediodes yvelanairi mcenareuli<br />
produqti.<br />
137
138<br />
cilis aseT struqturas meoreuli<br />
struqtura ewodeba.<br />
spiralis gareT mimarTulia aminomJavebis<br />
radikalebi R, romelTa SedgenilobaSi Sedis:<br />
karboqsilis jgufi – COOH, aminojgufi NH 2,<br />
hidroqsilis jgufi OH, sulfhidridis jgufi<br />
SH da sxva. spiralis gareT ganlagebuli<br />
es jgufebi urTierTzemoqmedebis Sedegad<br />
warmoqmnis sxvadasxva tipis bmebs: ionurs,<br />
kova lenturs, wyalbadurs da a.S. ris gamoc<br />
spirali ixveva da igragneba. cilis aseT struqturas<br />
cilis mesameuli struqtura ewodeba.<br />
rig SemTxvevebSi cila warmoqmnis meo Txeul<br />
struqturas. amis Sesaxeb 26-e paragrafSi visaubrebT.<br />
Tvisebebi<br />
cilis meoreul struqtura<br />
Si spiralis erTi xvia iseTi<br />
zomisaa, rom yoveli aminomJavas<br />
jgufis Tavze ganla-<br />
gebulia meoTxe aminomJavas<br />
jgufi, amitom maT Soris<br />
myar deba wyalbaduri bmebi,<br />
rac spiralis mdgradobas ganapirobebs.<br />
cilis mesameul struqtu<br />
ras aseTi forma aqvs.<br />
cilas mravalferovani Tvisebebi aqvs. zogierTi cila myari nivTierebaa,<br />
ar ixsneba wyalSi da marilTa xsnarebSi. Tumca, cilaTa umravlesoba Txevadi<br />
an labiseburi nivTierebaa, magaliTad, kvercxis cila albumini.<br />
wyalSi gaxsnisas cilebi warmoqmnis koloidur xsnars, wyalxsnaridan cila<br />
gamoileqeba mZime metalebis,<br />
tyviisa da vercxliswylis<br />
ma ri lebiT. amitomacaa mZime<br />
metalebis orTqli da maTi<br />
marilebi Zalian saSiSi co cxali<br />
organizmisaTvis.<br />
cilis gamoleqva SesaZlebelia<br />
mineraluri mJavebiT,<br />
magaliTad, azotmJaviT da<br />
gogirdmJaviT, formaliniT<br />
da fenoliT, agreTve eTilis<br />
spirtiT. swored amitomaa<br />
sur.41. cilis denaturirebis sqema<br />
SesaZlebeli mcenareebisa
da cxovelebis qsovilebis dakonserveba formalinisa da eTilis spirtis<br />
gamoyenebiT.<br />
susti gacxelebisas cilis meoreuli da mesameuli struqtura irRveva, rac<br />
iwvevs misi biologiuri funqciis dakargvas. am process cilis denaturireba<br />
ewodeba (sur. 41).<br />
Zlieri gaxurebisas cilis makromolekula mTlianad iSleba da gamoiyofa<br />
aqroladi produqtebi, romelTac damwvari bumbulis suni aqvs. am TvisebiT<br />
SesaZlebelia cilis amocnoba, cilovani warmoSobis boWkos, abreSumisa da<br />
Salis gansxvaveba sxva boWkoebisagan.<br />
hidrolizi cilis umniSvnelovanesi Tvisebaa, cila hidrolizdeba amiduri<br />
bmis xarjze, susti gaxurebisas mJavas an tutis xsnaris TanxlebiT:<br />
NH 2 – CH – C – N – CH – C – N – CN – C – ...<br />
| || | | || | | ||<br />
R 1 O H R 2 O H R 3 O →<br />
HO – H HO – H HO – H<br />
→ NH2 – CH – COOH + NH2 – CH – COOH + NH2 – CH – COOH + ..<br />
| | |<br />
R1 R2 R3 cilis hidrolizis procesSi warmoiqmneba sxvadasxva aminomJava.<br />
adamianisa da cxovelis organizmSi cilis hidrolizi fermentebis gavleniT<br />
mimdinareobs, warmoqmnili aminomJavebi sisxlSi gadadis da qsovilebsa da<br />
ujredebs miewodeba, sadac gardaiqmneba cocxali organizmisaTvis saWiro<br />
cilad.<br />
cilovani nivTierebebi didi xania mecnierTa yuradRebis centrSia. cilis<br />
struqturisa da funqciebis Seswavla SesaZlebels xdis memkvidreobis meqanizmis<br />
dadgenas, sxvadasxva daavadebaTa bunebis gamovlenas.<br />
Tavdapirvelad gamoikvlies SedarebiT dabali molekuluri masis mqone<br />
cilebi _ hormonebi da fermentebi. erT-erTi pirveli cila, romlis pirveladi<br />
struqturis dadgena mecnierebma moaxerxes, iyo hormoni – insulini. es<br />
hormoni aregulirebs sisxlSi Saqris Semcvelobas. aRmoCnda, rom insulinis<br />
molekula Sedgeba ori, araerTnairi polipeptiduri jaWvisagan. erTSi 21<br />
aminomJavas naSTia, meoreSi ki – 30-is. polipeptiduri jaWvebi erTmaneTTan<br />
dakavSirebulia sulfiduri -S‑S- xidebiT (sur. 42):<br />
sur.42. sulfiduri xidebi insulinis molekulaSi<br />
139
140<br />
cilis struqturis dadgenis Semdeg SesaZlebelia misi sinTezi. insulinis<br />
sinTezi namdvilad aris organuli qimiis udidesi miRweva. dReisaTvis<br />
qimikosebi swavloben gacilebiT rTuli Sedgenilobis cilebs. ukve cnobilia<br />
daaxloebiT 2000 cilis pirveladi struqtura, rac araerTi efeqturi medikamentis<br />
Seqmnis winapirobaa.<br />
1. ra mimarTeba arsebobs cnebebs Soris: `peptidi~, `polipeptidi~, `poli amidi~,<br />
`cila~?<br />
2. SeadgineT reaqciis toloba, romelic Seesabameba cilis hidrolizs, Tu misi<br />
zogadi formulaa:<br />
–NH – CH–CO–<br />
|<br />
R<br />
axseniT, ra mniSvneloba aqvs am process adamianis organizmisaTvis.<br />
<br />
3. moifiqreT da aRwereT meTodi, romliTac martivad ganasxvavebT:<br />
– Salis Zafs bambis Zafisagan;<br />
– xelovnur abreSums bunebrivisagan.<br />
axseniT, ras efuZneba Tqvens mier gamoyenebuli meTodi.
24<br />
polimerebi<br />
polimeri maRalmolekuluri naerTia, romelsac didi molekuluri<br />
masa aqvs – ramdenime aTasidan ramdenime milionamde. polimerebis makromolekulebi<br />
Sedgeba monomeruli erTeulebis didi raodenobisagan, makromolekulis<br />
sidide ganisazRvreba polimerizaciis xarisxiT.<br />
gTavazobT eTilenis polimerizaciis sqemas:<br />
nCH2 = CH2 → (...– CH2– CH2 –...) n<br />
am sqemis magaliTze gaixseneT cnebebi: po li meri,<br />
monomeri, monomeruli erTeuli, poli me ri zaciis<br />
xarisxi. ras aRniSnavs n po limerizaciis re aqciis<br />
sqemaSi? ra gansxvavebaa monomersa da mo no merul<br />
erTeuls Soris?<br />
Cven ki SegaxsenebT polimerizaciis reaqciis ganmartebas:<br />
literaturaSi SesaZloa<br />
SegxvdeT termi nebi:<br />
`ele mentaruli rgo li~,<br />
`struq turuli er Teuli~,<br />
`mo nomeruli er-<br />
Te uli~. gax sov deT, rom<br />
samive termini er Tsa da<br />
imave cnebas aRniSnavs.<br />
polimerizaciis reaqcia aris qimiuri procesi, romlis drosac<br />
erTmaneTs uerTdeba sawyisi dabalmolekuluri nivTierebis (monomeris)<br />
molekulebi da warmoqmnis polimeris makromolekulebs.<br />
polimerizaciis reaqciaSi Ses vlis unari aqvs iseT nivTierebebs, romelTa<br />
molekulaSic ujeri bmaa. e. i. po limerizaciis unari aqvs ujer naerTebs. es<br />
SeiZleba iyos erTi da igive mo nomeris, an sxvadasxva monomeris molekulebi.<br />
Tu reaqciaSi erTi da igive monomeris molekulebi monawileobs, reaqcias<br />
po limerizaciis reaqcia ewodeba. Tu reaqciaSi ori, an meti sxvadasxva niv-<br />
Ti e rebis molekulebi monawileobs, reaqcias Tana polimerizaciis reaqcia<br />
ewodeba.<br />
magaliTad,<br />
polimerizaciis reaqcias ganekuTvneba polieTilenis, polipropilenis,<br />
po li vinilqloridis miRebis reaqciebi:<br />
Tanapolimerizaciis reaqcias miekuTvneba, magaliTad, beta dien-sti rolis<br />
kauCukis miRebis reaqcia:<br />
n CH 2 = CH – CH = CH 2 + nCH 2 = CH → – CH 2 – CH = CH – CH 2 – CH 2 – CH –<br />
butadieni – 1,3<br />
n CH 2 = CH – CH 2 – CH –<br />
CH 3<br />
CH 3<br />
propileni polipropileni<br />
C 6H 5<br />
n<br />
C 6H 5<br />
stiroli butadien-stirolis kauCuki<br />
141
142<br />
axla ganvixiloT polikondensaciis reaqcia.<br />
polikondensaciis reaqcia qimiuri procesia, romlis drosac<br />
sawyisi mono merebis molekulaTa SeerTebisas polimeris makromolekulebad<br />
war mo iqmneba ara mxolod ZiriTadi pro duq ti<br />
polimeri, aramed Tanauri produqtic.<br />
Tanauri produqti aris dabalmolekuluri nivTiereba, ufro<br />
xSirad wya li.<br />
polikondensaciis reaqciaSi Sedis iseTi nivTierebebi, romelTa molekule<br />
bic funqciur jgufs Seicavs.<br />
rogorc polimerizaciis reaqciis SemTxvevaSi, poli kon densaciis<br />
reaqciac SesaZloa iyos homopolikondensaciis da Tanapolikondensaciis.<br />
homopolikondensaciis reaqciaSi monawileobs erTi da igive monomeris<br />
molekulebi, magaliTad, monosaqaridis glukozis molekulebisagan mce nareTa<br />
ujredebSi warmoiqmneba polisaqaridebi:<br />
nC 6H 12O 6 → (–C 6H 10O 5 – ) n + nH 2O<br />
glukoza polisaqaridi<br />
(saxamebeli, celuloza)<br />
Tanapolikondensaciis reaqciaSi polimeri warmoiqmneba ori an meti<br />
sawyisi monomeris molekulebisagan. magaliTad, fenol-formaldehiduri<br />
fisis miReba:<br />
nC 6H 5 – OH + n H – C<br />
O<br />
H<br />
OH<br />
CH2 –<br />
n<br />
+ n H 2O<br />
fenoli formaldehidi fenolformaldehiduri<br />
fisi<br />
polimeris Tviseba damokidebulia mis Sedgenilobaze, molekulur<br />
masasa da agebulebaze.<br />
polimeri, romlis SedgenilobaSic Sedis wyalbadi da naxSirbadi,<br />
iwvis. aseTia magaliTad, polieTileni da polipropileni, polimeris<br />
mikromolekulis monomerul erTeulSi halogenis gamoCena polimers<br />
axal Tvisebas sZens – is aRar iwvis, aseTia magaliTad, polivinilqloridi,<br />
fToroplasti.<br />
polimeris molekuluri masa mis agregatul mdgomareobas gansazRvravs:<br />
Tu polimerizaciis xarisxi arc ise maRalia, miiReba sqeli Txevadi masa,<br />
xolo monomeruli erTeulebis raodenobis gazrdis Sedegad miiReba myari<br />
produqti.<br />
molekuluri masis maRali mniSvneloba uzrunvelyofs polimeris araaqro<br />
ladobas, gansazRvravs lRobisa da daSlis temperaturul intervalebs.<br />
wrfivi agebulebis polimerebs didi molekuluri masa aqvs, Sesabamisad<br />
maTi molekulebi grZelia, rac maT moqnilobas ganapirobebs, polimeris
jaWvis moqniloba mniSvnelovnad<br />
izrdeba naxSirbadisa da<br />
polimeris Semadgeneli sxva<br />
atomebis Sinagani brunviT<br />
martivi bmis irgvliv. amis gamo<br />
SesaZlebelia mikro molekulis<br />
erTi nawilis Semobruneba meoris<br />
mimarT, es ki Tavis mxriv polimeris<br />
makromolekulis Zlieri<br />
gaRunvis da gorglebad daxvevis<br />
SesaZleblobas qmnis.<br />
wrfivi agebulebis myar poli<br />
merSi makromolekulebi<br />
erT ma neTTan ax lo saa ganla gebuli.<br />
amis gamo aRi Zvre ba mole<br />
ku laTaOSorisi ur Ti erT qmedebis<br />
Za lebi. mole ku laTa Sorisi<br />
Zalebi Za lian sustia, Cveulebriv<br />
qi miur bmaze 10-15-jer<br />
sus ti, magram vinaidan isini<br />
xor ci eldeba makromolekulis<br />
mTel sigr Zeze uzarmazari<br />
raodenobis monomeruli erTeulebis<br />
monawileobiT, mo le kulaTaSorisi<br />
Zalebis jamuri simtki<br />
ce mniSvnelovnad izrdeba.<br />
amiT aixsneba is, rom polimeri<br />
didi meqanikuri simtkiciT xa siaTdeba,<br />
organul gamxsnelSi ki<br />
blant xsnars warmoqmnis.<br />
polimerebi, romlebic wrfivi<br />
agebulebis makro mo le kulebisagan<br />
Sedgeba, gacxe lebisas<br />
jer rbildeba da mxolod<br />
Semdeg iwyebs lRo bas, dabalmolekuluri<br />
nivTiere bebi<br />
sagan gansxvavebiT polimers<br />
ar aqvs darbilebisa da lRobis<br />
fiq sirebuli temperatura. es<br />
imi tom, rom polimeris Sedge nilobaSi<br />
sxvadasxva zomis makromo<br />
lekulebi Sedis.<br />
wrfiv molekulebs SesaZlebelia<br />
hqondes rogorc kristaluri, ise amorfuli<br />
struqtura, polimeris kristalurobaSi<br />
igulisxmeba makromolekulebis an maTi<br />
nawilebis mowesrigebuli, paraleluri<br />
ganlageba. amorful mdgomareobaSi<br />
makromolekulebs mouwesrigebeli<br />
urTierTganlageba aqvs. ganStoebuli da<br />
sivrculi agebulebis polimerebi, rogorc<br />
wesi, amorfulia.<br />
rodesac kristalurobaze vlaparakobT,<br />
unda vicodeT, rom mxolod idealur<br />
SemTxvevaSia SesaZlebeli molekulebis<br />
erTmaneTis mimarT Nparaleluri ganlageba,<br />
sinamdvileSi makromolekulebis zogierTi<br />
monakveTi erTmaneTis mimarT ufro<br />
mowesrigebulad aris ganlagebuli,<br />
zogierTi ki – naklebad, erTi da igive<br />
makromolekulebi ganlagebulia rogorc<br />
kristalur ise amorful ubnebSi (sur. 43).<br />
erTi da igive polimeris kristalurobis<br />
xasiaTi SesaZloa Seicvalos. magaliTad,<br />
polimeris gaWimva xels uwyobs molekulebis<br />
paralelur ganlagebas, ris Sedegadac<br />
kristaluroba izrdeba.<br />
(2)<br />
sur. 43. polimeris amorfuli da<br />
kristaluri agebuleba<br />
(1) kristaluri agebulebis ubani<br />
(2) amorfuli agebulebis ubani.<br />
ganStoebuli, datotvili makromolekulebisagan agebul polimerebSi<br />
makromolekulebi erTmaneTisagan dacilebulia, molekulaTaSorisi Zalebi<br />
moqmedebs mxolod polimeris calkeul ubnebze. Sesabamisad, daqveiTebulia<br />
meqanikuri simtkice, lRobis temperatura. sivrciTi struqturis polimerebi<br />
meqanikurad mtkicea, ar ixsneba organul gamxsnelSi da ar lRveba – aseTi<br />
polimeri gaxurebisas iSleba.<br />
(1)<br />
143
144<br />
SegaxsenebT, rom polimeris Tvisebebze did gavlenas axdens regularoba,<br />
rac mJRavndeba polimeris makromolekulis warmomqmneli sawyisi monomeris<br />
molekulebis mkacrad gansazRvruli TanmimdevrobiT SeerTebaSi.<br />
polimerebs, romelTa makromolekulebi agebulia erTnairi sivrculi<br />
konfi gu raciis mqone elementaruli rgolebisagan, stereoregularuli<br />
ewodeba, po li meri stereoregularulia maSinac, rodesac mis makro mo lekulaSi<br />
gan sxva vebuli sivrculi konfiguraciis mqone monomeruli er Teulebia,<br />
magram maTi monacvleoba jaWvSi xasiaTdeba garkveuli Tan mimdevrobiT.<br />
polimers, romelSic sxvadasxva konfiguraciis monomeruli erTeulebi<br />
nebismieradaa ganlagebuli, arastereoregularuli ewodeba.<br />
stereoregularobas udidesi mniSvneloba aqvs, rogorc ukve iciT<br />
swored stereoregularuli agebulebis gamoa kauCuki elastikuri.<br />
elastikuroba ki kauCukis ganmsazRvreli Tvisebaa misi gamoyenebisas<br />
avto mo bilisa da TviTmfrinavis saburavebis dasamzadeblad, radgan<br />
avtomobilisa da safren bilikze TviTmfrinavis moZraobisas maTi saburavebi<br />
mravaljeradad deformirdeba.<br />
polimerebis makromolekulebs sxvadasxva agebuleba, sxvadasxva geo metriuli<br />
forma aqvs: wrfivi, ganStoebuli anu datotvili da sivrculi.<br />
wrfivi agebuleba aqvs magaliTad, polieTilenis, polipropilenis<br />
makro molekulebs. mravaljer ganmeorebadi elementaruli rgolebi<br />
am polimerebSi warmoqmnis zigzagisebur jaWvs. daxveuli gorglis<br />
msgavsi wrfivi agebuleba aqvs kauCukis makromolekulas. saxameblis<br />
makromolekulas datotvili agebuleba aqvs. rezins ki sivrculi, sadac<br />
makromolekulebs Soris warmoqmnilia qimiuri bmebi (sur. 44).<br />
a) b) g)<br />
sur. 44. polimeris geometriuli forma:<br />
a) wrfivi; b) ganStoebuli; g) sivrculi.
xelovnuri maRalmolekuluri naerTebi miiReba bunebrivi naerTebisagan<br />
qimiuri meTodebis gamoyenebiT ise, rom ar icvleba ZiriTadi jaWvi. erTerTi<br />
pirveli xelovnuri polimeri iyo viskoza, romelic celulozasgan<br />
miiRes. xelovnuria rezinic – romelic naturaluri kauCukis vulkanizaciis<br />
produqtia. sinTezuri maRalmolekuluri naerTebi miiReba iseTi<br />
nivTierebebis polimerizaciiT da polikondensaciiT, romlebic bunebaSi ar<br />
moipoveba da sinTezuri gziTaa miRebuli. pirveli sinTezuri polimerebi<br />
bakeliti da karboliti fenolformaldehiduri fisebia, romlebic miiReba<br />
fenolis polikondensaciiT formaldehidTan.<br />
1. SesaZlebelia Tu ara monomerul erTeulad miviCnioT polieTilenis<br />
molekulebSi – CH 2 – jgufi? pasuxi daasabuTeT.<br />
2. ras aRniSnavs n vinilqloridis polimerizaciis reaqciaSi?<br />
n CH2 = CH – CH2 – CH –<br />
Cl Cl<br />
n<br />
3. polieTilenis saSualo molekuluri masaa 28000. gamoiangariSeT misi<br />
polimerizaciis xarisxi.<br />
<br />
4. mecnierebi miiCneven, rom polimeris Termomedegobis gazrdis problema<br />
ukavSirdeba iseTi polimerebis sinTezis problemas, romelic jaWvSi ar Seicavs<br />
naxSirbadis atomebs. axseniT ras efuZneba mecnierTa aseTi mosazreba.<br />
145
146<br />
25<br />
elementorganuli polimerebi<br />
elementorganuli polimerebi es iseTiU polimerebia, romlebic ZiriTadad<br />
jaWvSi Seicavs ara naxSirbadis, aramed sxva qimiuri elementebis, magaliTad,<br />
Jangbadis, titanis, siliciumis atomebs. aseTi polimerebis gverdiT jaWvSi<br />
organuli radikalebia.<br />
ase magaliTad, 1935 wels miiRes siliciumorganuli polimeri – silikoni,<br />
romlis Sedgenilobac SeiZleba gamoisaxos ase:<br />
R R<br />
| |<br />
. . . – Si – O – Si – O – ...<br />
| |<br />
R R<br />
es polimeri xasiaTdeba maRali TermomedegobiT, kargi eleqtro sa izolacio<br />
TvisebebiT, qimiuri inertulobiT, hidrofoburia, e. i. ar sveldeba<br />
wyliT.<br />
plastmasi<br />
plastmasi ewodeba im masalas, romelic mzaddeba polimeris safuZvelze.<br />
plastmass SeuZlia gacxelebisas miiRos garkveuli forma da SeinarCunos<br />
gacivebisas.<br />
rogorc wesi plastmasi sxvadasxva nivTierebis narevia, polimeri am niv-<br />
TierebaTagan erT-erTi, Tumca yvelaze saWiro komponentia. swored polimeri<br />
akavSirebs plastmasis yvela komponents da erT mTlian, metnaklebad<br />
erTgvarovan masad gardaqmnis. amitom polimers uwodeben Semkvrels, makav-<br />
Sirebels.<br />
pirveli plastmasi miiRes bunebrivi polimerisagan – kauCukisagan, celulozas<br />
nawarmisagan da a. S. SemdegSi Semkvrelad gamoiyenes sinTezuri<br />
polimerebic – fenolformaldehiduri fisi, polieTeri da sxva.<br />
mza nakeTobis Sesaqmnelad im plastmasebis gamoyenebaa ufro mosaxerxebeli,<br />
romelTa gamyarebis da darbilebis procesi Seqcevadia. aseT polimers<br />
Termoplasti, anu Termoplastikuri polimeri ewodeba. aseTi polimeri<br />
gaxurebisas lRveba, SeuZlia miiRos gansazRvruli forma da SeinarCunos<br />
is gamyarebisas. Tu aseT polimers isev gavacxelebT, is kvlav darbildeba da<br />
saWiroebis SemTxvevaSi axal formas SeiZens, rac imas niSNnavs, rom arsebobs<br />
misi xelmeored gadamuSavebis SesaZlebloba.<br />
ase iReben, magaliTad, lavsanisa da kapronisagan boWkos, polieTilenisagan<br />
sxvadasxva daniSnulebis sagnebs, aseTi polimeria kauCukic.<br />
Tu nakeTobis formirebis procesSi xdeba polimeris makromolekulebis<br />
`Sekereba~ da polimeri gamyarebisas iZens badisebur agebulebas, maSin SeuZlebeli<br />
xdeba aseTi nivTierebis dabruneba blant da denad mdgomareobaSi<br />
gaxurebis an gaxsnis gziT. aseT plastmass reaqtoplasti, anu Termoreaqtiuli<br />
polimeri ewodeba. Termoreaqtiulia magaliTad fenoformaldepiduri<br />
fisi.<br />
Semkvreli polimeris garda plastmasSi xSirad SehyavT sxvadasxva daniSnulebis<br />
danamatebi, Semavseblebi, saRebrebi, nivTierebebi, romlebic aumjobesebs<br />
meqanikur Tvisebebs, Termomedegobas.
plastmasebi farTod gamoiyeneba samSeneblo industriaSi, manqanaTm-<br />
SeneblobaSi, eleqtroteqnikaSi, SesafuTi masalebis warmoebaSi, yoveldRiuri<br />
moxmarebis sagnebis dasamzadeblad.<br />
plastmasebis farTo gamoyenebas ganapirobebs maTi dabali fasi, gadamuSavebis<br />
SesaZlebloba da iseTi Tvisebebi, romliTac isini ar Camouvardeba<br />
metalebs, Senadnobebs, zog SemTxvevaSi ki ukeTesicaa maTze. ase magaliTad,<br />
plastmasis nakeTobebi Zalian msubuqia, mdgradia koroziis mimarT, mtkicea,<br />
gaaCnia Zalian kargi optikuri da saizolacio Tvisebebi.<br />
boWko<br />
boWko aris wrfivi agebulebis polimeri, romelic gamosadegia Zafebis,<br />
maryuJebis, safeiqro masalebis dasamzadeblad.<br />
bunebrivi boWko warmoSobis mixedviT aris:<br />
• mcenareuli, rogoricaa bamba, seli, qereli da sxva;<br />
• cxoveluri, rogoricaa matyli da abreSumi;<br />
• mineraluri, rogoricaa asbesti.<br />
qimiuri boWko miiReba boWkos warmomqmneli polimeris xsnaris an nal-<br />
Robisagan. qimiuri boWko SesaZloa iyos xelovnuri da sinTezuri.<br />
xelovnuri boWko miiReba bunebrivi polimerisagan, an maTi gadamuSavebis<br />
produqtisagan. ZiriTadad es aris celuloza da misi eTerebi. xelovnuria,<br />
magaliTad viskozuri da acetaturi boWko.<br />
sinTezuri boWko miiReba sinTezuri polimerisagan. aseTia magaliTad<br />
kaproni, lavsani, nailoni da sxva.<br />
1. boWkos misaRebad iyeneben aminomJavebs, magaliTad ε-aminokapronmJavas,<br />
Tumca iyeneben mxolod normaluri agebulebis aminomJavebs da ara maT izomerebs,<br />
magaliTad<br />
NH 2 – CH 2 – CH – CH 2 – CH 2 – COOH<br />
|<br />
CH 3<br />
axseniT, ra aris amis mizezi.<br />
2. praqtikaSi farTod gamoiyeneba boWko nitroni, romelic garegnulad gavs<br />
Sals. is kargad inaxavs siTbos, gamoirCeva didi simtkiciT, elastikuria, Termomedegia.<br />
nitroni, yvela sxva bunebrivsa da qimiur boWkosTan SedarebiT, mdgradia<br />
sinaTlisa da atmosferuli zemoqmedebis mimarT. misi monomeria akrilis mJavas<br />
nitrili, anu akrilonitrili CH 2 = CH – CN. SeadgineT nitrilis polimerizaciis<br />
reaqciis toloba.<br />
gamoyaviT monomeruli erTeuli.<br />
3. polipropilenis boWko gamoirCeva simsubuqiT. is wyalze msubuqia, gamoirCeva<br />
agreTve didi simtkiciT, tuteebisa da mJavebis mimarT mdgradobiT. gamoiyeneba<br />
sazRvao bagirebis, xaliCebis, TevzsaWeri badeebis, trikotaJis nawarmis dasamzadeblad<br />
da sxva. SeadgineT propilenis polimerizaciis reaqciis toloba.<br />
147
148<br />
4. Tu kauCuks 30%-ze met gogirds daamateben, miiReba xisti masala – eboniti,<br />
romelic plastmasebis miRebamde erT-erT saukeTeso izolatorad miiCneoda. rogor<br />
axsniT im faqts, rom eboniti aRar aris elastikuri?<br />
5. 45-e suraTze Tqven xedavT mowyobilobas,<br />
romlis saSualebiTac mimdinareobs<br />
kauCukis daSlis reaqcia. gaauferulebs Tu<br />
ara kauCukis daSlis produqti bromian wyals?<br />
daasabuTeT Tqveni varaudi.<br />
sur. 45. kauCukis daSla<br />
<br />
6. TqvenTvis kargadaa nacnobi rezinis nakeTobani. imisaTvis, rom kauCukis<br />
Tvisebebs gaecnoT, gaaSreT rezinis webo, romelic kauCukisa da benzinis narevia.<br />
SeadareT rezinisa da kauCukis Tvisebebi, gamoikvlieT, ra emarTeba maT gaTbobisas.
26<br />
biopolimerebi<br />
biopolimerebs ganekuTvneba TqvenTvis kargad nacnobi cilebi, polisaqaridebi<br />
da nukleinis mJavebi.<br />
cila iseTi biopolimeria, romelic α-aminomJavebis naSTebisagan Sedgeba.<br />
cilaSi gamoyofen oTxi donis struqturas. cilis pirveladi struqtura<br />
wrfivi struqturaa, is ganisazRvreba polipeptidur jaWvSi aminomJavebis<br />
molekulaTa naSTebis monacvleobiT. cilis pirveladi struqtura ganapirobebs<br />
dedamiwaze yvela cocxali organizmis cilovan individualobas.<br />
sul raRac 20 α-aminomJavisagan buneba qmnis cilaTa mravalferovnebas. iseve<br />
rogorc TiTis anabeWdi, yovel cocxal organizmSi cilaTa ganumeorebeli,<br />
mxo lod misTvis damaxasiaTebeli gansakuTrebuli nakrebia. organizmisaTvis<br />
`ucxo~ cilis nakrebis, magaliTad, mikrobebis miuReblobazea dafuZnebuli<br />
organizmis damcavi reaqcia – imuniteti.<br />
cilis meoreuli struqtura, romelic xSirad spiraliseburia, ganisaz-<br />
Rvreba cilis molekulis polipeptiduri jaWvis spiralad daxvevis TaviseburebiT.<br />
es daxveva xdeba C = O da – NH– jgufebs Soris wyalbaduri bmebis<br />
warmoqmnis xarjze.<br />
cilis mesameuli struqtura,<br />
romelic gorglismagvaria, ganisazRvreba<br />
cilovani spiralis<br />
sivrciTi mdebareobiT (sur. 46). am<br />
sivrciT mdebareobas ganapirobebs<br />
wyalbaduri, amiduri da disulfiduri<br />
bmebis warmoqmna. gansaz-<br />
Rvruli sivrciTi konfiguraciis<br />
mqone cilis mesameuli struqtura,<br />
SverilebiTa da RrmulebiT, gareT<br />
mimarTuli funqciuri jgufebiT<br />
ganapirobebs cilis molekulis<br />
specifikur biologiur aqtivobas. sur. 46. cilis mesameuli struqtura<br />
zogierT cilas, magaliTad hemoglobins,<br />
meoTxeuli struqtura aqvs (sur. 47). meoTxeuli struqtura<br />
warmoiqmneba im SemTxvevaSi, rodesac cilis makromolekulis SedgenilobaSi<br />
ramdenime polipeptiduri jaWvi Sedis. es struqtura ganisazRvreba im<br />
polipeptiduri jaWvebis sivrculi ganlagebiT, romlebic ar aris erTmaneTTan<br />
dakavSirebuli kovalenturi bmebiT.<br />
polisaqaridi iseTi biopolimerebia,<br />
romlebic Sedgeba monosaqaridis<br />
naSTebisagan.<br />
polisaqaridia saxamebeli da<br />
celuloza. saxameblisa da celulozas<br />
magaliTiT Cven kidev<br />
erTxel vrwmundebiT, Tu ra didi<br />
gavlena aqvs nivTierebis Tvisebebze<br />
sivrcul agebulebas. erTi<br />
sur. 47. cilis meoTxeuli struqtura<br />
da igive molekuluri formulis<br />
mqone (C6H10O5) n saxameblisa da ce-<br />
149
150<br />
lulozas Tvisebebs Soris marTlac TvalsaCino gansxvavebaa – saxamebeli<br />
sakvebi nivTierebaa, naxSirwyalia, romelsac mcenare ujredSi imaragebs.<br />
saxamebeli agebulia α-glukozis molekulis naSTebisagan.<br />
CH 2OH<br />
5 O<br />
H<br />
4<br />
H<br />
6<br />
H<br />
1<br />
OH OH<br />
3<br />
H<br />
2<br />
OH<br />
H<br />
OH<br />
glukozis α-forma<br />
CH 2OH CH 2OH CH 2OH<br />
O O O<br />
H H H H H H H H<br />
H<br />
... O O O ...<br />
H<br />
4<br />
OH<br />
OH H OH H OH H<br />
H OH H OH H OH<br />
6<br />
CH2OH 5<br />
H<br />
OH<br />
3<br />
H<br />
H<br />
2<br />
OH<br />
saxameblis makromolekulis naSTebi<br />
celuloza mcenareuli ujredis damatebiTi meqanikuri garsia, is agebulia<br />
β-glukozis molekulis naSTebisagan.<br />
O<br />
OH<br />
1<br />
H<br />
glukozis β-forma<br />
CH 2OH CH 2OH CH 2OH<br />
O O O<br />
H H H H H H<br />
... O O O ...<br />
OH H H OH H H OH H H<br />
H OH H OH H OH<br />
celulozas makromolekulis naSTebi.
polinukleotidi, anu nukleinis mJavebi<br />
nukleinis mJavebi iseTi biopolimerebia, romlebic nukleotidebisagan<br />
Sedgeba.<br />
nukleinis mJavebi xasiaTdeba maT makromolekulaSi sul oTxi saxis nukleotidis<br />
monacvleobiT. aqedan sami nukleotidi Sedis nebismieri nukleinis<br />
mJavas SedgenilobaSi. es nukleotidebia: adenini (a), guanini (g), citozini<br />
(c), meoTxe nukleotidi rnm-isaTvis aris uracili (u), xolo dnm-isaTvis Timini<br />
(T).<br />
dnm-is makromolekula spiralia, romelic<br />
saerTo RerZze daxveuli ori jaWvisagan Sedgeba<br />
(sur. 48). es dnm-is meoreuli struqturaa. iseve<br />
rogorc cilis mo le kulaSi, nukleinis mJavas<br />
SemTxvevaSic meoreuli struqtura wyalbaduri<br />
bmebis safuZvelze yalibdeba. Tavis<br />
mxriv wyal baduri bmebi myardeba makromolekulis<br />
sxvadasxva jaWvis azotovan fuZeebs Soris,<br />
romlebic cilis molekulebis Semadgeneli radikalebisagan<br />
gansxvavebiT mimarTulia ara spiralis<br />
gareT, aramed SigniT.<br />
nukleinis mJavebi rnm da dnm umniSvne lovanes<br />
rols asrulebs organizmis memkvidreobiTi<br />
informaciis Senaxvisa da gadacemis procesSi,<br />
cilis biosinTezSi.<br />
= a...T =<br />
a – adenini = a...T =<br />
g – guanini<br />
T – Timini<br />
c – citozini = c...g =<br />
= a...T =<br />
= T...a =<br />
= g...c =<br />
= c...g =<br />
= c...g =<br />
= a...T =<br />
= T...a =<br />
= g...c =<br />
= T...a =<br />
= c...g =<br />
sur. 48. dnm-is ormagi<br />
spiralis agebuleba<br />
151
152<br />
1. gaecaniT Semajamebel sqemas `polimerebis klasifikacia~. SeadgineT mcire<br />
moTxroba sqemis mixedviT.<br />
stereoregularuli<br />
stereoregula-<br />
robis mixedviT<br />
polimeri<br />
arastereore<br />
gu laruli<br />
miRebis xerxis mixedviT<br />
bunebrivi<br />
warmoSobis<br />
mixedviT<br />
polikondensaciis<br />
polimerizaciis<br />
xelovnuri<br />
sinTezuri<br />
wrfivi<br />
organuli<br />
ZiriTadi jaWvis Sedge-<br />
nilobis mixedviT<br />
makromolekulis for-<br />
mis mixedviT<br />
gaxurebis mimarT<br />
damokidebulebiT<br />
araorganuli<br />
sivrculi<br />
element<br />
organuli<br />
ganStoebuli<br />
Termoplastikuri<br />
Termoreaqtiuli
SeamowmeT Tqveni codna<br />
1 erT-erT laboratoriaSi Seamowmes Zexvis nimuSSi saxameblis Semcveloba,<br />
risTvisac gamoiyenes kaliumis iodidis xsnari, radganac Seferiloba<br />
ar Seicvala, `eqspertma~ daaskvna, rom Zexvis nimuSSi saxamebeli ar aris.<br />
ras fiqrobT amis Sesaxeb Tqven? ras urCevdiT eqsperts?<br />
2 sqemaze mocemulia 1 t mSrali<br />
xis naxerxis gadamuSavebiT miRebuli<br />
masalebis saxeoba da raodenoba.<br />
_ SeiZleba Tu ara celulozas gadamuSavebis<br />
process vuwodoT unarCeno<br />
warmoeba?<br />
daasabuTeT Tqveni mosazreba.<br />
Tu Tqveni pasuxi dadebiTia, axseniT<br />
ratom ar utoldeba produqtebis masaTa<br />
jami nedleulis masas?<br />
sur. 49<br />
3 suraTze Tqven xedavT diagramas, romelic asaxavs adamianis organizmis<br />
qimiur Sedgenilobas. gaiTvaliswineT Tqveni organizmis masa da daadgineT<br />
qimiuri Sedgeniloba.<br />
wyali 65%<br />
cximebi 10%<br />
cilebi 18%<br />
naxSirwylebi 5%<br />
sxva organuli da<br />
araorganuli<br />
nivTierebebi 2%<br />
153
154<br />
4 suraTze gamosaxuli cilis struqturebs Soris, romelia pirveladi,<br />
meoreuli da mesameuli? daasabuTeT Tqveni varaudi.<br />
a) b)<br />
g)