NCI National Clinical Trials Network (NCTN) Program Guidelines
NCI National Clinical Trials Network (NCTN) Program Guidelines
NCI National Clinical Trials Network (NCTN) Program Guidelines
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
PART 4: Appendices Section V – Important Definitions, Abbreviations, & NIH/<strong>NCI</strong> URLs<br />
V. Other Important <strong>NCI</strong>/NIH URLs, Federal Citations, and List of Abbreviations<br />
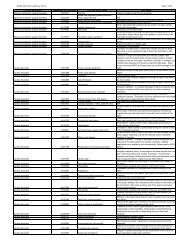
A listing of important URLs (links to websites) and abbreviations referenced in the text of these <strong>Guidelines</strong><br />
is provided below.<br />
A. Website URLs referenced in these <strong>Guidelines</strong><br />
<strong>NCI</strong> Website<br />
http://www.cancer.gov/<br />
<strong>NCI</strong> Biomarker, Imaging, and Quality of Life Studies Funding <strong>Program</strong> (BIQSFP)<br />
http://biqsfp.cancer.gov/<br />
<strong>NCI</strong> Cancer <strong>Trials</strong> Support Unit (CTSU) Website<br />
http://www.ctsu.org<br />
<strong>NCI</strong> Cancer Diagnosis <strong>Program</strong>’s Request for an Application (RFA) on Support for Human Specimen<br />
Banking in <strong>NCI</strong>-Supported Cancer <strong>Clinical</strong> <strong>Trials</strong><br />
http://grants.nih.gov/grants/guide/rfa-files/RFA-CA-05-017.html<br />
<strong>NCI</strong> Cancer Diagnosis <strong>Program</strong>’s Website<br />
http://cdp.cancer.gov/<br />
<strong>NCI</strong> Center for Coordinating <strong>Clinical</strong> <strong>Trials</strong><br />
http://ccct.cancer.gov/about/overview<br />
<strong>NCI</strong> Central IRB Website<br />
http://www.ncicirb.org<br />
<strong>NCI</strong> <strong>Clinical</strong> <strong>Trials</strong> and Translational Research Advisory Committee (CTAC)<br />
http://ccct.cancer.gov/committees/ctac<br />
<strong>NCI</strong> <strong>Clinical</strong> and Translational Research Operations Committee<br />
http://ccct.cancer.gov/committees/ctroc<br />
<strong>NCI</strong> CTWG Steering Committee System (Information on <strong>NCI</strong> Scientific Steering Committees)<br />
http://transformingtrials.cancer.gov/steering/overview<br />
<strong>NCI</strong> <strong>Clinical</strong> <strong>Trials</strong> Reporting <strong>Program</strong> (CTRP)<br />
http://www.cancer.gov/clinicaltrials/conducting/ncictrp/main/allpages<br />
<strong>NCI</strong> Guide to Readers to Information on Other <strong>NCI</strong> Divisions/Branches<br />
http://www.cancer.gov/aboutnci<br />
Diagnostics Evaluation Branch (DRB) of the Cancer Diagnosis <strong>Program</strong> (CDP) <strong>Program</strong> for the<br />
Assessment of <strong>Clinical</strong> Cancer Tests (PACCT) – <strong>Clinical</strong> Tumor Marker Study <strong>Guidelines</strong><br />
http://www.cancerdiagnosis.nci.nih.gov/diagnostics/advice/guidelines.htm<br />
Good <strong>Clinical</strong> Practice in FDA-Regulated <strong>Clinical</strong> <strong>Trials</strong><br />
http://www.fda.gov/oc/gcp/default.htm<br />
Page 216 of 241