K Nn mK - Amecamex.org.mx
K Nn mK - Amecamex.org.mx
K Nn mK - Amecamex.org.mx
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
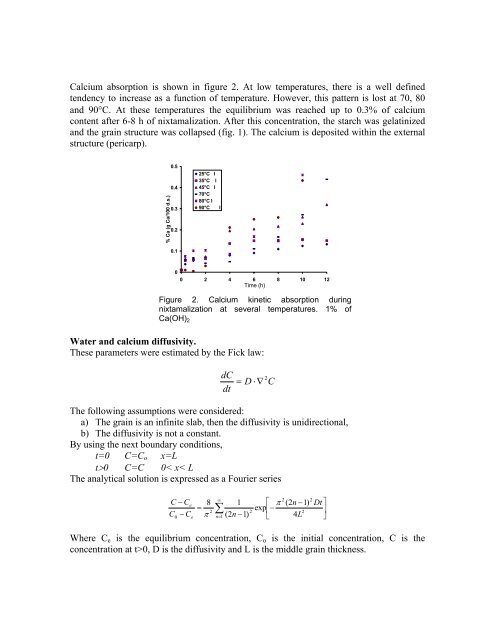
Calcium absorption is shown in figure 2. At low temperatures, there is a well defined<br />
tendency to increase as a function of temperature. However, this pattern is lost at 70, 80<br />
and 90°C. At these temperatures the equilibrium was reached up to 0.3% of calcium<br />
content after 6-8 h of nixtamalization. After this concentration, the starch was gelatinized<br />
and the grain structure was collapsed (fig. 1). The calcium is deposited within the external<br />
structure (pericarp).<br />
% Ca (g Ca/100 d.s.)<br />
0.5<br />
0.4<br />
0.3<br />
0.2<br />
0.1<br />
0<br />
25°C I<br />
35°C I<br />
45°C I<br />
70°C<br />
80°C I<br />
90°C I<br />
0 2 4 6 8 10 12<br />
Time (h)<br />
Figure 2. Calcium kinetic absorption during<br />
nixtamalization at several temperatures. 1% of<br />
Ca(OH)2<br />
Water and calcium diffusivity.<br />
These parameters were estimated by the Fick law:<br />
dC 2<br />
dt<br />
= D ⋅∇<br />
The following assumptions were considered:<br />
a) The grain is an infinite slab, then the diffusivity is unidirectional,<br />
b) The diffusivity is not a constant.<br />
By using the next boundary conditions,<br />
t=0 C=Co x=L<br />
t>0 C=C 0< x< L<br />
The analytical solution is expressed as a Fourier series<br />
∑ ∞<br />
2<br />
C − Ce<br />
8 1 ⎡ π ( 2n<br />
−1)<br />
=<br />
exp⎢−<br />
2<br />
2<br />
2<br />
C0<br />
− Ce<br />
π n=<br />
1 ( 2n<br />
−1)<br />
⎣ 4L<br />
Where Ce is the equilibrium concentration, Co is the initial concentration, C is the<br />
concentration at t>0, D is the diffusivity and L is the middle grain thickness.<br />
C<br />
2<br />
Dt ⎤<br />
⎥<br />
⎦