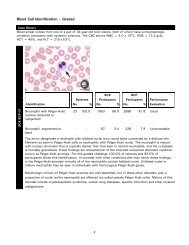
Urine Albumin Creatinine Ratio
Urine Albumin Creatinine Ratio
Urine Albumin Creatinine Ratio
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
<strong>Urine</strong> <strong>Albumin</strong> <strong>Creatinine</strong> <strong>Ratio</strong><br />
<strong>Urine</strong> albumin and estimated glomerular filtration rate (eGFR) are the two key markers<br />
for chronic kidney disease (CKD). The National Kidney Disease Education Program<br />
(NKDEP) recommends annual assessment of urine albumin excretion to diagnose and<br />
monitor kidney damage in patients who have type 1 diabetes for five years or more, or<br />
who have type 2 diabetes. However, more frequent monitoring may be needed in patients<br />
with changing clinical status or after therapeutic interventions. (1)<br />
<strong>Urine</strong> dipstick protein methods are not sensitive enough to detect microalbuminuria. (2)<br />
Lack of sensitivity means that a negative dipstick for albumin or protein does not rule out<br />
pathologic albuminuria or proteinuria. All forms of pathologic proteinuria are likely to<br />
begin with albuminuria, as albumin is, simultaneously, among the smallest and most<br />
prevalent proteins. A spot urine can be used, however, to compensate for variations in<br />
urine concentration, it is useful to compare the amount of albumin in the sample against<br />
its concentration of creatinine. This comparison is called the urine albumin to creatinine<br />
ratio (UACR).<br />
UACR is a ratio between two measured substances. Both the urine albumn (mg/dl) and<br />
the urine creatinine (g/dl) are measured values. UACR estimates 24-hour urine albumin<br />
excretion. UACR is reported in mg/g and approximates the albumin excretion in mg/day.<br />
Unlike a dipstick test for albumin, UACR is unaffected by variations in urine<br />
concentration. The UACR is measured using the first morning urine sample when<br />
possible. Twenty-four-hour collection and timed specimens are not necessary.<br />
<strong>Albumin</strong>uria is present when UACR is greater than 30 mg/g and is a marker for CKD<br />
(chronic kidney disease). <strong>Albumin</strong>uria is used to diagnose and monitor kidney disease.<br />
Changes in albuminuria may reflect positive or negative responses to therapy and risk for<br />
progression of kidney disease. A decrease in urine albumin may be associated with<br />
improved renal and cardiovascular outcomes.<br />
For the diagnosis of microalbuminuria, care must be taken when collecting samples for<br />
the urine UACR. As most people now know, one of the earliest signs of diabetic<br />
nephropathy is so-called microalbuminuria, An early morning sample is preferred. The<br />
patient should refrain from heavy exercises 24 hours before the test. A repeat test should<br />
be done 3 to 6 months after the first positive test for microalbuminuria. The UACR is<br />
inaccurate in a person with too much or too little muscle mass due to the variation in<br />
creatinine level, which is produced by the muscle. The UACR is a useful measure of<br />
renal function in diabetic renal disease.<br />
Microalbuminuria is defined as UACR ≥3.5 mg/mmol (female) or ≥2.5 mg/mmol(male),<br />
or, with both substances measured by mass, as a UACR between 30 and 300 µg<br />
albumin/mg creatinine. (2) An alternative definition of microalbuminuria is a UACR on a<br />
random urine sample of more than 30 mg (but less than 300 mg) of albumin per gram of<br />
creatinine. (2,3)
Proteinuria is defined as: UACR >30mg/mmol or albumin concentration >200mg/l.<br />
References<br />
Richard J Baltaro, MD PhD FCAP<br />
Chemistry Resource Committee<br />
1) National Kidney Disease Education Program,<br />
http://nkdep.nih.gov/professionals/chronic_kidney_disease.htm (last accessed February 5, 2012)<br />
2). American Diabetes Association: Nephropathy in diabetes. Diabetes Care 2004;27(Supplement<br />
1):S79- S83.<br />
3) Keane WE, Eknoyan G. Proteinuria, albuminuria, risk, assessment, detection, elimination<br />
(PARADE): a position paper of the National Kidney Foundation. Am J Kid Dis 1999; 33:1004-<br />
11.