Regioselectivity of the Reactions of Heteroatom-Stabilized Allyl ...
Regioselectivity of the Reactions of Heteroatom-Stabilized Allyl ...
Regioselectivity of the Reactions of Heteroatom-Stabilized Allyl ...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
676<br />
Scheme 29<br />
Scheme 30<br />
Scheme 31<br />
Scheme 32<br />
to give 179 followed by protodesilylation (Scheme<br />
30). 118<br />
<strong>Allyl</strong> sulfone 181 can form R-product 182 via an<br />
intramolecular transfer <strong>of</strong> an acyl group (Scheme<br />
31). 119<br />
The reaction <strong>of</strong> lithium and magnesium salts with<br />
acetone in <strong>the</strong> presence <strong>of</strong> a chiral diamine 183 gives<br />
R-substituted chiral sulfones 184. The enantioselectivity<br />
is improved up to a value <strong>of</strong> 50% ee by<br />
transmetalation from lithium to magnesium derivatives<br />
(Scheme 32). 120<br />
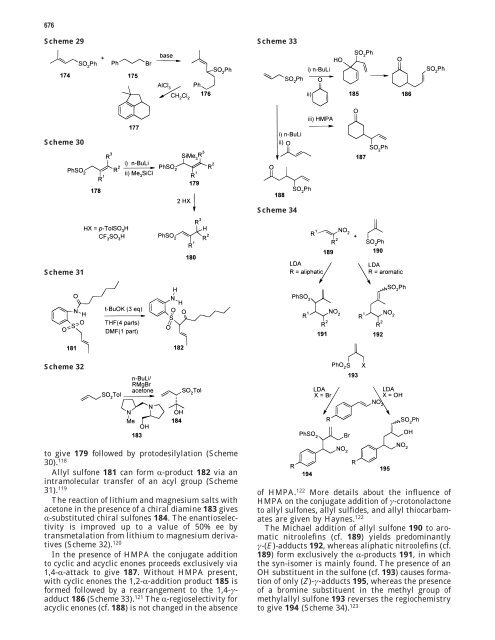
In <strong>the</strong> presence <strong>of</strong> HMPA <strong>the</strong> conjugate addition<br />
to cyclic and acyclic enones proceeds exclusively via<br />
1,4-R-attack to give 187. Without HMPA present,<br />
with cyclic enones <strong>the</strong> 1,2-R-addition product 185 is<br />
formed followed by a rearrangement to <strong>the</strong> 1,4-γadduct<br />
186 (Scheme 33). 121 The R-regioselectivity for<br />
acyclic enones (cf. 188) is not changed in <strong>the</strong> absence<br />
Scheme 33<br />
Scheme 34<br />
<strong>of</strong> HMPA. 122 More details about <strong>the</strong> influence <strong>of</strong><br />
HMPA on <strong>the</strong> conjugate addition <strong>of</strong> γ-crotonolactone<br />
to allyl sulfones, allyl sulfides, and allyl thiocarbamates<br />
are given by Haynes. 122<br />
The Michael addition <strong>of</strong> allyl sulfone 190 to aromatic<br />
nitroolefins (cf. 189) yields predominantly<br />
γ-(E)-adducts 192, whereas aliphatic nitroolefins (cf.<br />
189) form exclusively <strong>the</strong> R-products 191, in which<br />
<strong>the</strong> syn-isomer is mainly found. The presence <strong>of</strong> an<br />
OH substituent in <strong>the</strong> sulfone (cf. 193) causes formation<br />
<strong>of</strong> only (Z)-γ-adducts 195, whereas <strong>the</strong> presence<br />
<strong>of</strong> a bromine substituent in <strong>the</strong> methyl group <strong>of</strong><br />
methylallyl sulfone 193 reverses <strong>the</strong> regiochemistry<br />
to give 194 (Scheme 34). 123


![and 1-Amido-3-alkylimidazo[1,5-a]pyridines - Ark.chem.ufl.edu ...](https://img.yumpu.com/19437237/1/190x245/and-1-amido-3-alkylimidazo15-apyridines-arkchemufledu-.jpg?quality=85)











