- Page 1 and 2: Multicomponent Reactions. Edited by
- Page 3 and 4: Multicomponent Reactions Edited by
- Page 5 and 6: Contents Preface xiii Contributors
- Page 7 and 8: 2.3.4.2 Bycyclic Systems by Ugi-4CR
- Page 9 and 10: 8.3.1.5 Palladium-mediated Reaction
- Page 11 and 12: 12.5.1 Cyclic Ethers 364 12.5.2 Lac
- Page 13 and 14: XIV Preface entities, and the avail
- Page 15 and 16: XVI List of Contributors Stefano Ma
- Page 17 and 18: 2 1 Asymmetric Isocyanide-based MCR
- Page 19 and 20: 4 1 Asymmetric Isocyanide-based MCR
- Page 21 and 22: 6 1 Asymmetric Isocyanide-based MCR
- Page 23 and 24: 8 1 Asymmetric Isocyanide-based MCR
- Page 25 and 26: 10 1 Asymmetric Isocyanide-based MC
- Page 27 and 28: 12 1 Asymmetric Isocyanide-based MC
- Page 29 and 30: 14 1 Asymmetric Isocyanide-based MC
- Page 31 and 32: 16 1 Asymmetric Isocyanide-based MC
- Page 33 and 34: 18 1 Asymmetric Isocyanide-based MC
- Page 35 and 36: 20 1 Asymmetric Isocyanide-based MC
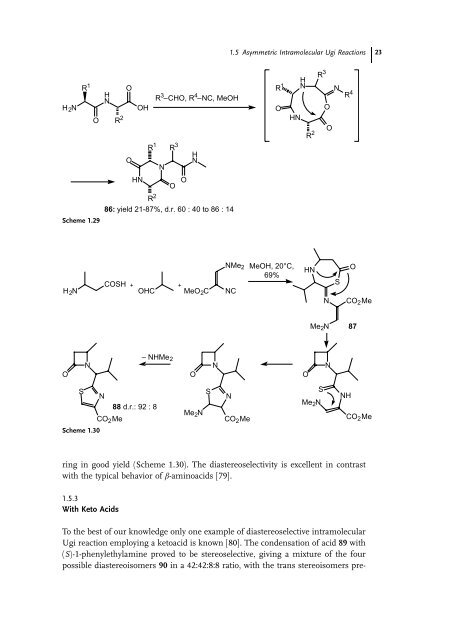
- Page 37: 22 1 Asymmetric Isocyanide-based MC
- Page 41 and 42: 26 1 Asymmetric Isocyanide-based MC
- Page 43 and 44: 28 1 Asymmetric Isocyanide-based MC
- Page 45 and 46: 30 1 Asymmetric Isocyanide-based MC
- Page 47 and 48: 32 1 Asymmetric Isocyanide-based MC
- Page 49 and 50: 34 2 Post-condensation Modification
- Page 51 and 52: 36 2 Post-condensation Modification
- Page 53 and 54: 38 2 Post-condensation Modification
- Page 55 and 56: Ph R1 R N H 2 Boc MeO 2C NC MeO 2C
- Page 57 and 58: 42 2 Post-condensation Modification
- Page 59 and 60: 44 2 Post-condensation Modification
- Page 61 and 62: 46 2 Post-condensation Modification
- Page 63 and 64: 48 2 Post-condensation Modification
- Page 65 and 66: 50 2 Post-condensation Modification
- Page 67 and 68: 52 2 Post-condensation Modification
- Page 69 and 70: 54 2 Post-condensation Modification
- Page 71 and 72: 56 2 Post-condensation Modification
- Page 73 and 74: 58 2 Post-condensation Modification
- Page 75 and 76: 60 2 Post-condensation Modification
- Page 77 and 78: 62 2 Post-condensation Modification
- Page 79 and 80: 64 2 Post-condensation Modification
- Page 81 and 82: 66 2 Post-condensation Modification
- Page 83 and 84: 68 2 Post-condensation Modification
- Page 85 and 86: 70 2 Post-condensation Modification
- Page 87 and 88: 72 2 Post-condensation Modification
- Page 89 and 90:
74 2 Post-condensation Modification
- Page 91 and 92:
76 3 The Discovery of New Isocyanid
- Page 93 and 94:
78 3 The Discovery of New Isocyanid
- Page 95 and 96:
80 3 The Discovery of New Isocyanid
- Page 97 and 98:
82 3 The Discovery of New Isocyanid
- Page 99 and 100:
HOOC 84 3 The Discovery of New Isoc
- Page 101 and 102:
86 3 The Discovery of New Isocyanid
- Page 103 and 104:
COOMe COOMe 88 3 The Discovery of N
- Page 105 and 106:
MeOOC NC MeOOC N H N N + NH 2 S 90
- Page 107 and 108:
92 3 The Discovery of New Isocyanid
- Page 109 and 110:
94 3 The Discovery of New Isocyanid
- Page 111 and 112:
96 4 The Biginelli Reaction the syn
- Page 113 and 114:
98 4 The Biginelli Reaction least 1
- Page 115 and 116:
Me Fe 100 4 The Biginelli Reaction
- Page 117 and 118:
102 4 The Biginelli Reaction Lewis
- Page 119 and 120:
104 4 The Biginelli Reaction One ot
- Page 121 and 122:
HO HO H EtO2C Me EtO 2C 106 4 The B
- Page 123 and 124:
108 4 The Biginelli Reaction O O Ph
- Page 125 and 126:
110 4 The Biginelli Reaction i-PrO
- Page 127 and 128:
112 4 The Biginelli Reaction i-PrO
- Page 129 and 130:
114 4 The Biginelli Reaction 4.9 Co
- Page 131 and 132:
116 4 The Biginelli Reaction 65 Y.
- Page 133 and 134:
118 4 The Biginelli Reaction 133 M.
- Page 135 and 136:
120 4 The Biginelli Reaction 196 D.
- Page 137 and 138:
122 5 The Domino-Knoevenagel-hetero
- Page 139 and 140:
124 5 The Domino-Knoevenagel-hetero
- Page 141 and 142:
126 5 The Domino-Knoevenagel-hetero
- Page 143 and 144:
D-glucose O O 128 5 The Domino-Knoe
- Page 145 and 146:
130 5 The Domino-Knoevenagel-hetero
- Page 147 and 148:
Ph 132 5 The Domino-Knoevenagel-het
- Page 149 and 150:
134 5 The Domino-Knoevenagel-hetero
- Page 151 and 152:
136 5 The Domino-Knoevenagel-hetero
- Page 153 and 154:
138 5 The Domino-Knoevenagel-hetero
- Page 155 and 156:
140 5 The Domino-Knoevenagel-hetero
- Page 157 and 158:
142 5 The Domino-Knoevenagel-hetero
- Page 159 and 160:
144 5 The Domino-Knoevenagel-hetero
- Page 161 and 162:
MeO MeO 146 5 The Domino-Knoevenage
- Page 163 and 164:
OMe H MeO rac-154 148 5 The Domino-
- Page 165 and 166:
169 150 5 The Domino-Knoevenagel-he
- Page 167 and 168:
O (MeO) 2P 183 OEt 88a X + O 183a:
- Page 169 and 170:
154 5 The Domino-Knoevenagel-hetero
- Page 171 and 172:
156 5 The Domino-Knoevenagel-hetero
- Page 173 and 174:
158 5 The Domino-Knoevenagel-hetero
- Page 175 and 176:
160 5 The Domino-Knoevenagel-hetero
- Page 177 and 178:
162 5 The Domino-Knoevenagel-hetero
- Page 179 and 180:
164 5 The Domino-Knoevenagel-hetero
- Page 181 and 182:
166 5 The Domino-Knoevenagel-hetero
- Page 183 and 184:
168 5 The Domino-Knoevenagel-hetero
- Page 185 and 186:
170 6 Free-radical-mediated Multico
- Page 187 and 188:
172 6 Free-radical-mediated Multico
- Page 189 and 190:
174 6 Free-radical-mediated Multico
- Page 191 and 192:
176 6 Free-radical-mediated Multico
- Page 193 and 194:
Cl 178 6 Free-radical-mediated Mult
- Page 195 and 196:
I 180 6 Free-radical-mediated Multi
- Page 197 and 198:
182 6 Free-radical-mediated Multico
- Page 199 and 200:
2 184 6 Free-radical-mediated Multi
- Page 201 and 202:
186 6 Free-radical-mediated Multico
- Page 203 and 204:
188 6 Free-radical-mediated Multico
- Page 205 and 206:
Me 3SiO 190 6 Free-radical-mediated
- Page 207 and 208:
192 6 Free-radical-mediated Multico
- Page 209 and 210:
EtO I O EtO (OC)5Mo O 194 6 Free-ra
- Page 211 and 212:
Ph 196 6 Free-radical-mediated Mult
- Page 213 and 214:
198 6 Free-radical-mediated Multico
- Page 215 and 216:
200 7 Multicomponent Reactions with
- Page 217 and 218:
202 7 Multicomponent Reactions with
- Page 219 and 220:
204 7 Multicomponent Reactions with
- Page 221 and 222:
206 7 Multicomponent Reactions with
- Page 223 and 224:
7.3.4 Synthesis of Iminodicarboxyli
- Page 225 and 226:
Boc 97 NH NH 2 MeO O (a) Me2C=CHCOC
- Page 227 and 228:
R4 R6 R5 R N 1 R2 B R O 5 R OR OR 6
- Page 229 and 230:
214 7 Multicomponent Reactions with
- Page 231 and 232:
Ph Finn [66] has reported that when
- Page 233 and 234:
Ph NH2 177 Ph Ph Ph O NHBoc 182, 73
- Page 235 and 236:
HO O OH 207 220 7 Multicomponent Re
- Page 237 and 238:
222 7 Multicomponent Reactions with
- Page 239 and 240:
224 8 Metal-catalyzed Multicomponen
- Page 241 and 242:
226 8 Metal-catalyzed Multicomponen
- Page 243 and 244:
228 8 Metal-catalyzed Multicomponen
- Page 245 and 246:
230 8 Metal-catalyzed Multicomponen
- Page 247 and 248:
232 8 Metal-catalyzed Multicomponen
- Page 249 and 250:
O 234 8 Metal-catalyzed Multicompon
- Page 251 and 252:
236 8 Metal-catalyzed Multicomponen
- Page 253 and 254:
238 8 Metal-catalyzed Multicomponen
- Page 255 and 256:
240 8 Metal-catalyzed Multicomponen
- Page 257 and 258:
242 8 Metal-catalyzed Multicomponen
- Page 259 and 260:
244 8 Metal-catalyzed Multicomponen
- Page 261 and 262:
246 8 Metal-catalyzed Multicomponen
- Page 263 and 264:
248 8 Metal-catalyzed Multicomponen
- Page 265 and 266:
250 8 Metal-catalyzed Multicomponen
- Page 267 and 268:
252 8 Metal-catalyzed Multicomponen
- Page 269 and 270:
254 8 Metal-catalyzed Multicomponen
- Page 271 and 272:
256 8 Metal-catalyzed Multicomponen
- Page 273 and 274:
258 8 Metal-catalyzed Multicomponen
- Page 275 and 276:
260 8 Metal-catalyzed Multicomponen
- Page 277 and 278:
262 8 Metal-catalyzed Multicomponen
- Page 279 and 280:
264 8 Metal-catalyzed Multicomponen
- Page 281 and 282:
266 8 Metal-catalyzed Multicomponen
- Page 283 and 284:
268 8 Metal-catalyzed Multicomponen
- Page 285 and 286:
270 8 Metal-catalyzed Multicomponen
- Page 287 and 288:
272 8 Metal-catalyzed Multicomponen
- Page 289 and 290:
274 8 Metal-catalyzed Multicomponen
- Page 291 and 292:
276 8 Metal-catalyzed Multicomponen
- Page 293 and 294:
278 9 Catalytic Asymmetric Multicom
- Page 295 and 296:
280 9 Catalytic Asymmetric Multicom
- Page 297 and 298:
O 2N R 1 282 9 Catalytic Asymmetric
- Page 299 and 300:
O O 284 9 Catalytic Asymmetric Mult
- Page 301 and 302:
R 1 N C N 286 9 Catalytic Asymmetri
- Page 303 and 304:
288 9 Catalytic Asymmetric Multicom
- Page 305 and 306:
O R 290 9 Catalytic Asymmetric Mult
- Page 307 and 308:
292 9 Catalytic Asymmetric Multicom
- Page 309 and 310:
294 9 Catalytic Asymmetric Multicom
- Page 311 and 312:
296 9 Catalytic Asymmetric Multicom
- Page 313 and 314:
298 9 Catalytic Asymmetric Multicom
- Page 315 and 316:
300 10 Algorithm-based Methods for
- Page 317 and 318:
302 10 Algorithm-based Methods for
- Page 319 and 320:
304 10 Algorithm-based Methods for
- Page 321 and 322:
306 10 Algorithm-based Methods for
- Page 323 and 324:
308 10 Algorithm-based Methods for
- Page 325 and 326:
310 10 Algorithm-based Methods for
- Page 327 and 328:
The most ancient classification con
- Page 329 and 330:
O O O O O N H 2 O Br OH O H 2 N OH
- Page 331 and 332:
N HO O F O HN N O F Fig. 10.2. Four
- Page 333 and 334:
In the second step, the right part
- Page 335 and 336:
and reaction time is a combinatoria
- Page 337 and 338:
342 12 Multicomponent Reactions in
- Page 339 and 340:
344 12 Multicomponent Reactions in
- Page 341 and 342:
346 12 Multicomponent Reactions in
- Page 343 and 344:
348 12 Multicomponent Reactions in
- Page 345 and 346:
350 12 Multicomponent Reactions in
- Page 347 and 348:
352 12 Multicomponent Reactions in
- Page 349 and 350:
354 12 Multicomponent Reactions in
- Page 351 and 352:
356 12 Multicomponent Reactions in
- Page 353 and 354:
358 12 Multicomponent Reactions in
- Page 355 and 356:
360 12 Multicomponent Reactions in
- Page 357 and 358:
362 12 Multicomponent Reactions in
- Page 359 and 360:
364 12 Multicomponent Reactions in
- Page 361 and 362:
366 12 Multicomponent Reactions in
- Page 363 and 364:
368 12 Multicomponent Reactions in
- Page 365 and 366:
370 12 Multicomponent Reactions in
- Page 367 and 368:
372 12 Multicomponent Reactions in
- Page 369 and 370:
374 12 Multicomponent Reactions in
- Page 371 and 372:
376 12 Multicomponent Reactions in
- Page 373 and 374:
378 12 Multicomponent Reactions in
- Page 375 and 376:
380 12 Multicomponent Reactions in
- Page 377 and 378:
382 12 Multicomponent Reactions in
- Page 379 and 380:
384 12 Multicomponent Reactions in
- Page 381 and 382:
386 12 Multicomponent Reactions in
- Page 383 and 384:
388 12 Multicomponent Reactions in
- Page 385 and 386:
390 12 Multicomponent Reactions in
- Page 387 and 388:
392 12 Multicomponent Reactions in
- Page 389 and 390:
394 12 Multicomponent Reactions in
- Page 391 and 392:
396 12 Multicomponent Reactions in
- Page 393 and 394:
398 13 The Modified Sakurai and Rel
- Page 395 and 396:
400 13 The Modified Sakurai and Rel
- Page 397 and 398:
402 13 The Modified Sakurai and Rel
- Page 399 and 400:
404 13 The Modified Sakurai and Rel
- Page 401 and 402:
406 13 The Modified Sakurai and Rel
- Page 403 and 404:
408 13 The Modified Sakurai and Rel
- Page 405 and 406:
R 410 13 The Modified Sakurai and R
- Page 407 and 408:
412 13 The Modified Sakurai and Rel
- Page 409 and 410:
414 13 The Modified Sakurai and Rel
- Page 411 and 412:
416 13 The Modified Sakurai and Rel
- Page 413 and 414:
418 13 The Modified Sakurai and Rel
- Page 415 and 416:
syn-123 anti-123 420 13 The Modifie
- Page 417 and 418:
R OH anti-126 422 13 The Modified S
- Page 419 and 420:
424 13 The Modified Sakurai and Rel
- Page 421 and 422:
426 13 The Modified Sakurai and Rel
- Page 423 and 424:
HO 428 13 The Modified Sakurai and
- Page 425 and 426:
171 OH R O 173 R H R O R 430 13 The
- Page 427 and 428:
432 13 The Modified Sakurai and Rel
- Page 429 and 430:
434 13 The Modified Sakurai and Rel
- Page 431 and 432:
436 13 The Modified Sakurai and Rel
- Page 433 and 434:
O R 200 Ph 438 13 The Modified Saku
- Page 435 and 436:
440 13 The Modified Sakurai and Rel
- Page 437 and 438:
442 13 The Modified Sakurai and Rel
- Page 439 and 440:
444 13 The Modified Sakurai and Rel
- Page 441 and 442:
446 13 The Modified Sakurai and Rel
- Page 443 and 444:
448 13 The Modified Sakurai and Rel
- Page 445 and 446:
450 13 The Modified Sakurai and Rel
- Page 447 and 448:
452 13 The Modified Sakurai and Rel
- Page 449 and 450:
454 Index allyl/allylic - alkoxide
- Page 451 and 452:
456 Index Brønsted - acids 98 - ba
- Page 453 and 454:
458 Index dialkoxydichlorotitanium
- Page 455 and 456:
460 Index furo[2,3-b]furan 358 furo
- Page 457 and 458:
462 Index 1-isocyano-1-cyclohexene
- Page 459 and 460:
464 Index neuropeptide, cyclic 331
- Page 461 and 462:
466 Index quinoline 246, 304 quinol
- Page 463:
468 Index tributyltin - enolate 183