Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
104 Chapter 4<br />
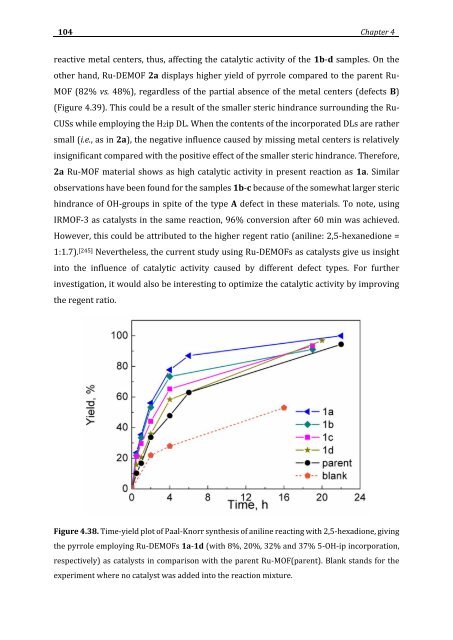
reactive metal centers, thus, affecting the catalytic activity of the 1b-d samples. On the<br />
other hand, Ru-DEMOF 2a displays higher yield of pyrrole compared to the parent Ru-<br />
MOF (82% vs. 48%), regardless of the partial absence of the metal centers (defects B)<br />
(Figure 4.39). This could be a result of the smaller steric hindrance surrounding the Ru-<br />
CUSs while employing the H2ip DL. When the contents of the incorporated DLs are rather<br />
small (i.e., as in 2a), the negative influence caused by missing metal centers is relatively<br />
insignificant compared with the positive effect of the smaller steric hindrance. Therefore,<br />
2a Ru-MOF material shows as high catalytic activity in present reaction as 1a. Similar<br />
observations have been found for the samples 1b-c because of the somewhat larger steric<br />
hindrance of OH-groups in spite of the type A defect in these materials. To note, using<br />
IRMOF-3 as catalysts in the same reaction, 96% conversion after 60 min was achieved.<br />
However, this could be attributed to the higher regent ratio (aniline: 2,5-hexanedione =<br />
1:1.7). [245] Nevertheless, the current study using Ru-DEMOFs as catalysts give us insight<br />
into the influence of catalytic activity caused by different defect types. For further<br />
investigation, it would also be interesting to optimize the catalytic activity by improving<br />
the regent ratio.<br />
Figure 4.38. Time-yield plot of Paal-Knorr synthesis of aniline reacting with 2,5-hexadione, giving<br />
the pyrrole employing Ru-DEMOFs 1a-1d (with 8%, 20%, 32% and 37% 5-OH-ip incorporation,<br />
respectively) as catalysts in comparison with the parent Ru-MOF(parent). Blank stands for the<br />
experiment where no catalyst was added into the reaction mixture.