pKa values and gas-phase acidities of superacid molecul
pKa values and gas-phase acidities of superacid molecul
pKa values and gas-phase acidities of superacid molecul
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
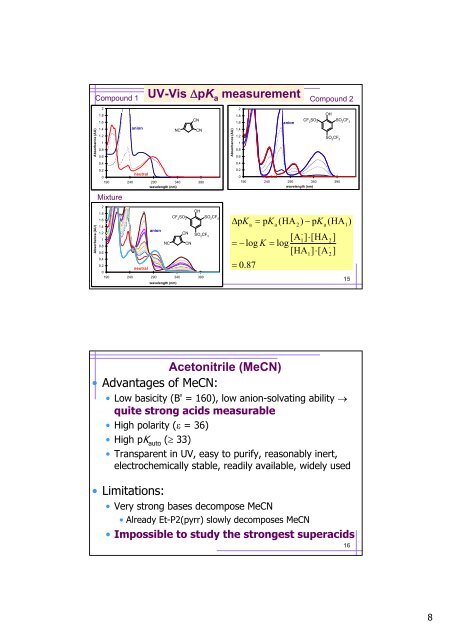
UV-Vis ΔpK a measurement<br />
Compound 1 Compound 2<br />
Absorbance (AU)<br />
Absorbance (AU)<br />
2<br />
1.8<br />
1.6<br />
1.4<br />
1.2<br />
1<br />
0.8<br />
0.6<br />
0.4<br />
0.2<br />
0<br />
anion<br />
neutral<br />
190 240 290 340 390<br />
wavelength (nm)<br />
Mixture<br />
2<br />
1.8<br />
1.6<br />
1.4<br />
1.2<br />
1<br />
0.8<br />
0.6<br />
0.4<br />
anion<br />
NC<br />
NC<br />
CN<br />
CN<br />
CN<br />
OH<br />
CF 2 SO 2<br />
CN SO 2 CF 3<br />
0.2<br />
neutral<br />
0<br />
190 240 290 340 390<br />
wavelength (nm)<br />
SO 2 CF 3<br />
Absorbance (AU)<br />
2<br />
1.8<br />
1.6<br />
1.4<br />
1.2<br />
1<br />
0.8<br />
0.6<br />
0.4<br />
0.2<br />
neutral<br />
190 240 290 340 390<br />
wavelength (nm)<br />
0<br />
ΔpK<br />
a<br />
= 0.87<br />
= pK<br />
a<br />
anion<br />
(HA ) − pK<br />
(HA )<br />
-<br />
[A1]<br />
⋅[HA2]<br />
= − log K = log<br />
-<br />
[HA ] ⋅[A<br />
]<br />
2<br />
CF 2 SO 2<br />
SO 2 CF 3<br />
1<br />
a<br />
OH<br />
2<br />
SO 2 CF 3<br />
1<br />
15<br />
Acetonitrile (MeCN)<br />
• Advantages <strong>of</strong> MeCN:<br />
• Low basicity (B' = 160), low anion-solvating ability →<br />
quite strong acids measurable<br />
• High polarity (ε = 36)<br />
• High pK auto<br />
(≥ 33)<br />
• Transparent in UV, easy to purify, reasonably inert,<br />
electrochemically stable, readily available, widely used<br />
• Limitations:<br />
• Very strong bases decompose MeCN<br />
• Already Et-P2(pyrr) slowly decomposes MeCN<br />
• Impossible to study the strongest <strong>superacid</strong>s<br />
16<br />
8