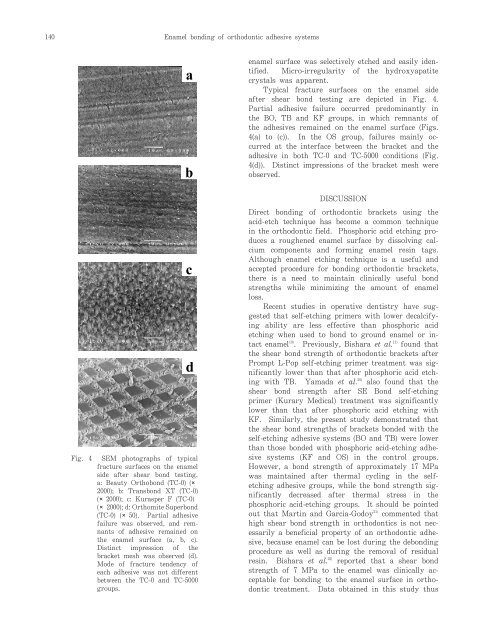
140<strong>Enamel</strong> bonding <strong>of</strong> orthodontic adhesive systemsabenamel surface was selectively <strong>etch</strong>ed <strong>and</strong> easily identified.Micro-irregularity <strong>of</strong> the hydroxyapatitecrystals was apparent.Typical fracture surfaces on the enamel sideafter shear bond testing are depicted in Fig. 4.Partial adhesive failure occurred predominantly inthe BO, TB <strong>and</strong> KF groups, in which remnants <strong>of</strong>the adhesives remained on the enamel surface (Figs.4(a) to (c)). In the OS group, failures mainly occurredat the interface between the bracket <strong>and</strong> theadhesive in both TC-0 <strong>and</strong> TC-5000 conditions (Fig.4(d)). Distinct impressions <strong>of</strong> the bracket mesh wereobserved.Fig. 4 SEM photographs <strong>of</strong> typicalfracture surfaces on the enamelside after shear bond testing.a: Beauty Orthobond (TC-0) (2000); b: Transbond XT (TC-0)(2000); c: Kurasper F (TC-0)(2000); d: Orthomite Superbond(TC-0) (50). Partial adhesivefailure was observed, <strong>and</strong> remnants<strong>of</strong> adhesive remained onthe enamel surface (a, b, c).Distinct impression <strong>of</strong> thebracket mesh was observed (d).Mode <strong>of</strong> fracture tendency <strong>of</strong>each adhesive was not differentbetween the TC-0 <strong>and</strong> TC-5000groups.cdDISCUSSIONDirect bonding <strong>of</strong> orthodontic brackets using theacid-<strong>etch</strong> technique has become a common techniquein the orthodontic field. <strong>Phosphoric</strong> acid <strong>etch</strong>ing producesa roughened enamel surface by dissolving calciumcomponents <strong>and</strong> forming enamel resin tags.Although enamel <strong>etch</strong>ing technique is a useful <strong>and</strong>accepted procedure for bonding orthodontic brackets,there is a need to maintain clinically useful bondstrengths while minimizing the amount <strong>of</strong> enamelloss.Recent studies in operative dentistry have suggestedthat self-<strong>etch</strong>ing primers with lower decalcifyingability are less effective than phosphoric acid<strong>etch</strong>ing when used to bond to ground enamel or intactenamel 19) . Previously, Bishara et al. 11) found thatthe shear bond strength <strong>of</strong> orthodontic brackets afterPrompt L-Pop self-<strong>etch</strong>ing primer treatment was significantlylower than that after phosphoric acid <strong>etch</strong>ingwith TB. Yamada et al. 20) also found that theshear bond strength after SE Bond self-<strong>etch</strong>ingprimer (Kurary Medical) treatment was significantlylower than that after phosphoric acid <strong>etch</strong>ing withKF. Similarly, the present study demonstrated thatthe shear bond strengths <strong>of</strong> brackets bonded with theself-<strong>etch</strong>ing adhesive systems (BO <strong>and</strong> TB) were lowerthan those bonded with phosphoric acid-<strong>etch</strong>ing adhesivesystems (KF <strong>and</strong> OS) in the control groups.However, a bond strength <strong>of</strong> approximately 17 MPawas maintained after thermal cycling in the self<strong>etch</strong>ingadhesive groups, while the bond strength significantlydecreased after thermal stress in thephosphoric acid-<strong>etch</strong>ing groups. It should be pointedout that Martin <strong>and</strong> Garcia-Godoy 21)commented thathigh shear bond strength in orthodontics is not necessarilya beneficial property <strong>of</strong> an orthodontic adhesive,because enamel can be lost during the debondingprocedure as well as during the removal <strong>of</strong> residualresin. Bishara et al. 22) reported that a shear bondstrength <strong>of</strong> 7 MPa to the enamel was clinically acceptablefor bonding to the enamel surface in orthodontictreatment. Data obtained in this study thus
KITAYAMA et al. 141suggested that all the adhesive systems evaluatedwere acceptable for routine clinical use.The effects <strong>of</strong> thermal changes on the bondstrength <strong>of</strong> resin-based materials to hard dental tissues,as well as on their mechanical properties (i.e.,fracture toughness, elastic modulus), have been welldocumented 23,24) . Thermal cycling stresses the bondbetween resin <strong>and</strong> tooth substance <strong>and</strong> might affectbond strength 25) . Christensen 26) commented that impressivein vitro bond strengths were transient whensubjected to temperature changes in the mouth.In the case <strong>of</strong> self-<strong>etch</strong>ing adhesive systems, theacidic monomers in the self-<strong>etch</strong>ing primers are responsiblefor both <strong>etch</strong>ing <strong>and</strong> bonding. As such, thedepth <strong>of</strong> demineralized enamel corresponds to thedepth <strong>of</strong> penetration <strong>of</strong> the adhesive to be polymerized.This mechanism thus circumvents problems associatedwith insufficient penetration as well asimproves the quality <strong>of</strong> hybridization 27-29) , thereby ensuingan excellent mechanical lock.<strong>Phosphoric</strong> acid <strong>etch</strong>ing creates resin tags formechanical retention between enamel <strong>and</strong> resin 30) .However, the resin may not completely infiltrate<strong>etch</strong>ed enamel 31) . A region <strong>of</strong> unprotected enamelprisms might be susceptible to hydrolytic degradationafter thermal cycling. In addition, water diffusioninto the bonded interface between adhesive <strong>and</strong>tooth surface was found to cause the resin to swell<strong>and</strong> become plasticized 32) , as well as reduce enamelhardness due to loss <strong>of</strong> surface calcium 33) .Basically, there are two opinions on the remainingadhesive following bracket debonding. One opinionlargely favors bracket-adhesive interface failurewith the adhesive resin left mainly on the enamelsurface 5,34) , when a heavy-filled resin is used to bondorthodontic attachments. The microporosities createdby <strong>etch</strong>ing are filled with the resin <strong>and</strong> provide mechanicalretention. The second opinion favors failureat the enamel-adhesive resin interface, because thereis less adhesive left to remove from the enamel surfaceafter debonding 34) . Martin <strong>and</strong> Garcia-Godoy 21)suggested that a weaker adhesive with a lowerstrength value might be preferable so as to increasefailure rate at the enamel-adhesive resin interface.In this way, minimal clean-up would be needed withreduced likelihood <strong>of</strong> damage to the enamel.The predominant mode <strong>of</strong> bracket failure for BO,TB, <strong>and</strong> KF was at the enamel-resin interface withless than 50<strong>of</strong> the adhesive on the enamel surface.In the cases <strong>of</strong> BO <strong>and</strong> TB, scratch lines with 600-grit silicone carbide paper were observed afterdebonding, which indicated that the self-<strong>etch</strong>ing adhesivesystems did not damage the enamel surface duringdebonding.As for OS, the predominant mode <strong>of</strong> bracket failurewas at the bracket-adhesive interface with morethan 50<strong>of</strong> the adhesive on the enamel surface. OSis MMA-based, the mechanical properties <strong>of</strong> whichare weaker than the light-cured adhesive resins 35,36) .Hotta et al. 37) reported that 4-MET, a hydrolysisproduct <strong>of</strong> 4-META, promoted effective diffusion <strong>of</strong>monomers into enamel. SEM observations <strong>of</strong> theconditioned enamel surfaces revealed that the self<strong>etch</strong>ingprimers produced less enamel dissolutioncompared with phosphoric acid <strong>etch</strong>ing. Moreover,the morphological appearances <strong>of</strong> the enamel surfacewere different between KF <strong>and</strong> OS, which was probablydue to the different concentrations <strong>of</strong> phosphoricacid in the <strong>etch</strong>ants. The 37phosphoric acid <strong>of</strong> K-<strong>etch</strong>ant in KF was more aggressive than the 65solution<strong>of</strong> Red Activator in OS 31) .Decalcification is a common side effect <strong>of</strong> fixedappliance orthodontic treatment 38) . On this note, orthodontictreatment with multibonded appliancesposes a significant caries risk 6,39) . To counter thisproblem, fluoride-releasing composites for bondingbrackets have attracted considerable attention <strong>and</strong>garnered much interest. This is because they mayinhibit the decalcification <strong>of</strong> enamel around thebrackets by delivering fluoride to the affectedenvironment 40,41) . Furthermore, the remineralizationcapability <strong>and</strong> antibacterial property <strong>of</strong> fluoride mayhelp eliminate the risk <strong>of</strong> dental caries 42) . BO containsS-PRG filler for the release <strong>and</strong> uptake <strong>of</strong> fluorideions, <strong>and</strong> might thus prevent demineralizationbut facilitate remineralization <strong>of</strong> the surroundingenamel 6) .<strong>Orthodontic</strong> bracket bonding is performed on intactenamel. The intact enamel surface ishypermineralized <strong>and</strong> contains more fluoride thanground enamel. Prismless layer on enamel surface 43)is less conducive to bonding by conventional acid gelconditioning 44) <strong>and</strong> self-<strong>etch</strong>ing primer application 45) .Kanemura et al. 45)reported that bond strength wassignificantly reduced when self-<strong>etch</strong>ing systems werebonded to intact enamel. Similarly, Senawongse etal. 46)reported that self-<strong>etch</strong>ing adhesive systems exhibitedsignificantly lower bond strengths than thephosphoric acid-<strong>etch</strong>ing adhesive systems on intactenamel. However, no statistically significant differenceswere found between self-<strong>etch</strong>ing adhesive systems<strong>and</strong> phosphoric acid-<strong>etch</strong>ing adhesive systems toground enamel. Furthermore, bond strengths <strong>of</strong> self<strong>etch</strong>ingadhesive systems to ground enamel were significantlyhigher than those to unground enamel,whereas phosphoric acid <strong>etch</strong>ing systems showed nosuch significant differences between intact <strong>and</strong>ground enamel. Based on the results obtained todate, further research is indeed needed to clarifywhether self-<strong>etch</strong>ing primer adhesive systems couldprovide sufficient bond strength to intact humanenamel.