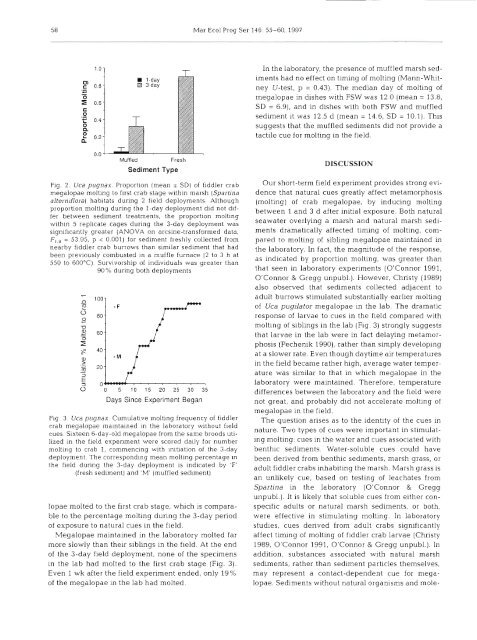
Mar Ecol Prog Ser 146: 55-60, 1997E4l-day3-dayIn the laboratory, the presence <strong>of</strong> muffled marsh sediment~had no effect on tim<strong>in</strong>g <strong>of</strong> molt<strong>in</strong>g (Mann-WhitneyU-test, p = 0.43). The median day <strong>of</strong> molt<strong>in</strong>g <strong>of</strong>megalopae <strong>in</strong> dishes with FSW was 12 0 (mean = 13.8,SD = 6.9), and <strong>in</strong> dishes with both FSW and muffledsediment it was 12.5 d (mean = 14.6, SD = 10.1). Thissuggests that the muffled sediments did not provide atactile cue for molt<strong>in</strong>g <strong>in</strong> the field.MuffledSediment TypeFreshFig. 2. Uca pugnax. Proportion (mean i SD) <strong>of</strong> <strong>fiddler</strong> <strong>crab</strong>megalopae molt<strong>in</strong>g to first <strong>crab</strong> stage with<strong>in</strong> marsh (Spart<strong>in</strong>aalterniflora) habitats dur<strong>in</strong>g 2 field deployments. Althoughproportion molt<strong>in</strong>g dur<strong>in</strong>g the l-day deployment did not differbetween sediment treatments, the proportion molt<strong>in</strong>gwith<strong>in</strong> 5 replicate cages dur<strong>in</strong>g the 3-day deployment wassignificantly greater (ANOVA on arcs<strong>in</strong>e-transformed data,F,,, = 53.95, p < 0.001) for sediment freshly collected fromnearby <strong>fiddler</strong> <strong>crab</strong> burrows than similar sediment that hadbeen previously combusted <strong>in</strong> a muffle furnace (2 to 3 h at550 to 600°C). Survivorship <strong>of</strong> ~ndividuals was greater than90% dur<strong>in</strong>g both deploymentsDays S<strong>in</strong>ce Experiment BeganFig. 3. Uca pugnax. Cumulative molt<strong>in</strong>g frequency <strong>of</strong> <strong>fiddler</strong><strong>crab</strong> megalopae ma<strong>in</strong>ta<strong>in</strong>ed <strong>in</strong> the laboratory without fieldcues. Sixteen 6-day-old megalopae from the same broods utlllzedIn the field expenment were scored daily for numbermoltlng to <strong>crab</strong> 1, commenc<strong>in</strong>g with ~nitiation <strong>of</strong> the 3-daydeployment. The correspond~ng mean molt<strong>in</strong>g percentage Inthe field dur<strong>in</strong>g the 3-day deployment is <strong>in</strong>dicated by 'F'(fresh sediment) and 'M' (muffled sediment)lopae molted to the first <strong>crab</strong> stage, which is comparableto the percentage molt<strong>in</strong>g dur<strong>in</strong>g the 3-day period<strong>of</strong> exposure to natural cues <strong>in</strong> the field.Megalopae ma<strong>in</strong>ta<strong>in</strong>ed <strong>in</strong> the laboratory molted farmore slo~vly than their sibl<strong>in</strong>gs <strong>in</strong> the field. At the end<strong>of</strong> the 3-day field deployment, none <strong>of</strong> the specimens<strong>in</strong> the lab had molted to the first <strong>crab</strong> stage (Fig. 3).Even 1 wk after the field experiment ended, only 19%<strong>of</strong> the megalopae <strong>in</strong> the lab had molted.DISCUSSIONOur short-term field experiment provides strong evidencethat natural cues greatly affect metamorphosis(molt<strong>in</strong>g) <strong>of</strong> <strong>crab</strong> megalopae, by <strong>in</strong>duc<strong>in</strong>g molt<strong>in</strong>gbetween 1 and 3 d after <strong>in</strong>itial exposure. Both naturalseawater overly<strong>in</strong>g a marsh and natural marsh sedimentsdramatically affected tim<strong>in</strong>g <strong>of</strong> molt<strong>in</strong>g, comparedto molt<strong>in</strong>g <strong>of</strong> sibl<strong>in</strong>g megalopae ma<strong>in</strong>ta<strong>in</strong>ed <strong>in</strong>the laboratory. In fact, the magnitude <strong>of</strong> the response,as <strong>in</strong>dicated by proportion rnolt<strong>in</strong>g, was greater thanthat seen <strong>in</strong> laboratory experiments (O'Connor 1991,O'Connor & Gregg unpubl.). However, Christy (1989)also observed that sediments collected adjacent toadult burrows stimulated substantially earlier molt<strong>in</strong>g<strong>of</strong> Uca pugilator megalopae <strong>in</strong> the lab. The dramaticresponse <strong>of</strong> larvae to cues <strong>in</strong> the field compared withmolt<strong>in</strong>g <strong>of</strong> sibl<strong>in</strong>gs <strong>in</strong> the lab (Fig. 3) strongly suggeststhat larvae <strong>in</strong> the lab were <strong>in</strong> fact delaylng metamorphosis(Pechenik 1990), rather than simply develop<strong>in</strong>gat a slower rate. Even though daytime air temperatures<strong>in</strong> the field became rather high, average water temperaturewas similar to that <strong>in</strong> which megalopae <strong>in</strong> thelaboratory were ma<strong>in</strong>ta<strong>in</strong>ed. Therefore, temperaturedifferences between the laboratory and the field werenot great, and probably did not accelerate molt<strong>in</strong>g <strong>of</strong>rnegalopae <strong>in</strong> the field.The question arises as to the identity <strong>of</strong> the cues <strong>in</strong>nature. Two types <strong>of</strong> cues were important <strong>in</strong> stimulat<strong>in</strong>gmolt<strong>in</strong>g: cues <strong>in</strong> the water and cues associated withbenthic sediments. Water-soluble cues could havebeen derived from benthic sediments, marsh grass, oradult <strong>fiddler</strong> <strong>crab</strong>s <strong>in</strong>habit<strong>in</strong>g the marsh. Marsh grass isan unlikely cue, based on test<strong>in</strong>g <strong>of</strong> leachates fromSpart<strong>in</strong>a <strong>in</strong> the laboratory (O'Connor & Greggunpubl.). It is likely that soluble cues from either conspecificadults or natural marsh sedirnents, or both,were effective <strong>in</strong> stimulat<strong>in</strong>g molt<strong>in</strong>g. In laboratorystudies, cues derived from adult <strong>crab</strong>s significantlyaffect tim<strong>in</strong>g <strong>of</strong> molt<strong>in</strong>g <strong>of</strong> <strong>fiddler</strong> <strong>crab</strong> larvae (Christy1989, O'Connor 1991, O'Connor & Gregg unpubl.). Inaddition, substances associated with natural marshsedirnents, rather than sediment particles themselves,may represent a contact-dependent cue for megalopae.Sediments without natural organisms and mole-
O'Connor & Judge- Molt<strong>in</strong>g <strong>of</strong> f~ddler <strong>crab</strong> megalopae 59cules (i.e. muffled marsh sediments <strong>in</strong> FSW) were noteffective at stimulat<strong>in</strong>g molt<strong>in</strong>g <strong>in</strong> the laboratory.Blue <strong>crab</strong> megalopae also molt sooner when exposedto potential habitat cues <strong>in</strong> the laboratory. Megalopalmolt<strong>in</strong>g is advanced by exposure to estuar<strong>in</strong>e seawatercompared with <strong>of</strong>fshore seawater (Wolcott & De Vries1994), and soluble materials leached from seagrassesare particularly effective at stimulat<strong>in</strong>g molt<strong>in</strong>g (Forwardet a1 1994, 1996). The present study providesadditional evidence that <strong>crab</strong> megalopae molt <strong>in</strong>response to chemical cues derived from adult habitats.Benthic <strong>in</strong>vertebrate larvae are known to utilize avariety <strong>of</strong> environmental cues to locate and metamorphose<strong>in</strong> suitable adult habitat. Hydrodynamic processes<strong>in</strong>fluence larval delivery and behavior at a variety<strong>of</strong> spatial scales (Eckman et al. 1983, Butman 1987).Surface characteristics may also serve as importanttactile cues for larvae (Rittsch<strong>of</strong> et al. 1984, Wethey1986). There is ample evidence that larvae activelyexplore such surfaces after contact (Mull<strong>in</strong>eaux & Butman1991, Walters 1992). In addition to their welldocumentedrole <strong>in</strong> crustacean b~ology, chemlcal cueshave been shown important <strong>in</strong> many benthlc groups,<strong>in</strong>clud<strong>in</strong>g polychaetes (Pawlik et al. 1991) and bivalves(Turner et al. 1994), even under simulated natural flowregimes. While we are not able to rank the relativeimportance <strong>of</strong> these cues <strong>in</strong> the present study, thestrong differential response under field cond~tions toour manipulations suggests that chemical cues are criticalto the settl<strong>in</strong>g larvae <strong>of</strong> benthic <strong>in</strong>vertebrates.The present study represents a new approach to thestudy <strong>of</strong> cues stimulat<strong>in</strong>g metamorphosis <strong>of</strong> <strong>in</strong>vertebratelarvae. The cag<strong>in</strong>g technique allows control <strong>of</strong>larval age at exposure to cues, and manipulation <strong>of</strong> thesource <strong>of</strong> the cues to which they are exposed. Our technlquealso ensures that equal numbers <strong>of</strong> larvae areexposed to different potential habitat cues. Other fieldexperiments, primarily deal<strong>in</strong>g with larval choice <strong>of</strong> settlementsite, rely on chance transport <strong>of</strong> planktonic larvaeto experimental sites, and must assume that larvaehave full access to different experimental substrata.Even <strong>in</strong> studies <strong>in</strong> which larvae are pelaglc for short periodsand can be tracked directly by human observers,e.g. relatively large ascidian larvae (Olson 1985, Stoner1990), researchers are unable to control larval abundanceor delivery to f~eld substrata The success <strong>of</strong> thepresent cag<strong>in</strong>g study should open new avenues <strong>of</strong> fieldexper~mentation on larval metamorphosis.Acknowledgenlents We are very grateful to our studentsAmanda Gregg, Kerry Cudmore McCarthy David Cnno, andDanlelle Cigliano for theii assistance <strong>in</strong> the laboratory and thefield We thank Richard B Foiward Jr, Jan Pechenik, and ananonymous reviewer for their comments on the manuscriptWe also thank the staff <strong>of</strong> Demarest Lloyd State Park forallow~ng us to set up our odd-look<strong>in</strong>g expeliment <strong>in</strong> the parkLlTERATURE CITEDButman CA (1987) Larval settlement <strong>of</strong> s<strong>of</strong>t-sediment <strong>in</strong>vertebrates,the spat~al scales <strong>of</strong> pattern expla<strong>in</strong>ed by actlvehabitat selection and the emerglng role <strong>of</strong> hydrodynamicalprocesses. Oceanogr Mar Biol A Rev 25:113-165Chr~sty JH (1989) Rapld development <strong>of</strong> megalopae <strong>of</strong> the <strong>fiddler</strong><strong>crab</strong> Uca pugilatorreared over sediment: implicationsfor models <strong>of</strong> larval recruitment. Mar Ecol Prog Ser 57.259-265Eckman JE (1983) Hydrodynamic processes affect<strong>in</strong>g benthicrecruitment. Limnol Oceanogr 28:241-257Eggleston DB. Armstrong DA (1995) Pre- and post-settlementdc,tei-m<strong>in</strong>ants <strong>of</strong> estuar<strong>in</strong>e Dungeness <strong>crab</strong> recruitmentEcol Monogr 65:193-216Fernandez M, Inbarne 0, Armstrong D (1993) Habitat selectionby young-<strong>of</strong>-the-year Dungeness <strong>crab</strong> Cancer maglsterand predation risk In <strong>in</strong>tertidal habitats Mar Ecol ProgSer 92:171-177Forward RB Jr, De Vries MC, Rittsch<strong>of</strong> D, Frankel DAZ,Bisch<strong>of</strong>f JP, Fisher CM, Welch JM (1996) Effects <strong>of</strong> environmentalcues on metamorphosis <strong>of</strong> the blue <strong>crab</strong> Calllnectessapidus Mar Ecol Prog Ser 131:165-177Forward RB Jr, Frankel DAZ, Rittsch<strong>of</strong> D (1994) Molt<strong>in</strong>g <strong>of</strong>megalopae from the blue <strong>crab</strong> Call<strong>in</strong>ectes sapldus: effects<strong>of</strong> <strong>of</strong>fshore and estuar<strong>in</strong>e cues Mar Ecol Prog Ser 113:55-59Heri-nk<strong>in</strong>d WF, Butler MJ IV (1986) Factors regulat<strong>in</strong>g postlarvalsettlement and juvenile microhab~tat use by sp<strong>in</strong>ylobsters Panullrus argus. Mar Ecol Prog Ser 34:23-30hlense DJ, Wenner EL (1989) Distribution and abundance <strong>of</strong>early life h~story stages <strong>of</strong> the blue <strong>crab</strong>, Calllnectessapidus, <strong>in</strong> tidal marsh creeks near Charleston, South Carol~na.Estuanes 12 157-168Mull~neaux LS, Butman CA (1991) lnitial contact, explorationand attachment <strong>of</strong> barnacle (Balanus amphltrite) cypndssettl<strong>in</strong>g <strong>in</strong> flow. Mar Biol 110.93-103NOAA (1995) T~de tables, high and low water predictions,east coast <strong>of</strong> North and South Amerlca, <strong>in</strong>clud~ng Greenland.US Dept <strong>of</strong> Commerce. Wash<strong>in</strong>gton, DCO'Connor NJ (1991) Flexib~lity <strong>in</strong> timlng <strong>of</strong> the metamorphicmolt by <strong>fiddler</strong> <strong>crab</strong> megalopae Uca pugilator. Mar EcolProg Ser 68:243-247O'Connor NJ (1993) Settlement and recruitment <strong>of</strong> the f~ddler<strong>crab</strong>s Uca pugnax and U pug~lator In a North Carol<strong>in</strong>a,USA, salt marsh Mar Ecol Prog Ser 93-227-234Olson RR (1985) The consequences <strong>of</strong> short-distance larvaldispersal <strong>in</strong> a sesslle mar<strong>in</strong>e <strong>in</strong>vertebrate Ecology 66-30-39Orth RJ, vdn Montfrans J (1987) Ut~lization <strong>of</strong> a seagrassmeadow and t~dal marsh creek by blue <strong>crab</strong>s Call<strong>in</strong>ectessapidus. I. Seasonal and annual vanations <strong>in</strong> abundancewith emphas~s on post-settlementjuveniles. Mar Ecol ProgSer 41 283-294Pabvhk JR. Butman CA, Starczak VAR (1991) Hydrodynamicfacilitation <strong>of</strong> gregarious settlement <strong>of</strong> a reef-budd<strong>in</strong>g tubeworm Science 251 421-424Pechenik JA (1990) Delayed metamorphosis by larvae <strong>of</strong> benthicmar<strong>in</strong>e <strong>in</strong>vertebrates: Does it occur? Is there a price topay? Ophelia 32.63-94R~ttsch<strong>of</strong> D, Branscomb ES, Costlo\v JD (1984) Settlement andbehavior <strong>in</strong> relation to flow and surface <strong>in</strong> larval barnacles,Balanus arnphltrite Darw<strong>in</strong> J Exp Mar Biol Ecol 82.131-146Stoner DS (1990) Recruitment <strong>of</strong> a troplcal colonial ascidian:relatlve ~mportance <strong>of</strong> pre-settlement vs post-settlementprocesses. Ecology 71.1682-1690