5200 R. Taylor, R. Krishna / Chemical Engineering Science 55 (2000) 5183}5229is claimed, but the "gures provided in their paper aresmall and hard to read.Barbosa and Doherty (1988d) point out that the EQstage model equations (including those that account forsimultaneous phase and chemical equilibrium) can berewritten so that they are identical in form to the EQmodel equations in the absence of chemical reactions.The actual #ows and compositions are replaced by thetransformed #ows and compositions, the latter beingde"ned by Eq. (3). The advantage of this approach is thatexisting algorithms and programs can be used to solvethe equations. All that is required is to replace that partof the program that carries out the phase equilibriumcalculations with a new procedure that computes thephase and chemical equilibrium computation and evaluatesthe transformed variables.3.3. Multiple steady-states with the EQ modelMultiple steady-states (MSS) in conventional<strong>distillation</strong> have been known from simulation andtheoretical studies dating back to the 1970s and havebeen a topic of considerable interest in the <strong>distillation</strong>community. However, it is only recently that experimentalveri"cation of their existence has been forthcoming.It is beyond the scope of this article to review thisbody of literature; readers are referred to GuK ttinger(1998) for citations of the original literature and discussionsof the di!erent kinds of multiplicity that havebeen found.The "rst report of MSS in RD appeared in the Russianliterature. Pisarenko, Epifanova and Sera"mov (1988a)found three steady-states for an RD column with just oneproduct stream, two of which were stable. Timofeev,Solokhin and Kalerin (1988) provided a simple analysisof their RD column con"guration. Karpilovsky,Pisarenko and Sera"mov (1997) developed an analysis ofsingle-product columns at in"nite re#ux. Pisarenko et al.(1993) used homotopy methods to locate MSS in RDwith more conventional con"gurations.RADFRAC has been used by, among others, Jacobsand Krishna (1993); Nijhuis, Kerkhof and Mak (1993),Hauan, Hertzberg and Lien (1995, 1997), Perez-Cisneros,Schenk and Gani (1997b) and Eldarsi and Douglas(1998a) for investigation of multiplicity of steady-states inRD columns. For MTBE synthesis using the Jacobs}Krishna column con"guration, shown in Fig. 20(a), varyingthe location of the stage to which methanol is fedresults in either a high or low conversion. When themethanol is fed to stages 10 or 11, steady-state multiplicityis observed (Baur, Higler, Taylor & Krishna, 1999).Explanation for the occurrence of MSS in the MTBEprocess was provided by Hauan et al. (1995, 1997).The ethylene glycol RD process also appears to beparticularly interesting for the investigation of MSS.Ciric and Miao (1994) found as many as nine steadystates,but Kumar and Daoutidis (1999) found `onlya"ve!GuK ttinger and Morari (1997, 1999a,b) develop theso-called R/R analysis for RD columns. The twoFig. 20. (a) Con"guration of the MTBE synthesis column, following Jacobs and Krishna (1993). The column consists of 17 stages. (a) High- andlow-conversion branches obtained by EQ and NEQ simulations. The bottoms #ow in these simulations was "xed at 203 mol/s. The details of thecalculations are given in Baur et al. (1999).
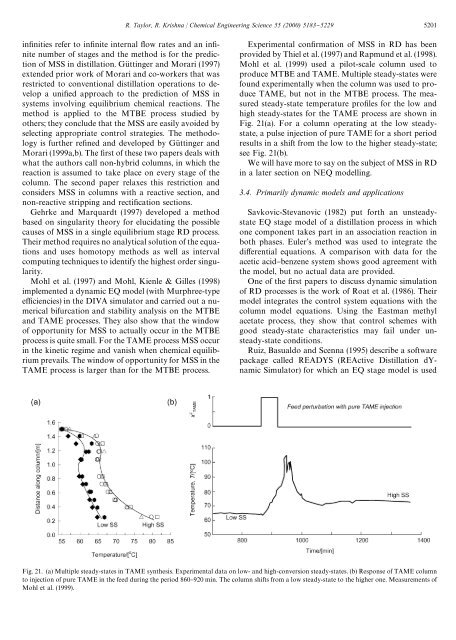
R. Taylor, R. Krishna / Chemical Engineering Science 55 (2000) 5183}5229 5201in"nities refer to in"nite internal #ow rates and an in"-nite number of stages and the method is for the predictionof MSS in <strong>distillation</strong>. GuK ttinger and Morari (1997)extended prior work of Morari and co-workers that wasrestricted to conventional <strong>distillation</strong> operations to developa uni"ed approach to the prediction of MSS insystems involving equilibrium chemical reactions. Themethod is applied to the MTBE process studied byothers; they conclude that the MSS are easily avoided byselecting appropriate control strategies. The methodologyis further re"ned and developed by GuK ttinger andMorari (1999a,b). The "rst of these two papers deals withwhat the authors call non-hybrid columns, in which thereaction is assumed to take place on every stage of thecolumn. The second paper relaxes this restriction andconsiders MSS in columns with a <strong>reactive</strong> section, andnon-<strong>reactive</strong> stripping and recti"cation sections.Gehrke and Marquardt (1997) developed a methodbased on singularity theory for elucidating the possiblecauses of MSS in a single equilibrium stage RD process.Their method requires no analytical solution of the equationsand uses homotopy methods as well as intervalcomputing techniques to identify the highest order singularity.Mohl et al. (1997) and Mohl, Kienle & Gilles (1998)implemented a dynamic EQ model (with Murphree-typee$ciencies) in the DIVA simulator and carried out a numericalbifurcation and stability analysis on the MTBEand TAME processes. They also show that the windowof opportunity for MSS to actually occur in the MTBEprocess is quite small. For the TAME process MSS occurin the kinetic regime and vanish when chemical equilibriumprevails. The window of opportunity for MSS in theTAME process is larger than for the MTBE process.Experimental con"rmation of MSS in RD has beenprovided by Thiel et al. (1997) and Rapmund et al. (1998).Mohl et al. (1999) used a pilot-scale column used toproduce MTBE and TAME. Multiple steady-states werefound experimentally when the column was used to produceTAME, but not in the MTBE process. The measuredsteady-state temperature pro"les for the low andhigh steady-states for the TAME process are shown inFig. 21(a). For a column operating at the low steadystate,a pulse injection of pure TAME for a short periodresults in a shift from the low to the higher steady-state;see Fig. 21(b).We will have more to say on the subject of MSS in RDin a later section on NEQ modelling.3.4. Primarily dynamic models and applicationsSavkovic-Stevanovic (1982) put forth an unsteadystateEQ stage model of a <strong>distillation</strong> process in whichone component takes part in an association reaction inboth phases. Euler's method was used to integrate thedi!erential equations. A comparison with data for theacetic acid}benzene system shows good agreement withthe model, but no actual data are provided.One of the "rst papers to discuss dynamic simulationof RD processes is the work of Roat et al. (1986). Theirmodel integrates the control system equations with thecolumn model equations. Using the Eastman methylacetate process, they show that control schemes withgood steady-state characteristics may fail under unsteady-stateconditions.Ruiz, Basualdo and Scenna (1995) describe a softwarepackage called READYS (REActive Distillation dYnamicSimulator) for which an EQ stage model is usedFig. 21. (a) Multiple steady-states in TAME synthesis. Experimental data on low- and high-conversion steady-states. (b) Response of TAME columnto injection of pure TAME in the feed during the period 860}920 min. The column shifts from a low steady-state to the higher one. Measurements ofMohl et al. (1999).
- Page 7 and 8: R. Taylor, R. Krishna / Chemical En
- Page 9 and 10: R. Taylor, R. Krishna / Chemical En
- Page 12 and 13: 5194 R. Taylor, R. Krishna / Chemic
- Page 14 and 15: 5196 R. Taylor, R. Krishna / Chemic
- Page 16 and 17: 5198 R. Taylor, R. Krishna / Chemic
- Page 20 and 21: 5202 R. Taylor, R. Krishna / Chemic
- Page 22 and 23: 5204 R. Taylor, R. Krishna / Chemic
- Page 24 and 25: 5206 R. Taylor, R. Krishna / Chemic
- Page 26 and 27: 5208 R. Taylor, R. Krishna / Chemic
- Page 28 and 29: 5210 R. Taylor, R. Krishna / Chemic
- Page 30 and 31: 5212 R. Taylor, R. Krishna / Chemic
- Page 32 and 33: 5214 R. Taylor, R. Krishna / Chemic
- Page 34 and 35: 5216 R. Taylor, R. Krishna / Chemic
- Page 36 and 37: 5218 R. Taylor, R. Krishna / Chemic
- Page 38 and 39: 5220 R. Taylor, R. Krishna / Chemic
- Page 40 and 41: 5222 R. Taylor, R. Krishna / Chemic
- Page 42 and 43: 5224 R. Taylor, R. Krishna / Chemic
- Page 44 and 45: 5226 R. Taylor, R. Krishna / Chemic
- Page 47: R. Taylor, R. Krishna / Chemical En