Dynamical Systems in Neuroscience:
Dynamical Systems in Neuroscience: Dynamical Systems in Neuroscience:
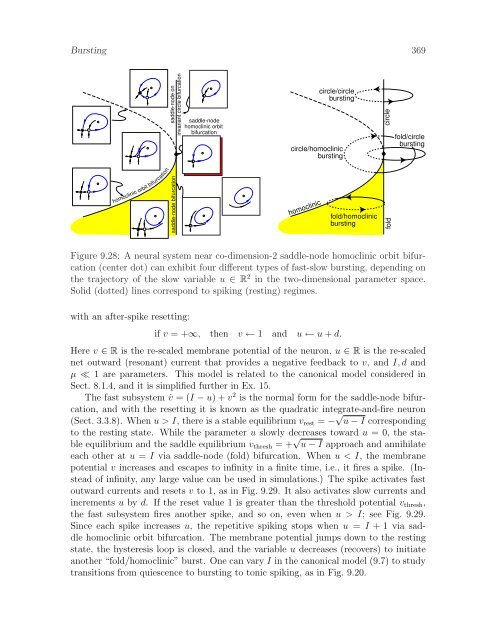
368 BurstingFigure 9.26: Putative “fold/homoclinic” bursting in a pancreatic β-cell (modified fromKinard et al. 1999).0 mV20 mV1 secFigure 9.27: Putative ”fold/homoclinic” bursting in a cell located in pre-Botzingercomplex of rat brain stem (data kindly shared by Christopher A. Del Negro and JackL. Feldman, Systems Neurobiology Laboratory, Department of Neurobiology, UCLA.)Suppose that the hysteresis loop oscillation of the slow variable u has a small amplitude.That is, the saddle-node bifurcation and the saddle homoclinic orbit bifurcationoccur for nearby values of the parameter u. In this case, the fast subsystem of (9.1)is near co-dimension-2 saddle-node homoclinic orbit bifurcation, depicted in Fig. 9.28and studied in Sect. 6.3.6. The figure shows a two-parameter unfolding of the bifurcation,treating u ∈ R 2 as the parameter. A stable equilibrium (resting state) exists inthe left half-plane, and a stable limit cycle (spiking state) exists in the right half-planeof the figure and in the shaded (bistable) region. “Fold/homoclinic” bursting occurswhen the bifurcation parameter, being a slow variable, oscillates between the restingand spiking states through the shaded region. Due to the bistability, the parametercould be one-dimensional. Other trajectories of the slow parameter correspond to othertypes of bursting.In Ex. 16 we prove that there is a piece-wise continuous change of variables thattransforms any “fold/homoclinic” burster with fast subsystem near such a bifurcationinto the canonical model (see Sect. 8.1.5)˙v = I + v 2 − u ,˙u = −µu ,(9.7)
Bursting 369homoclinic orbit bifurcationsaddle-node bifurcation saddle-node oninvariant circle bifurcationsaddle-nodehomoclinic orbitbifurcationcircle/homoclinicburstinghomocliniccircle/circleburstingfold/homoclinicburstingcirclefoldfold/circleburstingFigure 9.28: A neural system near co-dimension-2 saddle-node homoclinic orbit bifurcation(center dot) can exhibit four different types of fast-slow bursting, depending onthe trajectory of the slow variable u ∈ R 2 in the two-dimensional parameter space.Solid (dotted) lines correspond to spiking (resting) regimes.with an after-spike resetting:if v = +∞, then v ← 1 and u ← u + d.Here v ∈ R is the re-scaled membrane potential of the neuron, u ∈ R is the re-scalednet outward (resonant) current that provides a negative feedback to v, and I, d andµ ≪ 1 are parameters. This model is related to the canonical model considered inSect. 8.1.4, and it is simplified further in Ex. 15.The fast subsystem ˙v = (I − u) + v 2 is the normal form for the saddle-node bifurcation,and with the resetting it is known as the quadratic integrate-and-fire neuron(Sect. 3.3.8). When u > I, there is a stable equilibrium v rest = − √ u − I correspondingto the resting state. While the parameter u slowly decreases toward u = 0, the stableequilibrium and the saddle equilibrium v thresh = + √ u − I approach and annihilateeach other at u = I via saddle-node (fold) bifurcation. When u < I, the membranepotential v increases and escapes to infinity in a finite time, i.e., it fires a spike. (Insteadof infinity, any large value can be used in simulations.) The spike activates fastoutward currents and resets v to 1, as in Fig. 9.29. It also activates slow currents andincrements u by d. If the reset value 1 is greater than the threshold potential v thresh ,the fast subsystem fires another spike, and so on, even when u > I; see Fig. 9.29.Since each spike increases u, the repetitive spiking stops when u = I + 1 via saddlehomoclinic orbit bifurcation. The membrane potential jumps down to the restingstate, the hysteresis loop is closed, and the variable u decreases (recovers) to initiateanother “fold/homoclinic” burst. One can vary I in the canonical model (9.7) to studytransitions from quiescence to bursting to tonic spiking, as in Fig. 9.20.
- Page 328 and 329: 318 Simple Modelsthey are able to g
- Page 330 and 331: 320 Simple Modelsv r = −80 mV, an
- Page 332 and 333: 322 Simple Modelsshow in Fig. 7.36.
- Page 334 and 335: 324 Simple ModelsBibliographical No
- Page 336 and 337: 326 Simple ModelsExercises1. (Integ
- Page 338 and 339: 328 Simple Models17. [M.S.] Analyze
- Page 340 and 341: 330 Simple Modelsa35 mV350 msc-NAC
- Page 342 and 343: 332 Simple Modelsrat RTN neuronsimp
- Page 344 and 345: 334 Simple ModelsFigure 8.34: Class
- Page 346 and 347: 336 Simple Modelsspiny neuronlatenc
- Page 348 and 349: 338 Simple Models(a)stellate cellof
- Page 350 and 351: 340 Simple Modelsrat's mitral cell
- Page 352 and 353: 342 Bursting(a) cortical chattering
- Page 354 and 355: 344 Burstingmembranepotential (mV)-
- Page 356 and 357: 346 Burstingvoltage-gatedCa2+-gated
- Page 358 and 359: 348 Burstingslow dynamicsneuronvolt
- Page 360 and 361: 350 Burstingslow inactivation of in
- Page 362 and 363: 352 Bursting9.2.1 Fast-slow burster
- Page 364 and 365: 354 Burstingn-nullclinen slow =-0.0
- Page 366 and 367: 356 Bursting0maxmembrane potential,
- Page 368 and 369: 358 Bursting0membrane potential, V
- Page 370 and 371: 360 BurstingI=0I=4.5425 ms 25 mVI=5
- Page 372 and 373: 362 Burstingmembrane potential, V (
- Page 374 and 375: 364 Bursting9.3 ClassificationIn Fi
- Page 376 and 377: 366 Burstingbifurcation of spiking
- Page 380 and 381: 370 Bursting108spikingslow variable
- Page 382 and 383: 372 Bursting(a)membrane potential,
- Page 384 and 385: 374 Burstingdepending on the type o
- Page 386 and 387: 376 BurstingsubcriticalAndronov-Hop
- Page 388 and 389: 378 Bursting(a)membrane potential,
- Page 390 and 391: spiking380 Burstingfoldbifurcations
- Page 392 and 393: 382 BurstingspikingsupercriticalAnd
- Page 394 and 395: 384 Burstingaction potentials cut2
- Page 396 and 397: 386 Bursting9.4.3 BistabilitySuppos
- Page 398 and 399: 388 BurstingFigure 9.49: The instan
- Page 400 and 401: esting390 Burstingspikesynchronizat
- Page 402 and 403: 392 BurstingReview of Important Con
- Page 404 and 405: 394 BurstingspikingrestingFigure 9.
- Page 406 and 407: 396 Bursting0-10membrane potential,
- Page 408 and 409: 398 Bursting0membrane potential, V
- Page 410 and 411: 400 BurstingFigure 9.61: A cycle-cy
- Page 412 and 413: 402 Bursting28. [Ph.D.] Develop an
- Page 414 and 415: 404 Synchronization (see www.izhike
- Page 416 and 417: 406 Synchronization (see www.izhike
- Page 418 and 419: 408 Solutions to Exercises, Chap. 3
- Page 420 and 421: 410 Solutions to Exercises, Chap. 3
- Page 422 and 423: 412 Solutions to Exercises, Chap. 3
- Page 424 and 425: 414 Solutions to Exercises, Chap. 4
- Page 426 and 427: 416 Solutions to Exercises, Chap. 4
Burst<strong>in</strong>g 369homocl<strong>in</strong>ic orbit bifurcationsaddle-node bifurcation saddle-node on<strong>in</strong>variant circle bifurcationsaddle-nodehomocl<strong>in</strong>ic orbitbifurcationcircle/homocl<strong>in</strong>icburst<strong>in</strong>ghomocl<strong>in</strong>iccircle/circleburst<strong>in</strong>gfold/homocl<strong>in</strong>icburst<strong>in</strong>gcirclefoldfold/circleburst<strong>in</strong>gFigure 9.28: A neural system near co-dimension-2 saddle-node homocl<strong>in</strong>ic orbit bifurcation(center dot) can exhibit four different types of fast-slow burst<strong>in</strong>g, depend<strong>in</strong>g onthe trajectory of the slow variable u ∈ R 2 <strong>in</strong> the two-dimensional parameter space.Solid (dotted) l<strong>in</strong>es correspond to spik<strong>in</strong>g (rest<strong>in</strong>g) regimes.with an after-spike resett<strong>in</strong>g:if v = +∞, then v ← 1 and u ← u + d.Here v ∈ R is the re-scaled membrane potential of the neuron, u ∈ R is the re-scalednet outward (resonant) current that provides a negative feedback to v, and I, d andµ ≪ 1 are parameters. This model is related to the canonical model considered <strong>in</strong>Sect. 8.1.4, and it is simplified further <strong>in</strong> Ex. 15.The fast subsystem ˙v = (I − u) + v 2 is the normal form for the saddle-node bifurcation,and with the resett<strong>in</strong>g it is known as the quadratic <strong>in</strong>tegrate-and-fire neuron(Sect. 3.3.8). When u > I, there is a stable equilibrium v rest = − √ u − I correspond<strong>in</strong>gto the rest<strong>in</strong>g state. While the parameter u slowly decreases toward u = 0, the stableequilibrium and the saddle equilibrium v thresh = + √ u − I approach and annihilateeach other at u = I via saddle-node (fold) bifurcation. When u < I, the membranepotential v <strong>in</strong>creases and escapes to <strong>in</strong>f<strong>in</strong>ity <strong>in</strong> a f<strong>in</strong>ite time, i.e., it fires a spike. (Insteadof <strong>in</strong>f<strong>in</strong>ity, any large value can be used <strong>in</strong> simulations.) The spike activates fastoutward currents and resets v to 1, as <strong>in</strong> Fig. 9.29. It also activates slow currents and<strong>in</strong>crements u by d. If the reset value 1 is greater than the threshold potential v thresh ,the fast subsystem fires another spike, and so on, even when u > I; see Fig. 9.29.S<strong>in</strong>ce each spike <strong>in</strong>creases u, the repetitive spik<strong>in</strong>g stops when u = I + 1 via saddlehomocl<strong>in</strong>ic orbit bifurcation. The membrane potential jumps down to the rest<strong>in</strong>gstate, the hysteresis loop is closed, and the variable u decreases (recovers) to <strong>in</strong>itiateanother “fold/homocl<strong>in</strong>ic” burst. One can vary I <strong>in</strong> the canonical model (9.7) to studytransitions from quiescence to burst<strong>in</strong>g to tonic spik<strong>in</strong>g, as <strong>in</strong> Fig. 9.20.