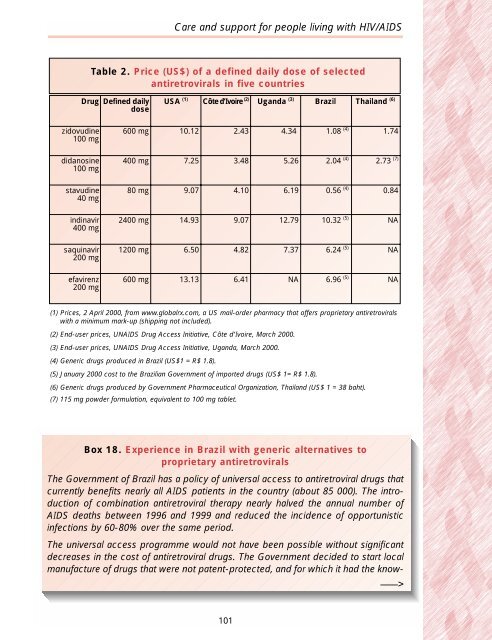
<strong>Care</strong> <strong>and</strong> <strong>support</strong> <strong>for</strong> <strong>people</strong> <strong>living</strong> <strong>with</strong> <strong>HIV</strong>/<strong>AIDS</strong>Table 2. Price (US$) of a defined daily dose of selectedantiretrovirals in five countriesDrug Defined dailydoseUSA (1) Côte d’Ivoire (2) Ug<strong>and</strong>a (3) Brazil Thail<strong>and</strong> (6)zidovudine100 mg600 mg 10.12 2.43 4.34 1.08 (4) 1.74didanosine100 mg400 mg 7.25 3.48 5.26 2.04 (4) 2.73 (7)stavudine40 mg80 mg 9.07 4.10 6.19 0.56 (4) 0.84indinavir400 mg2400 mg 14.93 9.07 12.79 10.32 (5) NAsaquinavir200 mg1200 mg 6.50 4.82 7.37 6.24 (5) NAefavirenz200 mg600 mg 13.13 6.41 NA 6.96 (5) NA(1) Prices, 2 April 2000, from www.globalrx.com, a US mail-order pharmacy that offers proprietary antiretrovirals<strong>with</strong> a minimum mark-up (shipping not included).(2) End-user prices, UN<strong>AIDS</strong> Drug Access Initiative, Côte d’Ivoire, March 2000.(3) End-user prices, UN<strong>AIDS</strong> Drug Access Initiative, Ug<strong>and</strong>a, March 2000.(4) Generic drugs produced in Brazil (US$1 = R$ 1.8).(5) January 2000 cost to the Brazilian Government of imported drugs (US$ 1= R$ 1.8).(6) Generic drugs produced by Government Pharmaceutical Organization, Thail<strong>and</strong> (US$ 1 = 38 baht).(7) 115 mg powder <strong>for</strong>mulation, equivalent to 100 mg tablet.Box 18. Experience in Brazil <strong>with</strong> generic alternatives toproprietary antiretroviralsThe Government of Brazil has a policy of universal access to antiretroviral drugs thatcurrently benefits nearly all <strong>AIDS</strong> patients in the country (about 85 000). The introductionof combination antiretroviral therapy nearly halved the annual number of<strong>AIDS</strong> deaths between 1996 <strong>and</strong> 1999 <strong>and</strong> reduced the incidence of opportunisticinfections by 60-80% over the same period.The universal access programme would not have been possible <strong>with</strong>out significantdecreases in the cost of antiretroviral drugs. The Government decided to start localmanufacture of drugs that were not patent-protected, <strong>and</strong> <strong>for</strong> which it had the know-–––>101
Report on the global <strong>HIV</strong>/<strong>AIDS</strong> epidemic – June 2000how <strong>and</strong> infrastructure. Local production, combined <strong>with</strong> bulk purchases of importedantiretrovirals, led to significant decreases in the programme’s drug costs. Theannual cost of double therapy <strong>with</strong> nucleoside analogues decreased on average by80% between 1996 <strong>and</strong> 2000, from US$ 3812 to US$ 763. For triple therapy <strong>with</strong> twonucleosides <strong>and</strong> one protease inhibitor, the cost reduction was 36% over the sameperiod (from US$ 7342 to US$ 4717) <strong>and</strong> <strong>for</strong> triple therapy <strong>with</strong> two nucleosides <strong>and</strong>one non-nucleoside it was 34% (from US$ 4584 to US$ 3009).The programme’s annual drug costs were approximately R$ 611 million (US$ 339million) in 1999, <strong>and</strong> are expected to rise to R$ 831 million (US$ 462 million) <strong>for</strong> theyear 2000, taking into account both a higher proportion of patients on triple therapy<strong>and</strong> a larger overall number of patients. Between 1997 <strong>and</strong> 1999, approximately146 000 <strong>AIDS</strong>-related hospitalizations were averted, resulting in savings of approximatelyR$ 521 million (US$ 289 million); this has partly offset the high cost of antiretroviraltherapy. At the same time, condom sales increased by nearly half (from 216million to 320 million pieces) between 1996 <strong>and</strong> 1999, <strong>and</strong> dem<strong>and</strong> <strong>for</strong> voluntary <strong>HIV</strong>counselling <strong>and</strong> testing rose 35% between 1996 <strong>and</strong> 1998.Yet another approach is to utilize a safeguard incorporated into the Agreement onTrade-Related Aspects of Intellectual Property Rights (TRIPS), an international treatythat protects patent rights, including those <strong>for</strong> drugs. Patent protection provides animportant incentive <strong>for</strong> innovative research <strong>and</strong> development of new <strong>HIV</strong>/<strong>AIDS</strong> drugs<strong>and</strong>, hopefully, the discovery of <strong>HIV</strong> vaccines, in particular, vaccines suitable <strong>for</strong> usein developing countries (see Box 14, page 68).The TRIPS agreement sets out minimum st<strong>and</strong>ards in relation to intellectual property<strong>for</strong> Member States of the World Trade Organization (WTO). Countries that wish to bemembers of WTO must comply <strong>with</strong> the TRIPS st<strong>and</strong>ards, where necessary bychanging their national laws <strong>and</strong> regulations. In practice, <strong>for</strong> many developing countries,this will mean giving patent protection <strong>for</strong> the first time to pharmaceuticalinventions (both products <strong>and</strong> processes). Under TRIPS, patent protection must beavailable <strong>for</strong> a minimum period of 20 years. However, the TRIPS agreement also<strong>for</strong>esees that in certain circumstances, such as national emergencies, governmentsmay grant third parties the right to produce <strong>and</strong> sell a patented product even <strong>with</strong>outthe consent of the patent holder, according to carefully prescribed conditions.This safeguard, known as “compulsory licensing”, was incorporated into the TRIPSagreement through negotiations by developing countries. Its maintenance as part ofTRIPS has been vigorously defended by nongovernmental organizations <strong>and</strong> activistgroups, such as Act Up, the Consumer Project on Technology <strong>and</strong> Médecins SansFrontières, who have conducted international campaigns <strong>for</strong> improved drug access.These organizations have also sought to obtain compulsory licences on drugs ofinterest to <strong>people</strong> <strong>with</strong> <strong>HIV</strong>/<strong>AIDS</strong> in South Africa <strong>and</strong> Thail<strong>and</strong>. The campaigns didnot result in the issuing of compulsory licences. However, access to the target drugs102