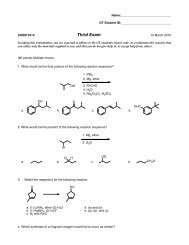
II Alkanes, Alkenes, and Alkynes
II Alkanes, Alkenes, and Alkynes
II Alkanes, Alkenes, and Alkynes
- No tags were found...
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
Conformational Isomers: Isomers that differ as a result of the degree ofrotation around a carbon-carbon single bondLet us take Ethane as an example to describe alkane conformation:Conformation or orientations of alkanes can be expressed using several differentmethods:A) Sawshorse Diagram: A way of representing conformational isomers withstick drawingsB) Newman Projection: A way of representing conformational isomers usingan end-on projection of a carbon-carbon bondEclipsed: Conformation around a carbon-carbon single bond in which attachedatoms are as close together as possibleStaggered: Conformation around a carbon-carbon single bond in which attachedatoms are as far apart as possibleTorsional or Dihedral Angle: Angle between the bonds in the Newman Projection4