Large Volume Inorganic Chemicals - Ammonia ... - ammk-rks.net
Large Volume Inorganic Chemicals - Ammonia ... - ammk-rks.net Large Volume Inorganic Chemicals - Ammonia ... - ammk-rks.net
Chapter 55.4.12 Use of entrainment separatorsDescriptionTo minimise contamination of the scrubber effluent with P 2 O 5 , vapours from vacuum flashcoolers and vacuum evaporators are usually first led through a separator for the removal ofphosphoric acid droplets that are entrained with the vapours.Despite the use of a separator, gas scrubber and condensor effluents may contain low levels ofphosphoric acid. Phosphate removal can be achieved by applying magnesium ammoniumphosphate (struvite) or by calcium phosphate precipitation. Although several phosphorusrecovery plants have been implemented, phosphate removal has not been practiced yet inphosphoric acid plants.Achieved environmental benefitsIncreased P 2 O 5 efficiency and reduced phosphate emissions to water.Cross-media effectsNone believed to be likely.Operational dataNo specific information provided.ApplicabilityGenerally applicable where flash cooling or vacuum evaporation is applied.A similar effect is achievable by using liquid ring pumps or scrubbing with recycling of the ringor scrubbing liquid.EconomicsNo specific information provided. Might be a requirement for the recovery of fluosilicic acid.Driving force for implementationPrevention of emissions.References to literature and example plants[29, RIZA, 2000, 31, EFMA, 2000]246 Large Volume Inorganic Chemicals – Ammonia, Acids and Fertilisers
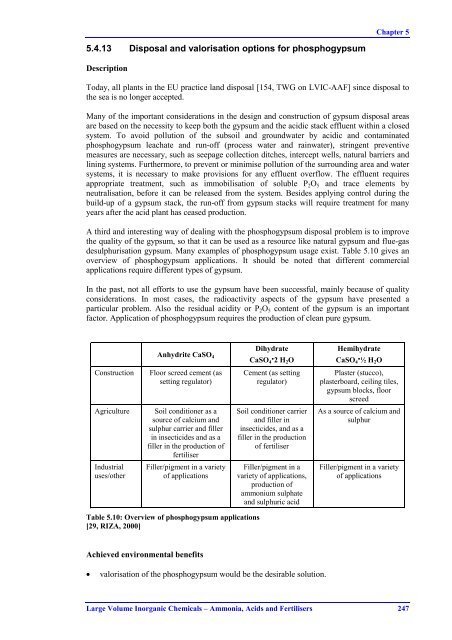
Chapter 55.4.13 Disposal and valorisation options for phosphogypsumDescriptionToday, all plants in the EU practice land disposal [154, TWG on LVIC-AAF] since disposal tothe sea is no longer accepted.Many of the important considerations in the design and construction of gypsum disposal areasare based on the necessity to keep both the gypsum and the acidic stack effluent within a closedsystem. To avoid pollution of the subsoil and groundwater by acidic and contaminatedphosphogypsum leachate and run-off (process water and rainwater), stringent preventivemeasures are necessary, such as seepage collection ditches, intercept wells, natural barriers andlining systems. Furthermore, to prevent or minimise pollution of the surrounding area and watersystems, it is necessary to make provisions for any effluent overflow. The effluent requiresappropriate treatment, such as immobilisation of soluble P 2 O 5 and trace elements byneutralisation, before it can be released from the system. Besides applying control during thebuild-up of a gypsum stack, the run-off from gypsum stacks will require treatment for manyyears after the acid plant has ceased production.A third and interesting way of dealing with the phosphogypsum disposal problem is to improvethe quality of the gypsum, so that it can be used as a resource like natural gypsum and flue-gasdesulphurisation gypsum. Many examples of phosphogypsum usage exist. Table 5.10 gives anoverview of phosphogypsum applications. It should be noted that different commercialapplications require different types of gypsum.In the past, not all efforts to use the gypsum have been successful, mainly because of qualityconsiderations. In most cases, the radioactivity aspects of the gypsum have presented aparticular problem. Also the residual acidity or P 2 O 5 content of the gypsum is an importantfactor. Application of phosphogypsum requires the production of clean pure gypsum.Anhydrite CaSO 4DihydrateCaSO 4·2 H 2 OHemihydrateCaSO 4·½ H 2 OConstructionFloor screed cement (assetting regulator)Cement (as settingregulator)Plaster (stucco),plasterboard, ceiling tiles,gypsum blocks, floorscreedAgricultureSoil conditioner as asource of calcium andsulphur carrier and fillerin insecticides and as afiller in the production offertiliserSoil conditioner carrierand filler ininsecticides, and as afiller in the productionof fertiliserAs a source of calcium andsulphurIndustrialuses/otherFiller/pigment in a varietyof applicationsFiller/pigment in avariety of applications,production ofammonium sulphateand sulphuric acidFiller/pigment in a varietyof applicationsTable 5.10: Overview of phosphogypsum applications[29, RIZA, 2000]Achieved environmental benefits• valorisation of the phosphogypsum would be the desirable solution.Large Volume Inorganic Chemicals – Ammonia, Acids and Fertilisers 247
- Page 224 and 225: Chapter 4EconomicsCost benefits can
- Page 226 and 227: Chapter 4Energy inputRecovery and l
- Page 228 and 229: Chapter 44.4.16 Minimisation and ab
- Page 230 and 231: Chapter 44.4.17 Minimisation of NO
- Page 232 and 233: Chapter 44.4.19 Tail gas scrubbing
- Page 234 and 235: Chapter 44.4.21 Tail gas treatment:
- Page 236 and 237: Chapter 4Economics[58, TAK-S, 2003]
- Page 238 and 239: Chapter 4EconomicsNo specific infor
- Page 240 and 241: Chapter 4BAT is to minimise and red
- Page 242 and 243: Chapter 55.2 Applied processes and
- Page 244 and 245: Chapter 55.2.2.1 Raw materials5.2.2
- Page 246 and 247: Chapter 5OriginChinaMine/regionRare
- Page 248 and 249: Chapter 55.2.2.2 GrindingDepending
- Page 250 and 251: Chapter 55.3 Current emission and c
- Page 252 and 253: Chapter 5Emission of mg/l g/tonne P
- Page 254 and 255: Chapter 55.4 Techniques to consider
- Page 256 and 257: Chapter 55.4.2 Hemihydrate process
- Page 258 and 259: Chapter 55.4.3 Hemi-dihydrate recry
- Page 260 and 261: Chapter 55.4.4 Hemi-dihydrate recry
- Page 262 and 263: Chapter 55.4.5 Di-hemihydrate recry
- Page 264 and 265: Chapter 55.4.6 RepulpingDescription
- Page 266 and 267: Chapter 55.4.7 Fluoride recovery an
- Page 268 and 269: Chapter 55.4.8 Recovery and abateme
- Page 270 and 271: Chapter 5Operational dataNo informa
- Page 272 and 273: Chapter 55.4.11 Decadmation of H 3
- Page 276 and 277: Chapter 5Cross-media effects• dis
- Page 278 and 279: Chapter 5Driving force for implemen
- Page 280 and 281: Chapter 5Cross-media effectsTable 5
- Page 282 and 283: Chapter 5ApplicabilityAt present, o
- Page 285 and 286: Chapter 66 HYDROFLUORIC ACID6.1 Gen
- Page 287 and 288: Chapter 6Component Portion (mass co
- Page 289 and 290: Chapter 66.2.4 Process gas treatmen
- Page 291 and 292: Chapter 66.3 Current emission and c
- Page 293 and 294: Chapter 6Emission of kg/tonne HF Re
- Page 295 and 296: 6.4 Techniques to consider in the d
- Page 297 and 298: Chapter 66.4.2 Energy recovery from
- Page 299 and 300: Chapter 66.4.4 Valorisation of fluo
- Page 301 and 302: Chapter 66.4.6 Scrubbing of tail ga
- Page 303 and 304: 6.4.7 Scrubbing of tail gases: fluo
- Page 305 and 306: Chapter 66.4.8 Abatement of dust em
- Page 307 and 308: Chapter 66.4.9 Waste water treatmen
- Page 309 and 310: Chapter 6Cross-media effects• 5 -
- Page 311 and 312: Chapter 77 NPK AND CN7.1 General in
- Page 313 and 314: Chapter 77.2 Applied processes and
- Page 315 and 316: Chapter 77.2.3 Direct neutralisatio
- Page 317 and 318: Chapter 7Fluorine compounds origina
- Page 319 and 320: Chapter7Per tonne productkWh Nm 3 k
- Page 321 and 322: Chapter7Emission levelmg/Nm 3 ppm k
- Page 323 and 324: Chapter7Emission levelmg/Nm 3 ppm k
Chapter 55.4.13 Disposal and valorisation options for phosphogypsumDescriptionToday, all plants in the EU practice land disposal [154, TWG on LVIC-AAF] since disposal tothe sea is no longer accepted.Many of the important considerations in the design and construction of gypsum disposal areasare based on the necessity to keep both the gypsum and the acidic stack effluent within a closedsystem. To avoid pollution of the subsoil and groundwater by acidic and contaminatedphosphogypsum leachate and run-off (process water and rainwater), stringent preventivemeasures are necessary, such as seepage collection ditches, intercept wells, natural barriers andlining systems. Furthermore, to prevent or minimise pollution of the surrounding area and watersystems, it is necessary to make provisions for any effluent overflow. The effluent requiresappropriate treatment, such as immobilisation of soluble P 2 O 5 and trace elements byneutralisation, before it can be released from the system. Besides applying control during thebuild-up of a gypsum stack, the run-off from gypsum stacks will require treatment for manyyears after the acid plant has ceased production.A third and interesting way of dealing with the phosphogypsum disposal problem is to improvethe quality of the gypsum, so that it can be used as a resource like natural gypsum and flue-gasdesulphurisation gypsum. Many examples of phosphogypsum usage exist. Table 5.10 gives anoverview of phosphogypsum applications. It should be noted that different commercialapplications require different types of gypsum.In the past, not all efforts to use the gypsum have been successful, mainly because of qualityconsiderations. In most cases, the radioactivity aspects of the gypsum have presented aparticular problem. Also the residual acidity or P 2 O 5 content of the gypsum is an importantfactor. Application of phosphogypsum requires the production of clean pure gypsum.Anhydrite CaSO 4DihydrateCaSO 4·2 H 2 OHemihydrateCaSO 4·½ H 2 OConstructionFloor screed cement (assetting regulator)Cement (as settingregulator)Plaster (stucco),plasterboard, ceiling tiles,gypsum blocks, floorscreedAgricultureSoil conditioner as asource of calcium andsulphur carrier and fillerin insecticides and as afiller in the production offertiliserSoil conditioner carrierand filler ininsecticides, and as afiller in the productionof fertiliserAs a source of calcium andsulphurIndustrialuses/otherFiller/pigment in a varietyof applicationsFiller/pigment in avariety of applications,production ofammonium sulphateand sulphuric acidFiller/pigment in a varietyof applicationsTable 5.10: Overview of phosphogypsum applications[29, RIZA, 2000]Achieved environmental benefits• valorisation of the phosphogypsum would be the desirable solution.<strong>Large</strong> <strong>Volume</strong> <strong>Inorganic</strong> <strong>Chemicals</strong> – <strong>Ammonia</strong>, Acids and Fertilisers 247