- Page 1 and 2: Organic Reactions VOLUME IV EDITORI
- Page 3 and 4: PREFACE TO THE SERIES In the course
- Page 5 and 6: CONTENTS CHAPTER PAGE 1. THE DIELS-
- Page 7 and 8: CHAPTER 1 THE DIELS-ALDER REACTION
- Page 9 and 10: 2. C6H5CH=CHA. DIENE SYNTHESIS I S
- Page 11 and 12: DIENE SYNTHESIS I 5 ^Lehmann, Ber.,
- Page 13 and 14: DIENE SYNTHESIS I 7 and IV for thes
- Page 15 and 16: DIENE SYNTHESIS I 9 tion of the com
- Page 17 and 18: Maleic anhydride Fumaric anhydride
- Page 19 and 20: DIENE SYNTHESIS I 13 jugated system
- Page 21 and 22: DIENE SYNTHESIS I 15 Mesaconic (met
- Page 23 and 24: DIENE SYNTHESIS I 17 An interesting
- Page 25 and 26: DIENE SYNTHESIS I 19 CH2 CH3 O Il I
- Page 27 and 28: CH C2H5 I CH2 XXX DIENE SYNTHESIS I
- Page 29 and 30: DIENE SYNTHESIS I cyclic dienes fre
- Page 31 and 32: DIENE SYNTHESIS I 25 Although l,2,3
- Page 33 and 34: DIENE SYNTHESIS I 27 Certain types
- Page 35 and 36: DIENE SYNTHESIS I 29 Hydrocarbons c
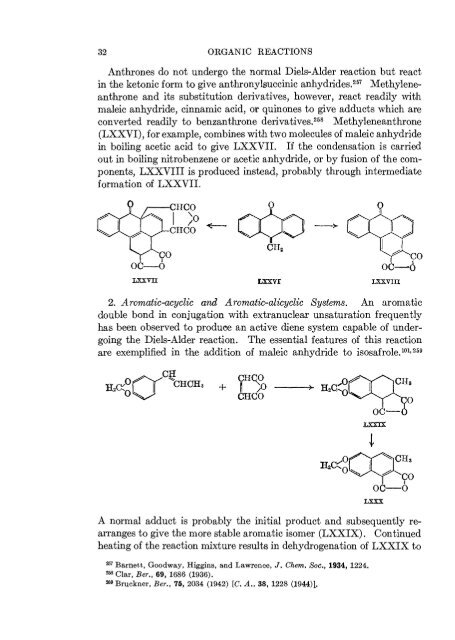
- Page 37: DIENE SYNTHESIS I 31 Diels-Alder re
- Page 41 and 42: DIENE SYNTHESIS I 35 Numerous other
- Page 43 and 44: DIENE SYNTHESIS I 37 Maleic anhydri
- Page 45 and 46: DIENE SYNTHESIS I 39 Indole derivat
- Page 47 and 48: DIENE SYNTHESIS I 41 anhydride. In
- Page 49 and 50: DIENE SYNTHESIS I 43 tetrahydronaph
- Page 51 and 52: DIENE SYNTHESIS I 45 TABLE III—Co
- Page 53 and 54: DIENE SYNTHESIS I 47 TABLE IV ADDtr
- Page 55 and 56: Cycloheptatriene DIENE SYNTHESIS I
- Page 57 and 58: BIENE SYNTHESIS I 51 TABLE V—Cont
- Page 59 and 60: DIENE SYNTHESIS I 53 TABLE VI ADDTJ
- Page 61 and 62: BIENE SYNTHESIS I 55 TABLE VII—Co
- Page 63 and 64: DIENE SYNTHESIS I 57 REFERENCES TO
- Page 65 and 66: DIENE SYNTHESIS I 59 422 Diels and
- Page 67 and 68: DIENE SYNTHESIS II 61 PAGE TABLKs O
- Page 69 and 70: DIENE SYNTHESIS II 63 The configura
- Page 71 and 72: DIENE SYNTHESIS II 65 Aromatic comp
- Page 73 and 74: DIENE SYNTHESIS II 67 55%). 13 Cycl
- Page 75 and 76: H8Cr H8C JCO2H XSV iOCH, DIENE SYNT
- Page 77 and 78: DIENE SYNTHESIS II 71 additions wit
- Page 79 and 80: DIENE SYNTHESIS II , 73 been report
- Page 81 and 82: DIENE SYNTHESIS II 75 drogenation u
- Page 83 and 84: DIENE SYNTHESIS II 77 Allyl, Vinyl,
- Page 85 and 86: DIENE SYNTHESIS II 79 (XLVII) that
- Page 87 and 88: ~r (PH.)* + CO2H CO5 :H CO2CH3 CO2C
- Page 89 and 90:
DIENE SYNTHESIS II 83 covered as th
- Page 91 and 92:
DIENE SYNTHESIS II 85 anhydride fro
- Page 93 and 94:
DIENE SYNTHESIS II 87 or even preve
- Page 95 and 96:
DIENE SYNTHESIS II 89 SELECTION OF
- Page 97 and 98:
DIENE SYNTHESIS II 91 liquid separa
- Page 99 and 100:
Acetylenic Dienophile Ethyl acetyle
- Page 101 and 102:
DIENE SYNTHESIS II 95 TABLE IV YIEL
- Page 103 and 104:
1-Diethylaminobutadiene 1,1-Dimeth
- Page 105 and 106:
1-Methylbutadiene (piperylene) l-Me
- Page 107 and 108:
1,5,5,6-Tetramethylcyclohexadiene (
- Page 109 and 110:
1-Methyl-l-phenylbtrfcadiene l-Met
- Page 111 and 112:
Benzalacetophenone Bicyclohexenyl r
- Page 113 and 114:
f$-Benzoylacrylic acid, 2, b-dimeth
- Page 115 and 116:
Bicyelohexenyl c r 2,3-Dimethylbuta
- Page 117 and 118:
Isoprene Cinnamic acid,2,6~ dimetho
- Page 119 and 120:
Isoprene Cinnamic acid,omeihoxy- (t
- Page 121 and 122:
Coumann Butadiene 2,3-Dimethylbutad
- Page 123 and 124:
1,3-Dimethylbutadiene 1,4-Dimethyl
- Page 125 and 126:
1,1,3-Trimethylbutadiene 1,1,4-Tri
- Page 127 and 128:
2,3-Dimethylbutadiene Croio7toladon
- Page 129 and 130:
2,3-Dimethylbutadiene2,3-Diphenylbu
- Page 131 and 132:
Cyclopentadiene 2,3-Dimethylbutadie
- Page 133 and 134:
Z}4rDihydro-5-bromo- 1\8-dimethoxy-
- Page 135 and 136:
Z^Dihydro-l-ftieihoxy-2-naphthoic a
- Page 137 and 138:
Ethyl ethytidenemalonate Butadiene
- Page 139 and 140:
1,4-Diphenylbutadiene2,3-Diphenylbu
- Page 141 and 142:
6 The product gradually decomposes
- Page 143 and 144:
Cyclohexadiene Cyclopentadiene 2,3-
- Page 145 and 146:
p-Nitrostyrenej 3,4-cfomethoxy- 2,3
- Page 147 and 148:
Dihydrothiophene-1 - dioxide Butadi
- Page 149 and 150:
Piperylcyclone Tetraphenyleyelopent
- Page 151 and 152:
AUyI iodide Cyclopentadiene AUyI is
- Page 153 and 154:
Tetraphenylcyclopentadienone Pheney
- Page 155 and 156:
Indene a,a , -Diphenyl-/3,j3'isoben
- Page 157 and 158:
1,5,5-Trimethyleyclopentadiene Tri
- Page 159 and 160:
Addends Acetylene Tetraphenylcyelop
- Page 161 and 162:
Trimethylcyclopentadiene? (Damsky)
- Page 163 and 164:
Cyclopentadiene 9,10-Dibromoaiithra
- Page 165 and 166:
Propargyl aldehyde 2,3-Dimethylbuta
- Page 167 and 168:
Addends Acetylenedicarboxylic acid
- Page 169 and 170:
I-* SO N-ce-Dimethylindole 3,5-Dim
- Page 171 and 172:
Oi N-Methylindole a-Methylpyrrole N
- Page 173 and 174:
OS Quinaldine Quinoline CX | JCH. j
- Page 175 and 176:
2,3,4-Trimethylpyrrole H3Cp CCO2CH3
- Page 177 and 178:
1,3-Dimethylbutadiene 1,1,3-Trimet
- Page 179 and 180:
DIENE SYNTHESIS II 110 Diels and Al
- Page 181 and 182:
PREPARATION OF AMINES BY REDUCTIVE
- Page 183 and 184:
PREPARATION OF AMINES BY REDUCTIVE
- Page 185 and 186:
PREPARATION OF AMINES BY REDUCTIVE
- Page 187 and 188:
PREPARATION OF AMINES BY REDUCTIVE
- Page 189 and 190:
PREPARATION OF AMINES BY REDUCTIVE
- Page 191 and 192:
PREPARATION OF AMINES BY REDUCTIVE
- Page 193 and 194:
PREPARATION OF AMINES BY REDUCTIVE
- Page 195 and 196:
PREPARATION OF AMINES BY REDUCTIVE
- Page 197 and 198:
PREPARATION OF AMINES BY REDUCTIVE
- Page 199 and 200:
PREPARATION OF AMINES BY REDUCTIVE
- Page 201 and 202:
PREPARATION OF AMINES BY REDUCTIVE
- Page 203 and 204:
PREPARATION OF AMINES BY REDUCTIVE
- Page 205 and 206:
PREPARATION OF AMINES BY REDUCTIVE
- Page 207 and 208:
PREPARATION OF AMINES BY REDUCTIVE
- Page 209 and 210:
PREPARATION OF AMINES BY REDUCTIVE
- Page 211 and 212:
PREPARATION OF AMINES BY REDUCTIVE
- Page 213 and 214:
PREPARATION OF AMINES BY REDUCTIVE
- Page 215 and 216:
CH3CH=CHCHO CH3(CHS)2CH=C(CH2CH3)CH
- Page 217 and 218:
(CHs)2C=CHCOCH3 (CH3)2C=CH(CH2)2COC
- Page 219 and 220:
CH3COCH3 CH3COCH3 CH3COCH2CH3 CH3CO
- Page 221 and 222:
H2N (CH2) 2NH2 H2N(CH2)2NH2 H2N{CH2
- Page 223 and 224:
P-HOC6H4CH2CH(CH3)NH2 CH2O P-CH3OC6
- Page 225 and 226:
CH3NH2 CH3NH2 CH3NH2 CH3NH2 CH3NH2
- Page 227 and 228:
HO(CH2)2NH2 HO(CHa)2NH2 HO(CH2)2NH2
- Page 229 and 230:
(GHg)2COHCH2NH2 (CHs)2COHCH2NH2 CH3
- Page 231 and 232:
Amine, Nitro, Nitroso, or Azo Compo
- Page 233 and 234:
P-HOC6H4NH2 P-HOC6H4NH2 - P-HOC6H4N
- Page 235 and 236:
CeHgNHs C6H5NH2 C6HgNH2 C6H5NH2 P-C
- Page 237 and 238:
HN=C(CH3)CO2H CH2 SehifPs Base / \
- Page 239 and 240:
CH3CH2N=CHC6H5J CH3(CH2)2N=CHCH3f C
- Page 241 and 242:
C6HuN=CH(CH2)2CH3t C6Hi1N=CH(CH2)2C
- Page 243 and 244:
p-C6H5(CH2)2N=CHC6H40H f p-C6H5(CH2
- Page 245 and 246:
C6H5N-=CHCH2CH(CH3)2f C6H5N=CH(CHOH
- Page 247 and 248:
2,4'-CH3C6H4N=CHC6H4CIf 2,4 / -CH3C
- Page 249 and 250:
0-CH3OC6H4N=CH(CHOH)4CH2OH m-CH3OC6
- Page 251 and 252:
Amine Used CH3CH2NH2 Amine Used CH3
- Page 253 and 254:
Nitro Compound Used ^HOC6H4NO2 P-H2
- Page 255 and 256:
C6H5CHOHCH2CH(CH3)- NHCH3 C6H5CHOHC
- Page 257 and 258:
Amine Used (CHs)2NH (CHs)2NH (CH3)2
- Page 259 and 260:
C6H5NH(CHs)2CH3 C6H5NH(CHs)3CH3 C6H
- Page 261 and 262:
PREPARATION OF AMINES BY REDUCTIVE
- Page 263 and 264:
THE ACYLOINS 257 euphony and to avo
- Page 265 and 266:
THE ACYLOINS 259 A mechanism simila
- Page 267 and 268:
THE ACYLOINS 261 There is a strikin
- Page 269 and 270:
TSE ACYLOINS 263 upon the rigid exc
- Page 271 and 272:
THE ACYLOINS 265 carbonate. This pr
- Page 273 and 274:
THE ACYLOINS 267 is thought that py
- Page 275 and 276:
CHAPTER 6 THE SYNTHESIS OF BENZOINS
- Page 277 and 278:
THE SYNTHESIS OF BENZOINS 271 are i
- Page 279 and 280:
THE SYNTHESIS OF BENZOINS 273 metri
- Page 281 and 282:
THE SYNTHESIS OF BENZOINS 275 Symme
- Page 283 and 284:
THE SYNTHESIS OF BENZOINS 277 cent
- Page 285 and 286:
THE SYNTHESIS OF BENZOINS 279 Since
- Page 287 and 288:
Benzoin Reaction 1 * 54 Benzoin Ben
- Page 289 and 290:
LESS STABLE V C6H5COCHOHC6H4OCH3^ V
- Page 291 and 292:
THE SYNTHESIS OF BENZOINS 285 66-86
- Page 293 and 294:
THE SYNTHESIS OF BENZOINS 287 Aliph
- Page 295 and 296:
THE SYNTHESIS OF BENZOINS 289 the i
- Page 297 and 298:
THE SYNTHESIS OF BENZOINS 291 The e
- Page 299 and 300:
THE SYNTHESIS OF BENZOINS 293 effec
- Page 301 and 302:
THE SYNTHESIS OF BENZOINS 295 The e
- Page 303 and 304:
THE SYNTHESIS OF BENZOINS 297 ethox
- Page 305 and 306:
THE SYNTHESIS OF BENZOINS 299 C6HBC
- Page 307 and 308:
Formula CI5HHO2 CI6HHO3 CI6HHO3 CI5
- Page 309 and 310:
Formula C19H22O2 C20H1GO2 C20H24O2
- Page 311 and 312:
CHAPTER 6 SYNTHESIS OF BENZOQUINONE
- Page 313 and 314:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 315 and 316:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 317 and 318:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 319 and 320:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 321 and 322:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 323 and 324:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 325 and 326:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 327 and 328:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 329 and 330:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 331 and 332:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 333 and 334:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 335 and 336:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 337 and 338:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 339 and 340:
SYNTHESIS OF BENZOQUINpNES BY OXIDA
- Page 341 and 342:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 343 and 344:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 345 and 346:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 347 and 348:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 349 and 350:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 351 and 352:
Quinone 5,6-Dimethoxy-2hydi oxyquin
- Page 353 and 354:
Quinone 3-Hydroxythymoqui- J none |
- Page 355 and 356:
Quinone 2-Methoxy-6-tridecylquinone
- Page 357 and 358:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 359 and 360:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 361 and 362:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 363 and 364:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 365 and 366:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 367 and 368:
SYNTHESIS OF BENZOQUINONES BY OXIDA
- Page 369 and 370:
ROSENMUND REDUCTION 363 For accompl
- Page 371 and 372:
ROSENMUND REDUCTION 365 acid in alm
- Page 373 and 374:
ROSENMUND REDUCTION 367 The Hydroge
- Page 375 and 376:
ROSENMUND REDUCTION 369 solution of
- Page 377 and 378:
ROSENMUND REDUCTION 371 TABLE I ALI
- Page 379 and 380:
Acid Chloride Benzoyl chloride p-Ca
- Page 381 and 382:
Acid Chloride 3-Furoic- 4~Carbometh
- Page 383 and 384:
ROSENMUND REDUCTION 377 106 Shoesmi
- Page 385 and 386:
THE WOLFF-KISHNER REDUCTION 379 INT
- Page 387 and 388:
THE W0LFF-KISHNER REDUCTION 381 mol
- Page 389 and 390:
THE WOLFF-KISHNER REDUCTION 383 dan
- Page 391 and 392:
THE WOLFF-KISHNER REDUCTION 385 Sod
- Page 393 and 394:
THE WOLFF-KISHNER REDUCTION 387 TAB
- Page 395 and 396:
THE WOLFF-KISHNER REDUCTION 389 Red
- Page 397 and 398:
THE WOLFF-KISHNER REDUCTION 391 Dir
- Page 399 and 400:
C7Hi2O C7H14O C7H0O2 C7H8O2 C7H6O4
- Page 401 and 402:
C9Hi0O C9Hi2O C9Hi4O Formula C9Hi6O
- Page 403 and 404:
Formula Ci0Hi6O CioHisO Ci0H6O2 Ci0
- Page 405 and 406:
Formula CnH16O CnHi8O CnHi6O2 CnHi8
- Page 407 and 408:
Formula Ci3H22O C13H10O3 Ci3Hi8O3 C
- Page 409 and 410:
Formula CI6HHO2 CI6HHO3 CI6H24O3 C1
- Page 411 and 412:
Formula Ci8H13ON Ci8H2IO3N Ci8Hi9O4
- Page 413 and 414:
Formula C2IH34O C21H30O2 C21H32O2 C
- Page 415 and 416:
Formula C23H32O7 C23Hi7ON C24H3603
- Page 417 and 418:
Formula C25H40O7 C26Hi6O C26H44O C2
- Page 419 and 420:
Formula C29H46O C29H46O2 C29H48O3 C
- Page 421 and 422:
Formula C32H52O3 C32H46O4 C32H50O4
- Page 423 and 424:
THE WOLFF-KISHNER REDUCTION 417 133
- Page 425 and 426:
THE WOLFF-KISHNER REDUCTION 419 235
- Page 427 and 428:
THE WOLFF-KISHNER REDUCTION 421 331
- Page 429 and 430:
DEX Numbers in bold-face type r :ef
- Page 431 and 432:
Dialkylaminoquinonedisulfonates, 35
- Page 433 and 434:
3-Methylpyrene, preparation by Wolf