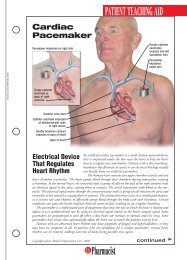
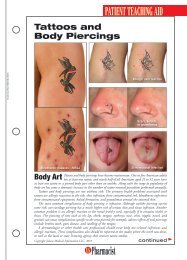
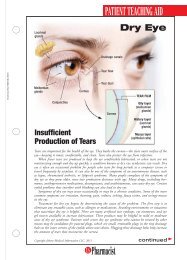
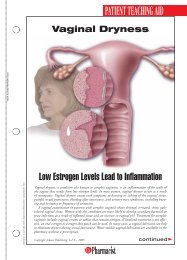
FDA Fast FactsNew LeukemiaTreatment Fast TrackedThe FDA approved ofatumumab(Arzerra) fortreating patients withchronic lymphocyticleukemia (CLL), a bloodand bone marrow cancer.Ofatumumab wasapproved using the acceleratedapproval process forpatients with CLL whosecancer is uncontrolled byother types of chemotherapy.The effectiveness ofthe drug was evaluated in59 patients with CLLwhose disease no longerresponded to the availabletherapies. CLL, whicharises from B cells foundin the body’s immune system,mainly afflictspatients older than 50years, killing about 4,400people each year.“The approval ofArzerra illustrates FDA’scommitment to using theaccelerated approvalprocess to approve drugsfor patients who havelimited therapeuticoptions,” said RichardPazdur, MD, director ofthe Office of OncologyDrug Products in theFDA’s Center for DrugEvaluation and Research.The manufacturer, Glaxo-SmithKline, is currentlyconducting a clinical trialto verify that ofatumumabadded to standardchemotherapy delaysCLL progression.Marketing of IllegalOpioids HaltedThe FDA warned fourFDA Marks AIDS Program MilestoneThe U.S. Department of Health and Human Services (HHS) marked therecent approval of the 100th antiretroviral drug in association with thePresident’s Emergency Plan for AIDS Relief (PEPFAR), which is designedto prevent HIV/AIDS and to treat and care for patients and thoseaffected by the disease worldwide. The PEPFAR program is a joint effortinvolving the FDA and other HHS agencies, the State Department’sOffice of the U.S. Global AIDS Coordinator, the U.S. Department ofDefense, other federal agencies, host country governments, and otherinternational partners. “This milestone exemplifies the dedication, caring,and hard work of all who strive to better the lives of those infected withor affected by HIV/AIDS,” said HHS Secretary Kathleen Sebelius.Drug products used in PEPFAR receive a tentative approval and cannotbe approved for marketing in the United States due to current patentsand marketing exclusivity, yet the drugs must meet the same manufacturing,safety, and efficacy standards required for marketing in this country.As of October 6, 2009, more than 100 products had been assessed by theFDA as part of the PEPFAR program and either fully or tentativelyapproved. Of these, 29 were new products, 71 were versions of drugsapproved in the U.S., and 22 were new combinations or regimens notpreviously authorized in the U.S. In 2008, PEPFAR provided almost $1.6billion in support of HIV/AIDS treatment programs.companies that theymust stop marketingunapproved codeine sulfatetablets, opioid analgesicsused to treat pain.These particular productshave not receivedFDA approval, and theagency has no evidencethat they are safe andeffective. Another manufacturer,Roxane Laboratories,markets approvedcodeine sulfate tablets,and the FDA does notexpect a shortage in thesupply of these drugs.The products and themanufacturers and distributorsthat receivedthe warning letters are:Codeine Sulfate Tablets,30 mg, 60 mg (LehighValley Technologies Inc.,Allentown, Pennsylvania);Codeine SulfateTablets, 30 mg, 60 mg(Cerovene Inc., ValleyCottage, New York);Codeine Sulfate Tablets,30 mg (Dava InternationalInc., Fort Lee,New Jersey); andCodeine Sulfate Tablets,30 mg, 60 mg (GlenmarkGenerics Inc.USA, Mahwah, New Jersey).Companies receivingthe warning lettershave 15 days to give theFDA a plan to discontinuemarketing theunapproved drugs.New Web PageDetails DisposalInstructionsThe FDA launched aWeb page for consumerswith information onhow to dispose of certaindrugs, includinghigh-potency opioidsand other potentiallyharmful controlled substances.The FDA recommendsthat thesemedicines be disposed ofby flushing them downthe sink or toilet to keepthem away from childrenand others. Medicinesnot listed shouldbe thrown away in thehousehold trash aftermixing them with someunpalatable substance,such as coffee grounds,and sealing them in abag or other container.Another option is to disposeof them throughdrug take-back programs,federal and statelaw permitting.24U.S. <strong>Pharmacist</strong> • November 2009 • www.uspharmacist.com
Visit us atASHP Booth 1646QS/1 ®When you think you need a miraclecure for managing your outpatientpharmacy, think QS/1.Until there’s a miracle cure for the aches and pains of managing an outpatientpharmacy, count on real pharmacy management solutions from QS/1. QS/1has the industry’s most complete suite of pharmacy management tools, alldesigned to work together to improve efficiency, safety and operations in busypharmacies like yours. Join the thousands of pharmacies backed by the leaderin pharmacy management. Think QS/1.© 2009, J M SMITH CORPORATION. QS/1 is a registered trademark of the J M Smith Corporation.1-800-231-7776www.qs1.com