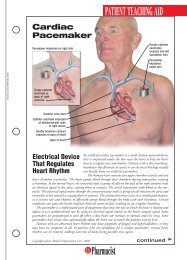
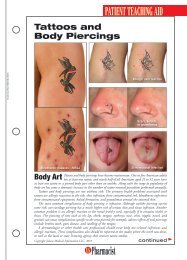
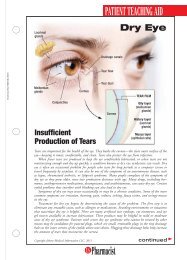
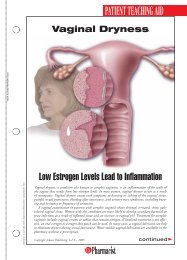
HEALTH SYSTEMS EDITIONSchizophreniaA Review ofPharmacologic andNonpharmacologicTreatments© JUPITERIMAGESPatients with mental illness constitute a populationthat can be challenging for any health care provider,but they offer great opportunities for a pharmacistto make a meaningful contribution. The pharmacist’srole might involve assisting in the selection of medicationregimens, managing adverse drug reactions, orfacilitating adherence. These interventions can be particularlyimportant for patients with schizophrenia.DIAGNOSIS AND CLINICAL PRESENTATIONSchizophrenia is a chronic, debilitating mental illnessthat affects approximately 1% of the population. 1 Owingto the 10% lifetime prevalence of suicide in patientswith the disorder, early recognition and appropriatetreatment are imperative. 2 Schizophrenia typically presentsin late adolescence and persists throughout thepatient’s life, and males typically experience symptoms5 to 7 years earlier than females. 1,3Symptoms of schizophrenia may be classified as positive,negative, or cognitive. Positive symptoms includedisorganized speech or behavior and psychotic characteristicssuch as delusions or hallucinations. Delusions,or fixed false beliefs, occur when a patient misinterpretsan experience, leading toerroneous beliefs involving paranoiaor persecution. Hallucinations involvemore vivid sensory disruptions, withauditory hallucinations being themost common. 1,3 Negative symptoms,including affective flattening, alogia,avolition, and reduced social driveStacy Eon, PharmDPGY2 Pharmacy Resident in PsychiatryUniversity of North Carolina HospitalsChapel Hill, North CarolinaJennifer Durham, PharmDClinical <strong>Pharmacist</strong>Moses Cone Memorial HospitalGreensboro, North Carolina(see TABLE 1), involve the diminution or absence ofnormal functions; they often are refractory to treatment.3,4 Cognitive symptoms are highly prevalent andmay include difficulties with verbal fluency, attention,and working memory. 3 Antipsychotic medicationshave little effect on cognitive symptoms; as aresult, these symptoms affect a patient’s ability to obtainemployment, establish personal relationships, and functionin many social settings. 3According to Diagnostic and Statistical Manual ofMental Disorders-IV-TR criteria, a patient can bediagnosed with schizophrenia if he or she presents withat least two of the following symptoms in addition tosocial dysfunction for a significant amount of time duringa 1-month period, with some symptoms persistingfor at least 6 months: delusions, hallucinations, disorganizedspeech, grossly disorganized or catatonicbehavior, or negative symptoms. 4 The diagnosis canalso be made solely on the basis of bizarre delusionsor hallucinations involving voices. 4PATHOPHYSIOLOGYAlthough the neurochemistry behind schizophrenia isnot fully understood, the disorder isthought to be caused by increaseddopaminergic transmission. 1 Thisincrease in dopamine effect causes thebrain to be hypersensitive to stimuli,making it difficult for the patientto distinguish between reality and hisor her delusions or hallucinations. 1HS-2U.S. <strong>Pharmacist</strong> • November 2009 • www.uspharmacist.com
IN THE TREATMENT OF MRSA BACTEREMIA AND MRSA COMPLICATED SKIN INFECTIONSINSIDE.OUTSIDE.ON HIS SIDE.n Landmark clinical trial of CUBICIN 6 mg/kg once daily demonstratedefficacy in S. aureus bacteremia caused by MRSA and MSSAn Proven clinical success of CUBICIN 4 mg/kg once daily in S. aureuscomplicated skin infections —both MRSA and MSSAINDICATIONS AND IMPORTANT SAFETY INFORMATIONCUBICIN is indicated for the following infections:Complicated skin and skin structure infections caused by susceptibleisolates of the following Gram-positive microorganisms: S. aureus (includingmethicillin-resistant isolates), Streptococcus pyogenes, Streptococcusagalactiae, Streptococcus dysgalactiae subspecies equisimilis, andEnterococcus faecalis (vancomycin-susceptible isolates only). Combinationtherapy may be clinically indicated if the documented or presumedpathogens include Gram-negative or anaerobic organisms.S. aureus bloodstream infections (bacteremia), including those withright-sided infective endocarditis, caused by methicillin-susceptible andmethicillin-resistant isolates. Combination therapy may be clinically indicatedif the documented or presumed pathogens include Gram-negative oranaerobic organisms.The efficacy of CUBICIN in patients with left-sided infective endocarditis dueto S. aureus has not been demonstrated. The clinical trial of CUBICIN inpatients with S. aureus bloodstream infections included limited data frompatients with left-sided infective endocarditis; outcomes in these patientswere poor. CUBICIN has not been studied in patients with prosthetic valveendocarditis or meningitis.Patients with persisting or relapsing S. aureus infection or poor clinicalresponse should have repeat blood cultures. If a culture is positive forwww.cubicin.com©2007 Cubist Pharmaceuticals, Inc.3974071907 September 2007CUBICIN is a registered trademark of Cubist Pharmaceuticals,Inc.S. aureus, MIC susceptibility testing of the isolate should be performedusing a standardized procedure, as well as diagnostic evaluation to ruleout sequestered foci of infection. Appropriate surgical intervention (eg,debridement, removal of prosthetic devices, valve replacement surgery)and/or consideration of a change in antibiotic regimen may be required.CUBICIN is not indicated for the treatment of pneumonia.Clostridium difficile-associated diarrhea (CDAD) has been reported with theuse of nearly all antibacterial agents, including CUBICIN, and may range inseverity from mild diarrhea to fatal colitis. CDAD has been reported to occurover 2 months post-antibiotic treatment. If CDAD is suspected, antibiotictreatment may need to be suspended.Patients receiving CUBICIN should be monitored for the development ofmuscle pain or weakness, particularly of the distal extremities. In patientswho receive CUBICIN, creatine phosphokinase (CPK) levels should bemonitored weekly, and more frequently in patients who received recent prioror concomitant therapy with an HMG-CoA reductase inhibitor. In patientswith renal insufficiency, both renal function and CPK should be monitoredmore frequently. Patients who demonstrate unexplained elevations in CPKwhile receiving CUBICIN should be monitored more frequently.CUBICIN should be discontinued in patients with unexplained signs andsymptoms of myopathy in conjunction with CPK elevation >1000 U/L(~5X ULN), or in patients without reported symptoms who have markedelevations in CPK >2000 U/L (≥10X ULN).Most adverse events reported in CUBICIN clinical trials were mild to moderatein intensity. The most common CUBICIN adverse events were anemia,constipation, diarrhea, nausea,vomiting, injection-site reactions,and headache.Please see Brief Summaryof Prescribing Informationon adjacent page.