Systematic review, meta-analysis and economic modelling of ...
Systematic review, meta-analysis and economic modelling of ... Systematic review, meta-analysis and economic modelling of ...
Assessment of diagnostic and prognostic accuracyTABLE 6 Index and reference standard tests used in studies of TnTStudy Index test assayIndex testthreshold (µg/l)99thpercentile,10% CV andLoD (µg/l) Reference troponin assay and timing a Reference troponinthreshold (µg/l) Prevalence MI (%)Body 2011 46 Roche Diagnostics Elecsys fourthgenerationTnT0.01 0.010.035Not statedRoche fourth-generation TnT at 12 hours 0.035 18Cete 2010 58 Not stated 0.1 Not stated Unspecified TnT at 6 hours 0.1 33Christ 2010 60 Roche Diagnostics Elecsys fourthgenerationTnT0.010.040.010.035Not statedRoche fourth-generation TnT at 6 hoursafter presentation0.04 15Christ 2010 60 Roche Diagnostics Elecsys HsTnT 0.0030.0140.0140.0130.002Roche fourth-generation TnT at 6 hoursafter presentation0.04 15Collinson Roche Diagnostics Elecsys thirdgeneration2006 62 TnT0.03 0.010.030.01Roche third-generation TnT at 24 hours 0.05 29Haltern 2010 67 Roche Diagnostics Elecsys TnT 0.03 0.01Roche TnT at 12 hours 0.03 330.030.01Keller 2009 20 Roche Diagnostics Elecsys TnT 0.010.030.010.030.01Roche TnT or Siemens Dimension RxL TnIwithin 6 hours of admission0.3 (TnT)0.14 (TnI)2926NIHR Journals Library
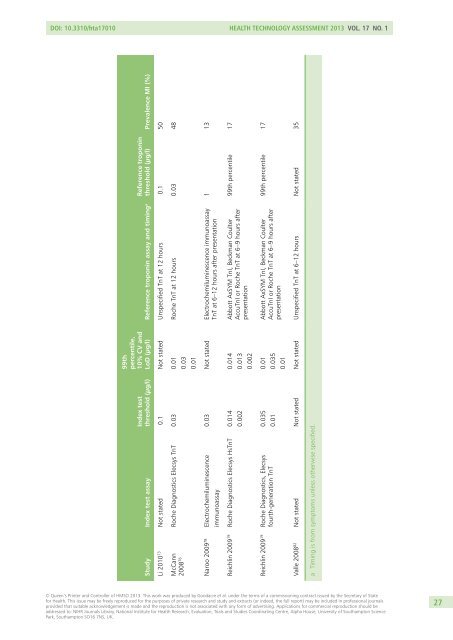
DOI: 10.3310/hta17010 Health Technology Assessment 2013 Vol. 17 No. 1Study Index test assayIndex testthreshold (µg/l)99thpercentile,10% CV andLoD (µg/l) Reference troponin assay and timing a Reference troponinthreshold (µg/l) Prevalence MI (%)Li 2010 73 Not stated 0.1 Not stated Unspecified TnT at 12 hours 0.1 50McCann Roche Diagnostics Elecsys TnT 0.03 0.012008 76 0.030.01Roche TnT at 12 hours 0.03 48Naroo 2009 78 Electrochemiluminescenceimmunoassay0.03 Not stated Electrochemiluminescence immunoassayTnT at 6–12 hours after presentation1 13Reichlin 2009 19 Roche Diagnostics Elecsys HsTnT 0.0140.0020.0140.0130.002Abbott AxSYM TnI, Beckman CoulterAccuTnI or Roche TnT at 6–9 hours afterpresentation99th percentile 17Reichlin 2009 19 Roche Diagnostics, Elecsysfourth-generation TnT0.0350.010.010.0350.01Abbott AxSYM TnI, Beckman CoulterAccuTnI or Roche TnT at 6–9 hours afterpresentation99th percentile 17Valle 2008 82 Not stated Not stated Not stated Unspecified TnT at 6–12 hours Not stated 35a Timing is from symptoms unless otherwise specified.© Queen’s Printer and Controller of HMSO 2013. This work was produced by Goodacre et al. under the terms of a commissioning contract issued by the Secretary of Statefor Health. This issue may be freely reproduced for the purposes of private research and study and extracts (or indeed, the full report) may be included in professional journalsprovided that suitable acknowledgement is made and the reproduction is not associated with any form of advertising. Applications for commercial reproduction should beaddressed to: NIHR Journals Library, National Institute for Health Research, Evaluation, Trials and Studies Coordinating Centre, Alpha House, University of Southampton SciencePark, Southampton SO16 7NS, UK.27
- Page 1: Health Technology AssessmentVOLUME
- Page 7: DOI: 10.3310/hta17010 Health Techno
- Page 10 and 11: ContentsAppendix 5 Expected discoun
- Page 13 and 14: DOI: 10.3310/hta17010 Health Techno
- Page 15 and 16: DOI: 10.3310/hta17010 Health Techno
- Page 17 and 18: DOI: 10.3310/hta17010 Health Techno
- Page 19: DOI: 10.3310/hta17010 Health Techno
- Page 22 and 23: BackgroundTABLE 1 Hospital admissio
- Page 24 and 25: Background2. investigation of the c
- Page 26 and 27: BackgroundComputed tomographic coro
- Page 28 and 29: Research questionsiii. the diagnost
- Page 30 and 31: Assessment of diagnostic and progno
- Page 32 and 33: Assessment of diagnostic and progno
- Page 34 and 35: Assessment of diagnostic and progno
- Page 36 and 37: Assessment of diagnostic and progno
- Page 38 and 39: Assessment of diagnostic and progno
- Page 40 and 41: Assessment of diagnostic and progno
- Page 42 and 43: Assessment of diagnostic and progno
- Page 44 and 45: Assessment of diagnostic and progno
- Page 48 and 49: Assessment of diagnostic and progno
- Page 50 and 51: Assessment of diagnostic and progno
- Page 52 and 53: Assessment of diagnostic and progno
- Page 54 and 55: Assessment of diagnostic and progno
- Page 56 and 57: Assessment of diagnostic and progno
- Page 58 and 59: Assessment of diagnostic and progno
- Page 60 and 61: Assessment of diagnostic and progno
- Page 62 and 63: Assessment of diagnostic and progno
- Page 64 and 65: Assessment of diagnostic and progno
- Page 66 and 67: Assessment of diagnostic and progno
- Page 68 and 69: Assessment of diagnostic and progno
- Page 70 and 71: Assessment of diagnostic and progno
- Page 72 and 73: Assessment of diagnostic and progno
- Page 74 and 75: Assessment of diagnostic and progno
- Page 76 and 77: Assessment of diagnostic and progno
- Page 78 and 79: Assessment of diagnostic and progno
- Page 80 and 81: Assessment of diagnostic and progno
- Page 82 and 83: Assessment of diagnostic and progno
- Page 84 and 85: Assessment of diagnostic and progno
- Page 86 and 87: Assessment of diagnostic and progno
- Page 88 and 89: Assessment of diagnostic and progno
- Page 91 and 92: DOI: 10.3310/hta17010 Health Techno
- Page 93 and 94: DOI: 10.3310/hta17010 Health Techno
- Page 95 and 96: DOI: 10.3310/hta17010 Health Techno
DOI: 10.3310/hta17010 Health Technology Assessment 2013 Vol. 17 No. 1Study Index test assayIndex testthreshold (µg/l)99thpercentile,10% CV <strong>and</strong>LoD (µg/l) Reference troponin assay <strong>and</strong> timing a Reference troponinthreshold (µg/l) Prevalence MI (%)Li 2010 73 Not stated 0.1 Not stated Unspecified TnT at 12 hours 0.1 50McCann Roche Diagnostics Elecsys TnT 0.03 0.012008 76 0.030.01Roche TnT at 12 hours 0.03 48Naroo 2009 78 Electrochemiluminescenceimmunoassay0.03 Not stated Electrochemiluminescence immunoassayTnT at 6–12 hours after presentation1 13Reichlin 2009 19 Roche Diagnostics Elecsys HsTnT 0.0140.0020.0140.0130.002Abbott AxSYM TnI, Beckman CoulterAccuTnI or Roche TnT at 6–9 hours afterpresentation99th percentile 17Reichlin 2009 19 Roche Diagnostics, Elecsysfourth-generation TnT0.0350.010.010.0350.01Abbott AxSYM TnI, Beckman CoulterAccuTnI or Roche TnT at 6–9 hours afterpresentation99th percentile 17Valle 2008 82 Not stated Not stated Not stated Unspecified TnT at 6–12 hours Not stated 35a Timing is from symptoms unless otherwise specified.© Queen’s Printer <strong>and</strong> Controller <strong>of</strong> HMSO 2013. This work was produced by Goodacre et al. under the terms <strong>of</strong> a commissioning contract issued by the Secretary <strong>of</strong> Statefor Health. This issue may be freely reproduced for the purposes <strong>of</strong> private research <strong>and</strong> study <strong>and</strong> extracts (or indeed, the full report) may be included in pr<strong>of</strong>essional journalsprovided that suitable acknowledgement is made <strong>and</strong> the reproduction is not associated with any form <strong>of</strong> advertising. Applications for commercial reproduction should beaddressed to: NIHR Journals Library, National Institute for Health Research, Evaluation, Trials <strong>and</strong> Studies Coordinating Centre, Alpha House, University <strong>of</strong> Southampton SciencePark, Southampton SO16 7NS, UK.27