You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
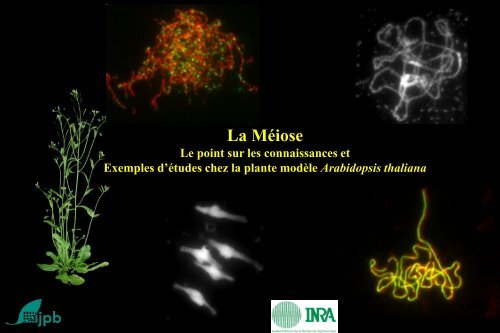
La Méiose<br />
Le point sur les connaissances et<br />
Exemples d’études chez la plante modèle Arabidopsis thaliana
Diploid<br />
Cells<br />
(2n)<br />
Meiosis<br />
Gametes<br />
(haploid cells, n)<br />
Fertilization
One replication + two divisions = ploidy halving<br />
Recombination
Objectif de la méiose: réduire le niveau de ploïdie<br />
Le principe : regrouper pour mieux séparer<br />
Un bivalent<br />
Un monovalent<br />
Phase S<br />
Méiose I<br />
Méiose II
Two sister chromatids<br />
Phase S<br />
Two homologues
Phase S<br />
DAPI<br />
PROPHASE I<br />
Leptotene<br />
ASY1 (axis)<br />
SCC3 (cohesins)
DBA breaks formation<br />
Phase S<br />
PROPHASE I<br />
Leptotène
Double Strand Break formation (DSBs)<br />
PROPHASE I<br />
Leptotene
ASY1 (axis)<br />
DMC1 (recombinase)<br />
DAPI<br />
PROPHASE I<br />
Zygotene<br />
ASY1 (axis)<br />
ZYP1<br />
(synaptonemal complex)
PROPHASE I<br />
Pachytene<br />
ASY1 (axis)<br />
ZYP1<br />
(synaptonemal complex)
DAPI<br />
MLH1<br />
PROPHASE I<br />
Pachytene
PROPHASE I<br />
Diplotene
PROPHASE I<br />
Diakinesis<br />
MLH1
The bivalent!<br />
End of prophase
Metaphase I
Metaphase I/AnaphaseI
Metaphase I/AnaphaseI
Metaphase I/AnaphaseI
Anaphase I
Anaphase I
Anaphase I
telophase I
Metaphase II
Metaphase/Anaphase II
Metaphase/Anaphase II
What meiosis needs?<br />
recombination : at least one<br />
Crossing-Over/bivalent<br />
orientation of the kinetochores<br />
Sister chromatid cohesion<br />
+ Cell cycle control
La cohesion
L’orientation des kinetochores
Christian Huyghe
Le cycle cellulaire
Cyclin/CDK activity<br />
Cyclin<br />
Cyclin Degradation<br />
by APC/C<br />
Cyclin<br />
Cyclin<br />
CDK<br />
APC<br />
CDK<br />
CDK<br />
Active CDK-Cyclin<br />
M-phase entry<br />
Progression through M-phase<br />
Inactive CDK<br />
M-phase exit<br />
Cyclins bind and activate cyclin-dependent kinase (Cdk) to effect cell cycle progression.<br />
Degradation of cyclin by the anaphase-promoting complex<br />
APC : Anaphase-Promoting Complex (E3 ubiquitin ligase) target cell cycle proteins for degradation<br />
by the 26S proteasome
Mitosis<br />
Meiosis<br />
Meiosis Cell Cycle
La recombinaison<br />
• CO essential comme lien entre homologues<br />
• CO et NCO<br />
• Distribution des CO (non homogène, points<br />
chauds, CO obligatoire, interference)
MI<br />
In the absence of COs<br />
AI<br />
Wild type<br />
spo11-1 -/-<br />
Random segregation of<br />
chromosomes….
La Recombinaison méiotique<br />
1) Initiée par des cassures double-brin programmées<br />
2) Consiste en la réparation de ces cassures<br />
3) Deux types de produits: - Crossing-over (CO)<br />
- Conversion génique (NCO)<br />
CO<br />
Nbre de NCO > Nbre de CO<br />
NCO
Wild type<br />
tetrads of pollen grains<br />
CO<br />
Berchowitz LE, Copenhaver GP.<br />
Nat Protoc. 2008;
Gene conversion in A. thaliana<br />
Francis KE, et al. (2007) Proc Natl Acad Sci U S A 104(10): 3913-3918.<br />
NCO
Modified from<br />
Youds & Boulton 2011<br />
ZMMs<br />
SPO11s, PRDs<br />
DMC1, RAD51,<br />
MND1/HOP2, MRE11,<br />
RAD50, COM1,<br />
ASY1, SDS<br />
+<br />
~230<br />
+<br />
FANCM<br />
ROCO2<br />
ROCO4<br />
ZMMs<br />
MLH1/3<br />
RMI/TOP3/BLM<br />
MUS81<br />
~9<br />
~1.5
Modified from<br />
Youds & Boulton 2011<br />
ZMMs<br />
SPO11s, PRDs<br />
DMC1, RAD51,<br />
MND1/HOP2, MRE11,<br />
RAD50, COM1,<br />
ASY1, SDS<br />
+<br />
~230<br />
+<br />
FANCM<br />
ROCO2<br />
ROCO4<br />
ZMMs<br />
MLH1/3<br />
RMI/TOP3/BLM<br />
MUS81<br />
~9<br />
~1.5
SPO11 and friends: Initiation of recombination<br />
Metaphase I<br />
Anaphase I<br />
Wild type<br />
spo11-1<br />
spo11-2<br />
prd1<br />
prd2<br />
prd3<br />
Grelon et al. EMBO J. 2001<br />
Demuyt et al EMBO J. 2007<br />
Demuyt et al, PloS Genet 2009
Modified from<br />
Youds & Boulton 2011<br />
ZMMs<br />
SPO11s, PRDs<br />
DMC1, RAD51,<br />
MND1/HOP2, MRE11,<br />
RAD50, COM1,<br />
ASY1, SDS<br />
+<br />
~230<br />
+<br />
FANCM<br />
ROCO2<br />
ROCO4<br />
ZMMs<br />
MLH1/3<br />
RMI/TOP3/BLM<br />
MUS81<br />
~9<br />
~1.5
Modified from<br />
Youds & Boulton 2011<br />
ZMMs<br />
SPO11s, PRDs<br />
DMC1, RAD51,<br />
MND1/HOP2, MRE11,<br />
RAD50, COM1,<br />
ASY1, SDS<br />
+<br />
~230<br />
+<br />
FANCM<br />
ROCO2<br />
ROCO4<br />
ZMMs<br />
MLH1/3<br />
RMI/TOP3/BLM<br />
MUS81<br />
~9<br />
~1.5
Wild type:<br />
Anaphase I<br />
Atmre11<br />
Puizina et al. 2004<br />
Atrad51<br />
Li et al. 2004
Test for meiotic DSB formation<br />
Wild type:<br />
Anaphase I<br />
Atspo11-1 Atmre11<br />
or<br />
Atspo11-1 Atrad51<br />
Atmre11<br />
Puizina et al. 2004<br />
Atrad51<br />
Li et al. 2004
Modified from<br />
Youds & Boulton 2011<br />
ZMMs<br />
SPO11s, PRDs<br />
DMC1, RAD51,<br />
MND1/HOP2, MRE11,<br />
RAD50, COM1,<br />
ASY1, SDS<br />
+<br />
~230<br />
+<br />
FANCM<br />
ROCO2<br />
ROCO4<br />
ZMMs<br />
MLH1/3<br />
RMI/TOP3/BLM<br />
MUS81<br />
~9<br />
~1.5
Wild-type<br />
zmm-/-<br />
(msh4, msh5, mer3,zip4,<br />
shoc1, ptd1, hei10)<br />
Higgins et al, G&D 2004<br />
Mercier at al. Curr Biol 2005<br />
Chelysheva et al, PLoS gen 2007<br />
Macaisne et al, Curr Biol 2008<br />
Higgins, Vignard et al, Plant J. 2008<br />
Macaisne et al, J cell Science 2011
Modified from<br />
Youds & Boulton 2011<br />
ZMMs<br />
SPO11s, PRDs<br />
DMC1, RAD51,<br />
MND1/HOP2, MRE11,<br />
RAD50, COM1,<br />
ASY1, SDS<br />
+<br />
~230<br />
+<br />
FANCM<br />
ROCO2<br />
ROCO4<br />
ZMMs<br />
MLH1/3<br />
RMI/TOP3/BLM<br />
MUS81<br />
~9<br />
~1.5
Modified from<br />
Youds & Boulton 2011<br />
ZMMs<br />
SPO11s, PRDs<br />
DMC1, RAD51,<br />
MND1/HOP2, MRE11,<br />
RAD50, COM1,<br />
ASY1, SDS<br />
+<br />
~230<br />
+<br />
FANCM<br />
ROCO2<br />
ROCO4<br />
ZMMs<br />
MLH1/3<br />
RMI/TOP3/BLM<br />
MUS81<br />
~9<br />
~220<br />
~1.5
Homologous Recombination Pathways<br />
modified after Lorenz & Whitby, 2006<br />
9 th European Meiosis Meeting, 20/09/2009
La distribution des COs<br />
Kong A, et al. (2002) Nat Genet 31(3):<br />
241-247.<br />
Non homogène<br />
Peu de crossover<br />
Interférence<br />
CO obligatoire
La distribution des COs n’est pas homogène le<br />
long des chromosomes<br />
male<br />
female<br />
cM / Mb<br />
20<br />
Kong A, et al. (2002) Nat Genet 31(3):<br />
241-247.<br />
15<br />
10<br />
5<br />
Human Chromosome 3<br />
Kong et al., Nature Genet. , 2002<br />
Mb<br />
18<br />
16<br />
14<br />
12<br />
10<br />
8<br />
6<br />
4<br />
2<br />
0<br />
0<br />
Mb<br />
15 10<br />
5<br />
knob<br />
NOR<br />
Drouaud J, et al. (2007) PLoS Genet 3(6): e106.<br />
Chromosome 4<br />
Arabidopsis thaliana
La distribution et le taux des COs le long des chromosomes<br />
entre la méiose mâle et la méiose femelle<br />
male<br />
female<br />
cM / Mb<br />
20<br />
15<br />
10<br />
5<br />
0<br />
Mb<br />
18<br />
16<br />
14<br />
12<br />
10<br />
8<br />
6<br />
4<br />
2<br />
0<br />
Mb<br />
15 10<br />
5<br />
knob<br />
NOR<br />
Drouaud Chromosome J, et al. (2007) 4 Arabidopsis PLoS Genet thaliana 3(6): e106.
CO rate (cM/Mb)<br />
taux de crossovers<br />
(cM/Mb)<br />
Pre<br />
Points chauds de recombinaison méiotique<br />
cM/Mb<br />
25<br />
20<br />
15<br />
10<br />
5<br />
0<br />
0 1 2 3 4 5 6 7 8 9 10 12 14 16 18000 kb<br />
10<br />
0<br />
80<br />
60<br />
400<br />
350<br />
20<br />
300<br />
250<br />
A<br />
0<br />
200<br />
150<br />
14a1<br />
AT4G35070<br />
AT4G35080<br />
14a2<br />
100<br />
50<br />
cM/Mb<br />
0<br />
140<br />
120<br />
100<br />
80<br />
60<br />
40<br />
20<br />
chromosome coordinate (kb)<br />
0<br />
0 100 200 300 400 500 600 700 800<br />
Kb
Crossing-over<br />
Crossing-over obligatoire et interférence
Crossing-over<br />
Crossing-over obligatoire et interférence
Crossing-over<br />
Crossing-over obligatoire et interférence
Crossing-over<br />
Crossing-over obligatoire et interférence
Measure of genetic interference<br />
No interference<br />
r I1<br />
Without CO in I2<br />
r I1<br />
With CO in I2<br />
Complete<br />
interference<br />
***
De grandes questions restent sans réponse :<br />
- Comment les chromosomes se reconnaissent-ils?<br />
- Décision CO / NCO ?<br />
- Comment sont contrôlés les crossing-overs :<br />
formation ?<br />
distribution ? …<br />
- Mécanisme de l’interférence<br />
-Mécanismes cohésion/relargage de la cohésion<br />
- Rôle du complexe synaptonémal<br />
- Contrôle du cycle cellulaire<br />
……
What are the molecular mechanisms?<br />
The main model species in the field:
Molecular data available on plant meiosis<br />
50<br />
45<br />
40<br />
Number of identified genes<br />
35<br />
30<br />
25<br />
20<br />
15<br />
10<br />
5<br />
0<br />
1 2 3 4 5 6 7 8 9 10 11 12<br />
1998 2009<br />
A. thaliana<br />
Maize<br />
Rice
Forward and Reverse genetics<br />
Collection of mutants<br />
Phenotype ?<br />
Meiotic<br />
mutant<br />
Phenotypic screen<br />
Mutants<br />
400 000<br />
Insertions<br />
Phenotypic<br />
reversion<br />
mutagenesis<br />
Mutants<br />
Genes<br />
Genes<br />
Genes<br />
Sequence<br />
similarity<br />
OR<br />
Expression<br />
profile
Summary: The genes identified through these screens:<br />
• Initiation of recombination<br />
– SPO11 (Grelon et al. EMBO J. 2001)<br />
– MEI1 (Grelon et al., Plant J 2003)<br />
– PRD1 (Demuyt et al EMBO 2007)<br />
– SKI8 (Jolivet et al, Genes to Cells, 2006)<br />
– PRD2, PRD3 (Demuyt et al, PloS Genet 2009)<br />
• Progression of recombination<br />
– MER3 (Mercier at al. Curr Biol 2005)<br />
– MND1 (Kerzendorfer at al JCS 2006, Vignard et<br />
al, PLoS Gen. 2007)<br />
– ZIP4 (Chelysheva et al, PLoS gen 2007)<br />
– SHOC1/PTD1 (Macaisne et al, Curr Biol 2008;<br />
JCS 2011)<br />
– RMI1 (Chelysheva et al, PloS gen 2008)<br />
– MSH5 (Higgins et al, Plant J. 2008)<br />
• cohesion, cell cycle…<br />
– SWI1 (Mercier et al. Gen. and Dev. 2001)<br />
– MEI1 (Grelon et al., Plant J 2003)<br />
– SCC3/REC8 (Chelysheva et al, JCS 2005)<br />
– AtPS1 (D’erfurth et al, PLoS genet 2008)<br />
– <strong>OSD1</strong> (D’erfurth et al, PLoS Biol 2009)<br />
– TAM1 (D’erfurth et al, PLoS Genet, 2010)
To Mix:<br />
The control of Crossover formation
Modified from<br />
Youds & Boulton 2011<br />
ZMMs<br />
SPO11s, PRDs<br />
DMC1, RAD51,<br />
MND1/HOP2, MRE11,<br />
RAD50, COM1,<br />
ASY1, SDS<br />
+<br />
~250<br />
+<br />
FANCM<br />
ROCO2<br />
ROCO4<br />
ZMMs<br />
MLH1/3 RMI/TOP3/BLM<br />
MUS81 ?<br />
~9<br />
~1.5
How can we identify anti-Crossover activities?<br />
= how can we find mutants with more<br />
crossovers?
Wild-type (fertile)<br />
Meiotic mutant (sterile)<br />
zmm-/- (T-DNA)
Wild-type (fertile)<br />
Meiotic mutant (sterile)<br />
zmm-/- (T-DNA)<br />
zmm-/- (T-DNA)<br />
gene?-/- (EMS)
It works!
oco for RestOration of CrossOvers<br />
roco1-/-zip4-/-<br />
zip4-/-<br />
At least 5 times more CO<br />
in roco1/zip4 than in zip4
Patronus
Mutants<br />
patronus<br />
wt
télophase I
Cohesine<br />
patronus<br />
separase
Expression profile<br />
<strong>OSD1</strong><br />
243 aa<br />
Conserved only in plants<br />
Unknown function
osd1 mutant<br />
male meiosis:<br />
100% dyads<br />
Wild type<br />
osd1
osd1 mutant<br />
selfing<br />
osd1<br />
(2N)<br />
85% 4n<br />
15% 3n
osd1 produces male and female diploid gametes<br />
X<br />
Wild type ♀<br />
(2n)<br />
osd1 ♂<br />
(2n)<br />
100% 3n (2n+n)<br />
X<br />
osd1 ♀<br />
Wild type ♂<br />
85% 3n (n+2n)<br />
15% 2n (n+n)
Meiosis in osd1<br />
Normal meiosis I<br />
No meiosis II
osd1 skip meiosis II?<br />
Meiosis I<br />
Meiosis II
Mother<br />
plant<br />
Meiosis I<br />
Gametes
Genotype of osd1 gametes<br />
(molecular markers on triploid offspring)<br />
Male<br />
♂ 0 79 45 58 43<br />
0 78 46 41 46<br />
0 79 45 58 43<br />
♀<br />
female 0 78 46 41 46<br />
% d’heterozygote 0-20 20-40 40-60 60-80 80-100
Wild type<br />
tetrads of pollen grains<br />
Berchowitz LE, Copenhaver GP.<br />
Nat Protoc. 2008;
osd1<br />
dyads of pollen grains
Genotype of osd1 gametes<br />
(molecular markers on triploid offspring + fluorescent markers on pollen)<br />
male 0 79<br />
45<br />
58<br />
43<br />
9 43 78<br />
femelle 0 78<br />
46<br />
41<br />
46<br />
% d’heterozygote 0-20 20-40 40-60 60-80 80-100
<strong>OSD1</strong> is required for the meiosis I-meiosis II transition
Cyclin/CDK activity<br />
Control of meiosis progression<br />
(similar to Mes1 in pombe?)<br />
<strong>OSD1</strong><br />
?<br />
APC<br />
APC<br />
wt<br />
CDK<br />
Cyclin<br />
Entry threshold<br />
Exit threshold<br />
S<br />
Prophase<br />
(G2)<br />
Meiosis I<br />
Meiosis II<br />
Modified from Marston and Amon. 2004
Apomixis<br />
(clonal reproduction through seeds)<br />
- Relatively common in the plant kingdom<br />
Erigeon Taraxacum Heracium Tripsacum Boechera Ranunculus<br />
- Absent in crops<br />
- Its introduction in crops has potential revolutionary<br />
application
T. Dresselhaus
Sexual reproduction<br />
Diploid<br />
Cells<br />
(2n)<br />
Meiosis<br />
Gametes<br />
(recombined, haploid)<br />
Fertilization
Sexual reproduction<br />
Clonal reproduction<br />
Mitosis<br />
Diploid<br />
Cells<br />
(2n)<br />
Meiosis<br />
Gametes<br />
(clonal, diploid)<br />
Fertilization<br />
Parthenogenesis
1)<br />
Can we replace meiosis by a mitosis?<br />
Three features distinguish meiosis from mitosis<br />
-Two rounds of chromosome segregation<br />
-Co-segregation of sister chromatids at division I<br />
-Recombination
spo11-1 : no recombination<br />
spo11-1<br />
Grelon et al, 2001
1)<br />
Can we replace meiosis by a mitosis?<br />
Three features distinguish meiosis from mitosis<br />
-Two rounds of chromosome segregation<br />
-Co-segregation of sister chromatids at division I<br />
-Recombination
osd1<br />
Wild type<br />
D’erfurth et al, PLoS Biol 2009<br />
D’erfurth, Cromer et al, PLoS Genet, 2010
osd1<br />
dyads of pollen grains<br />
D’erfurth et al, PLoS Biol 2009<br />
D’erfurth, Cromer et al, PLoS Genet, 2010
1)<br />
Can we replace meiosis by a mitosis?<br />
Three features distinguish meiosis from mitosis<br />
-Two rounds of chromosome segregation<br />
-Co-segregation of sister chromatids at division I<br />
-Recombination
spo11/rec8: no recombination and separation of sisters<br />
(but second division still occurs)<br />
spo11/rec8<br />
(Chelysheva et al, JCS 2005)
The combination of the three mutations should<br />
turn meiosis into mitosis (and produce clonal 2n gametes)<br />
-Two rounds of chromosome segregation<br />
-Co-segregation of sister chromatids at division I<br />
-Recombination
d’Erfurth et al, Plos Biol 2009.<br />
d’erfurth, Cromer et al, Plos genet 2010<br />
MiMe<br />
(osd1/spo11/rec8)<br />
male<br />
Female<br />
100% triploid when crossed by wt
98% (n=248)<br />
K5<br />
osd1<br />
Pollen grains<br />
MiMe pollen grains
Mitosis<br />
Diploid<br />
Cells<br />
(2n)<br />
Meiosis<br />
Gametes<br />
(clonal, diploid)<br />
Fertilization
Doubling of ploidy at each generation in the MiMe line<br />
2n<br />
(10 chr)<br />
meiosis<br />
4n<br />
(20 chr)<br />
8n<br />
(40 chr)
Mitosis<br />
Diploid<br />
Cells<br />
(2n)<br />
Meiosis<br />
Gametes<br />
(clonal, diploid)<br />
Fertilization
diploid mother cell<br />
recombined diploid<br />
gamete<br />
haploid GEM<br />
gamete<br />
Fertilization<br />
Simon Chan<br />
Ravi et al, Nature, 2010<br />
Haploid<br />
embryo
Clonal reproduction<br />
Mitosis<br />
Diploid<br />
Cells<br />
(2n)<br />
Meiosis<br />
Gametes<br />
(clonal, diploid)<br />
Fertilisation<br />
with the genome<br />
elimination line (GEM)
cross (♀ x ♂)<br />
Seeds per<br />
siliqua<br />
Germination<br />
rate (%)<br />
Total plants<br />
analysed<br />
Hybrid<br />
diploid* (%)<br />
Triploid (%)<br />
Aneuploid<br />
(%)<br />
Clones* (%)<br />
MiMe x GEM 15 92 156 0.6 13 53 34<br />
GEM x MiMe 23 0.5 12 0 25 33 42<br />
cloned MiMe x GEM 14 91 79 1.3 20 54 24<br />
Marimuthu, Jolivet et al. Science 2011
cross (♀ x ♂)<br />
Seeds per<br />
siliqua<br />
Germination<br />
rate (%)<br />
Total plants<br />
analysed<br />
Hybrid<br />
diploid* (%)<br />
Triploid (%)<br />
Aneuploid<br />
(%)<br />
Clones* (%)<br />
MiMe x GEM 15 92 156 0.6 13 53 34<br />
GEM x MiMe 23 0.5 12 0 25 33 42<br />
cloned MiMe x GEM 14 91 79 1.3 20 54 24<br />
Marimuthu, Jolivet et al. Science 2011
cross (♀ x ♂)<br />
Seeds per<br />
siliqua<br />
Germination<br />
rate (%)<br />
Total plants<br />
analysed<br />
Hybrid<br />
diploid* (%)<br />
Triploid (%)<br />
Aneuploid<br />
(%)<br />
Clones* (%)<br />
MiMe x GEM 15 92 156 0.6 13 53 34<br />
GEM x MiMe 23 0.5 12 0 25 33 42<br />
cloned MiMe x GEM 14 91 79 1.3 20 54 24
Mitosis instead of Meiosis (Apomeiosis)<br />
+ Cross with the genome elimination line<br />
= Clonal seeds !<br />
Marimuthu, Jolivet et al. Science 2011
Mitosis instead of Meiosis (Apomeiosis)<br />
+ Cross with the genome elimination line<br />
= Clonal seeds !<br />
Current limitations:<br />
- dependency on the cross<br />
- Efficiency (35%)<br />
- Not a crop!<br />
Marimuthu, Jolivet et al. Science 2011