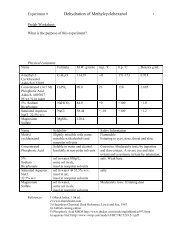
Org. Chem II Experiment 9 Synthesis of Luminol - OChemOnline
Org. Chem II Experiment 9 Synthesis of Luminol - OChemOnline
Org. Chem II Experiment 9 Synthesis of Luminol - OChemOnline
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
<strong>Org</strong>. <strong>Chem</strong> <strong>II</strong> <strong>Experiment</strong> 9 <strong>Synthesis</strong> <strong>of</strong> <strong>Luminol</strong> 2<br />
In neutral solution, luminol exists largely as a dipolar ion (zwitterion). This dipolar ion exhibits a<br />
blue fluorescence after being exposed to light. However, in alkaline solution, luminol is<br />
converted into its dianion.<br />
NH 3<br />
O<br />
NH 2<br />
O<br />
C<br />
C<br />
N<br />
NH<br />
C<br />
N<br />
+ 2 OH + H 2 O<br />
N<br />
C<br />
luminol<br />
O<br />
O<br />
The dianion <strong>of</strong> luminol undergoes a reaction with molecular oxygen to form peroxide <strong>of</strong><br />
unknown structure. This peroxide is unstable and decomposes with the evolution <strong>of</strong> nitrogen gas,<br />
producing the 3-aminophthalate dianion in an electronically excited state.<br />
NH 2<br />
O<br />
NH 2<br />
O<br />
*<br />
C<br />
C<br />
N<br />
N<br />
C<br />
O<br />
+ O ? 2<br />
+ N 2<br />
O<br />
C<br />
O<br />
O<br />
The excited dianion emits a photon, which is observed as visible light.<br />
NH 2 O<br />
*<br />
NH 2 O<br />
C<br />
O<br />
O<br />
C<br />
O<br />
O<br />
+ hν<br />
C<br />
C<br />
O<br />
O<br />
http://www.metnet.state.mt.us/Special/Natives%20Are%20Nice/HTM/gifs/firefly.gif