standard operating procedure (sop) index page – internal - Accord
standard operating procedure (sop) index page – internal - Accord
standard operating procedure (sop) index page – internal - Accord
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
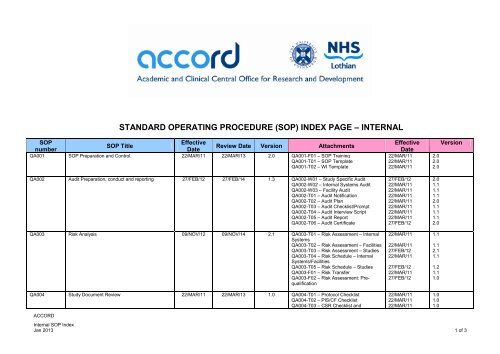
STANDARD OPERATING PROCEDURE (SOP) INDEX PAGE <strong>–</strong> INTERNAL<br />
SOP<br />
Effective<br />
SOP Title<br />
number<br />
Date<br />
Review Date Version Attachments<br />
QA001 SOP Preparation and Control. 22/MAR/11 22/MAR/13 2.0 QA001-F01 <strong>–</strong> SOP Training<br />
QA001-T01 <strong>–</strong> SOP Template<br />
QA001-T02 <strong>–</strong> WI Template<br />
QA002 Audit Preparation, conduct and reporting 27/FEB/12 27/FEB/14 1.3 QA002-W01 <strong>–</strong> Study Specific Audit<br />
QA002-W02 <strong>–</strong> Internal Systems Audit<br />
QA002-W03 <strong>–</strong> Facility Audit<br />
QA002-T01 <strong>–</strong> Audit Notification<br />
QA002-T02 <strong>–</strong> Audit Plan<br />
QA002-T03 <strong>–</strong> Audit Checklist/Prompt<br />
QA002-T04 <strong>–</strong> Audit Interview Script<br />
QA002-T05 <strong>–</strong> Audit Report<br />
QA002-T06 <strong>–</strong> Audit Certificate<br />
QA003 Risk Analysis 09/NOV/12 09/NOV/14 2.1 QA003-T01 <strong>–</strong> Risk Assessment <strong>–</strong> Internal<br />
Systems<br />
QA003-T02 <strong>–</strong> Risk Assessment <strong>–</strong> Facilities<br />
QA003-T03 <strong>–</strong> Risk Assessment <strong>–</strong> Studies<br />
QA003-T04 <strong>–</strong> Risk Schedule <strong>–</strong> Internal<br />
Systems/Facilities<br />
QA003-T05 <strong>–</strong> Risk Schedule <strong>–</strong> Studies<br />
QA003-F01 <strong>–</strong> Risk Transfer<br />
QA003-F02 <strong>–</strong> Risk Assessment: Prequalification<br />
QA004 Study Document Review 22/MAR/11 22/MAR/13 1.0 QA004-T01 <strong>–</strong> Protocol Checklist<br />
QA004-T02 <strong>–</strong> PIS/CF Checklist<br />
QA004-T03 <strong>–</strong> CSR Checklist and<br />
ACCORD<br />
Effective<br />
Date<br />
22/MAR/11<br />
22/MAR/11<br />
22/MAR/11<br />
27/FEB/12<br />
22/MAR/11<br />
22/MAR/11<br />
22/MAR/11<br />
22/MAR/11<br />
22/MAR/11<br />
22/MAR/11<br />
22/MAR/11<br />
27/FEB/12<br />
22/MAR/11<br />
22/MAR/11<br />
27/FEB/12<br />
22/MAR/11<br />
27/FEB/12<br />
22/MAR/11<br />
27/FEB/12<br />
22/MAR/11<br />
22/MAR/11<br />
22/MAR/11<br />
2.0<br />
2.0<br />
2.0<br />
2.0<br />
1.1<br />
1.1<br />
1.1<br />
2.0<br />
1.1<br />
1.1<br />
1.1<br />
2.0<br />
1.1<br />
1.1<br />
2.1<br />
1.1<br />
1.2<br />
1.1<br />
1.0<br />
1.0<br />
1.0<br />
1.0<br />
Version<br />
Internal SOP Index<br />
Jan 2013 1 of 3
SOP<br />
number<br />
SOP Title<br />
Effective<br />
Date<br />
Review Date Version Attachments<br />
Effective Version<br />
Date<br />
BS60001 sampling<br />
QA004-T04 <strong>–</strong> Work plan checklist 22/MAR/11 1.0<br />
QA005 Performance Indicators 22/MAR/11 22/MAR/13 1.1 QA005-T01 <strong>–</strong> Performance indicator<br />
measurements<br />
22/MAR/11 1.1<br />
QA006 Hosting an Audit or Inspection of ACCORD 22/MAR/11 22/MAR/13 1.0<br />
HR001 Establishing and Maintaining a training record 12/AUG/11 12/AUG/13 1.0 HR001-T01 <strong>–</strong> Staff Training Record 12/AUG/11 1.0<br />
HR002 Staff Induction, training and continuity 12/AUG/11 12/AUG/13 1.0<br />
HR003 Clinical monitoring competency 21/MAY/12 21/MAY/14 1.0 HR003-F01 <strong>–</strong> Monitoring Competency<br />
record<br />
Study evaluation 07/MAY/12 07/MAY/14 1.2 GS001-W01 <strong>–</strong> Agreement Preparation <strong>–</strong><br />
GS001<br />
Sponsor<br />
GS001-W02 <strong>–</strong> Research Review<br />
GS001-W03 <strong>–</strong> Scientific and Risk<br />
Assessment Meeting<br />
GS001-W04 <strong>–</strong> Agreement Preparation <strong>–</strong><br />
Host<br />
GS001-W05 <strong>–</strong> Amendments<br />
GS001-F01 <strong>–</strong> SARA review form<br />
GS001-F02 <strong>–</strong> Governance Review<br />
Checklist<br />
GS001-F03 <strong>–</strong> Amendments checklist<br />
GS001-T01 <strong>–</strong> MA Template &optional<br />
inserts<br />
21/MAY/12 1.0<br />
25/APR/11<br />
07MAY/12<br />
01/FEB/12<br />
25/APR/11<br />
15/FEB/2012<br />
21/MAY/12<br />
21/MAY/12<br />
15/JAN/13<br />
01/MAR/12<br />
1.0<br />
2.1<br />
2.0<br />
1.0<br />
1.0<br />
1.3<br />
1.2<br />
1.2<br />
3.1<br />
GS002<br />
FA001<br />
Combined risk analysis 09/NOV/12 09/NOV/14 1.0 GS002-T01 <strong>–</strong> Combined risk assessment 09/NOV/12 1.0<br />
Facilitating a research project 25/APR/11 25/APR/13 1.0 FA001-T01 <strong>–</strong> Clinical investigational agent<br />
classification<br />
FA001-T02 <strong>–</strong> Clinical investigational agent<br />
flowchart<br />
25/APR/11<br />
25/APR/11<br />
1.0<br />
1.0<br />
ACCORD<br />
Internal SOP Index<br />
Jan 2013 2 of 3
SOP<br />
number<br />
SOP Title<br />
Effective<br />
Date<br />
Review Date Version Attachments<br />
FA001-T03 <strong>–</strong> Facilitation checklist<br />
FA001-T04 <strong>–</strong> Quality and safety checklist<br />
FA001-W01 <strong>–</strong> Clinical investigational<br />
agent identification<br />
Effective<br />
Date<br />
12/AUG/11<br />
25/APR/11<br />
25/APR/11<br />
2.0<br />
1.0<br />
1.0<br />
Version<br />
CM001<br />
CM002<br />
CM003<br />
Site initiation visits 14/SEP/11 14/SEP/13 1.0 CM001-T01 <strong>–</strong> SIV & green light report 14/SEP/11 1.0<br />
Site monitoring visits 14/SEP/11 14/SEP/13 1.0 CM002-T01 <strong>–</strong> Site monitoring visit report 14/SEP/11 1.0<br />
Close out visits 14/SEP/11 14/SEP/13 1.0 CM003-T01 <strong>–</strong> Close out visit checklist<br />
CM003-T02 <strong>–</strong> Close out visit report<br />
CR = Clinical Research QA = Quality Assurance CM = Clinical Monitoring<br />
W = Work Instruction<br />
T = Template<br />
F = Form<br />
HR = Human Resources<br />
GS = Governance and Sponsorship<br />
FA = Facilitation<br />
14/SEP/11<br />
14/SEP/11<br />
1.0<br />
1.0<br />
ACCORD<br />
Internal SOP Index<br />
Jan 2013 3 of 3