Cell-Based Assays for Hepatotoxicity:
Cell-Based Assays for Hepatotoxicity: Cell-Based Assays for Hepatotoxicity:
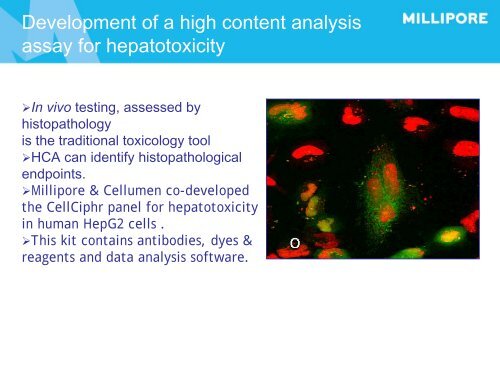
Development of a high content analysis assay for hepatotoxicity ‣In vivo testing, assessed by histopathology is the traditional toxicology tool ‣HCA can identify histopathological endpoints. ‣Millipore & Cellumen co-developed the CellCiphr panel for hepatotoxicity in human HepG2 cells . ‣This kit contains antibodies, dyes & reagents and data analysis software.
Development of a high content analysis assay for hepatotoxicity Eleven hepatotoxicity endpoints in human HepG2 cells are measured using the Cell Ciphr Assay ‣ Cell Loss ‣ Cell Cycle Arrest ‣ DNA Degradation/Apoptosis ‣ Nuclear Size ‣ Oxidative Stress ‣ Stress Kinase Activation ‣ DNA Damage ‣ Mitochondrial Membrane Potential ‣ Mitochondrial Mass ‣ Mitotic Arrest ‣ Cytoskeletal Integrity.
- Page 1 and 2: Cell-Based Assays for Hepatotoxicit
- Page 3 and 4: Now: High Content Analysis Automate
- Page 5: Inability to accurately screen for
- Page 9 and 10: Development of a high content analy
- Page 11 and 12: HepG2 cells treated with Campotheci
- Page 13 and 14: Development of high content analysi
- Page 15 and 16: Cellular Targets for Neurotoxicity
- Page 17 and 18: Assays for detecting toxicity in as
- Page 19 and 20: Detecting changes in neuronal and a
- Page 21 and 22: Assessment of toxin effects on syna
- Page 23 and 24: Preliminary data: comparison of HCA
- Page 25 and 26: Preliminary data: comparison of HCA
- Page 27 and 28: Summary Cell-based assays for hepat
Development of a high content analysis<br />
assay <strong>for</strong> hepatotoxicity<br />
‣In vivo testing, assessed by<br />
histopathology<br />
is the traditional toxicology tool<br />
‣HCA can identify histopathological<br />
endpoints.<br />
‣Millipore & <strong>Cell</strong>umen co-developed<br />
the <strong>Cell</strong>Ciphr panel <strong>for</strong> hepatotoxicity<br />
in human HepG2 cells .<br />
‣This kit contains antibodies, dyes &<br />
reagents and data analysis software.