Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
B/2, Mahalaxmi Chambers,<br />
22, Bhulabhai Desai Road,<br />
Post Box No. 26511,<br />
Mumbai 400 026, India<br />
<strong>Press</strong> <strong>Release</strong><br />
For Immediate publication<br />
<strong>Glenmark</strong> Pharma posts 26.65% revenue growth and 13.42%<br />
increase in net profits in first quarter FY 2006 for its India<br />
operations<br />
Mumbai, July 29, 2005: <strong>Glenmark</strong> Pharmaceuticals Limited [<strong>Glenmark</strong>], one of the<br />
country’s leading research-based pharmaceutical companies, has registered a standalone<br />
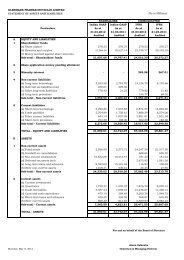
revenue of Rs. 12485.81 lakhs [USD 28.7 mn] for the first quarter ended 30 th<br />
June 2005, a growth of 26.65% compared to the corresponding period of the<br />
previous year. In addition, the Company’s US, Brazil and Swiss subsidiaries<br />
generated total revenues of Rs. 4045.56 lakhs [USD 9.3 mn], including Rs. 2610.60<br />
lakhs [USD 6.00 mn] of NCE milestones received by the Swiss subsidiary during the<br />
quarter. Net profit of the stand-alone India operations for the quarter was Rs.<br />
1108.84 lakhs [USD 2.55 mn], a growth of 13.42% compared to Q1 in FY 2005.<br />
Revenues for Q1 from the domestic formulations segment grew by a healthy 62.57%<br />
to Rs. 8727.87 lakhs [USD 20.06 mn]. As per ORG June 2005 MAT, the retail<br />
demand for the Company’s products showed an increase of 6.5% vis-à-vis the<br />
industry growth of 1.9%. The International business generated revenues of Rs.<br />
2445.11 lakhs [USD 5.62 mn], reflecting a modest growth of 14.09% compared to<br />
the same period last year. The Domestic API and Co-marketing segment generated<br />
revenues of Rs. 1312.83 lakhs [USD 3.02 mn] reflecting a de-growth of 44.05% over<br />
the same period last year.<br />
<strong>Glenmark</strong> has planned several initiatives to improve profitability including<br />
commissioning of its plant at Baddi [Himachal Pradesh] in August 2005. The tax<br />
benefits offered by the location will help offset the impact of the MRP-based Excise.<br />
On the API front, the Company is already working on several new products for the<br />
India market and is also planning to address concerns of co-marketing customers<br />
through the manufacture of co-marketed products at Baddi.<br />
Glenn Saldanha, Managing Director and CEO of <strong>Glenmark</strong> said: “We are confident of<br />
closing the distance between our present performance and targets and maintain our<br />
guidance on revenue and profit for the financial year 2005-2006.”<br />
The Company’s NCE research program has made good progress. The Company<br />
signed a collaboration agreement with Teijin Pharma in Japan to develop, register<br />
and market <strong>Glenmark</strong>’s Asthma/COPD molecule GRC 3886 in Japan. The Company is<br />
now actively seeking a partner in Europe for the molecule. GRC 3886 is on course to<br />
commence Phase II clinical trials in the US by December 2005/January 2006; the<br />
Phase II trials will be conducted by Forest Laboratories, <strong>Glenmark</strong>’s North American<br />
partner for the molecule. The Company is also on-track to file its anti-diabetes lead<br />
molecule GRC 8200 for Phase I trials during the second quarter. Work on other<br />
programmes is progressing well.<br />
About <strong>Glenmark</strong><br />
<strong>Glenmark</strong> Pharmaceuticals Limited is an emerging global, fully integrated, researchbased<br />
pharmaceutical company headquartered in Mumbai, India. The Company has<br />
generic formulation and API business interests in over 70 countries across the world<br />
1 of 2
B/2, Mahalaxmi Chambers,<br />
22, Bhulabhai Desai Road,<br />
Post Box No. 26511,<br />
Mumbai 400 026, India<br />
including the highly regulated markets of USA and Europe. The formulation business<br />
spans several product segments such as Dermatology, Internal Medicine, Paediatrics,<br />
Gynaecology, ENT and Diabetes.<br />
The Company in also engaged in cutting-edge research in discovering new molecules<br />
and runs discovery projects in the areas of inflammation [Asthma/COPD, etc] and<br />
metabolic disorders [Diabetes, Obesity, etc]. It recently announced the licensing of<br />
its first Asthma/COPD molecule to Forest Labs and Teijin Pharma for the North<br />
American and Japanese markets, respectively. This compound has subsequently<br />
completed Phase I clinical trials successfully. [www.glenmarkpharma.com]<br />
For further Information, please contact<br />
Ms. Vaijayanti Karande<br />
Senior Manager<br />
[Corporate Communications]<br />
Tel: 5654 9999 [Ext. 811]<br />
Fax: 23519652<br />
Email : vaijayantik@glenmarkpharma.com<br />
2 of 2





![Formulations [India] â Product List - Glenmark](https://img.yumpu.com/46601329/1/190x245/formulations-india-a-product-list-glenmark.jpg?quality=85)
![Formulation [India] â Product List - Glenmark](https://img.yumpu.com/44013338/1/190x245/formulation-india-a-product-list-glenmark.jpg?quality=85)



![Formulations [India] â Product List - Glenmark](https://img.yumpu.com/35994839/1/190x245/formulations-india-a-product-list-glenmark.jpg?quality=85)

