Download - Glenmark
Download - Glenmark
Download - Glenmark
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
Category<br />
Method<br />
Developed<br />
Methods<br />
Validated<br />
Methods<br />
Transferred<br />
to the<br />
manufacturing<br />
Site<br />
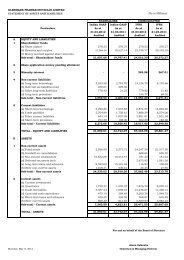
Oral Solid Dosages 18 32 18<br />
Derma products 18 29 18<br />
Oncology products 7 10 3<br />
MDI 11 18 8<br />
DPI 6 – 2<br />
Nasal 1 – 1<br />
NCE 4 5 3<br />
API 3 2 17<br />
Total 68 96 70<br />
Documents for<br />
Drug Substance<br />
(STP, Specs etc.)<br />
– – 170<br />
In Analytical Research activities for NCE research:<br />
a) New analytical test procedures are developed to<br />
establish the structure and evaluate the quality<br />
of NCE prior to initial biological screening.<br />
During pre-clinical studies, we generated<br />
analytical data for establishing the quality and<br />
setting up specification for the release testing<br />
of Drug substances. The methods used to<br />
release the drug substances which are used in<br />
clinical trials, are validated as per International<br />
Regulatory Standards.<br />
b) Physicochemical properties of new chemical<br />
entity are established and characterization<br />
studies are conducted.<br />
c) CMC related Dossiers, study protocols and<br />
study reports were prepared to support various<br />
pre-clinical studies and clinical trial applications<br />
with Regulatory Agencies.<br />
d) We perform polymorphic evaluation and<br />
salt selection studies on various NCEs drug<br />
substance and drug products.<br />
e) Reference standards of NCE were generated<br />
and supplied to CROs and manufacturing sites.<br />
2. Future plan of action<br />
R & D is working on new molecules in the following<br />
segment;<br />
- Antifungal molecules<br />
- Antibacterial molecules<br />
- Antiasthmatic molecules<br />
- Antidiabetic products<br />
- Antiaging products<br />
- Antiinflammatory products<br />
- Atihyperlipidemic products<br />
- Antiosteoporosis products<br />
- Antiemetic products<br />
- Antihypertensive molecules<br />
- Nutraceuticals<br />
- Sunscreens Products<br />
- Technology – such as micro spheres and<br />
aerosols foam Mousse.<br />
- Development of formulations for Semi regulatory<br />
market.<br />
- Development of formulations for Latin American<br />
market.<br />
- Development of formulations for US market.<br />
- Metered dose inhaler products for India Brazil/<br />
US market.<br />
- Development of specialized NDDS products for<br />
Indian/SRM.<br />
- Nasal sprays for Semi regulatory market and<br />
US market.<br />
TECHNOLOGY ABSORPTION, ADOPTION<br />
AND INNOVATION:<br />
1. Efforts in brief towards technology absorption,<br />
adoption and innovation<br />
Our efforts in the area of technology absorption,<br />
adoption and innovation are based on our own<br />
efforts in R & D. They include improvement<br />
in yield and quality, efficacy, improvement of<br />
processes and development of new processes<br />
with validation studies.<br />
2. Benefits derived:<br />
Benefits derived are introduction of new products,<br />
improvement in the yield and quality, safety and<br />
efficacy of products, cost reduction of products<br />
and processes without affecting the quality of the<br />
products and process efficacy. Our R & D Centre<br />
is recognised by D.S.I.R., Ministry of Science<br />
and Technology, Government of India.<br />
3. Information regarding technology imported<br />
during the last five years – Nil.<br />
3. Expenditure on R & D:<br />
(` in Million)<br />
2010-11 2009-10<br />
a) Capital<br />
Expenditure 89.98 57.97<br />
b) Revenue<br />
Expenditure 569.20 460.55<br />
c) Total 659.18 518.52<br />
d) R & D<br />
Expenditure as<br />
a percentage of<br />
total turnover 5.30% 4.99%<br />
C. FOREIGN EXCHANGE EARNINGS AND<br />
OUTGO<br />
1. Activities relating to exports; initiatives taken<br />
to increase exports; development of new<br />
export markets for products and services; and<br />
export plans; The Management Discussion and<br />
Analysis report forming a part of the Directors<br />
Report deals with the same.<br />
2. Total foreign exchange earned was ` 3339.26<br />
million and outflow was ` 1558.86 million.<br />
Mumbai<br />
Date: 10 May 2011<br />
For and on behalf of the Board of Directors<br />
Glenn Saldanha<br />
Chairman & Managing Director<br />
36<br />
GLENMARK PHARMACEUTICALS LIMITED





![Formulations [India] â Product List - Glenmark](https://img.yumpu.com/46601329/1/190x245/formulations-india-a-product-list-glenmark.jpg?quality=85)
![Formulation [India] â Product List - Glenmark](https://img.yumpu.com/44013338/1/190x245/formulation-india-a-product-list-glenmark.jpg?quality=85)



![Formulations [India] â Product List - Glenmark](https://img.yumpu.com/35994839/1/190x245/formulations-india-a-product-list-glenmark.jpg?quality=85)

