Download - Glenmark
Download - Glenmark
Download - Glenmark
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
Annexures to the Directors’ Report<br />
ANNEXURE - A<br />
Information under Section 217(1)(e) of the Companies Act, 1956 read with Companies (Disclosure of Particulars in the<br />
Report of the Board of Directors) Rules, 1988 and forming part of the Directors’ Report.<br />
A. CONSERVATION OF ENERGY<br />
Energy Generation Measures Taken<br />
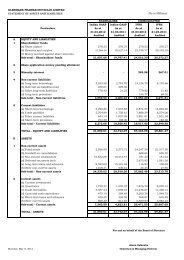
A. Power and Fuel Consumption 2010-11 2009-10<br />
1. Electricity<br />
(a) Purchased<br />
Unit (in ‘000 Kwhrs) 11010.20 7995.98<br />
Total Amount (` in ’000’s) 52398.82 35385.00<br />
Rate/Unit (`) 4.76 4.43<br />
(b) Own Generation<br />
(i) Through Diesel Generator<br />
Unit (in ‘000 Kwhrs) 1686.72 1187.92<br />
Units per Ltr. of Diesel Oil 3.35 3.33<br />
Cost/Unit (`) 11.07 9.51<br />
(ii) Through Steam Turbine/Generator NIL NIL<br />
2. Coal NIL NIL<br />
Qty.<br />
Total Cost<br />
Average Rate<br />
3. Furnace Oil/Light Diesel Oil<br />
Qty. (K. Ltr.) 714.58 52.40<br />
Total Amount (` in ’000’s) 26252.47 2391.40<br />
Average Rate (`/K. Ltr.) 38.19 45.64<br />
4. i) Internal generation<br />
Light Diesel Oil<br />
Qty. (In Ltr. ’000’s) NIL NIL<br />
Total Cost (` in ’000’s) NIL NIL<br />
Rate/Unit (`) NIL NIL<br />
ii) Natural Gas<br />
Qty. (M 3 ’000’s) NIL NIL<br />
Total Cost (` in ’000’s) NIL NIL<br />
Rate/Unit (`) NIL NIL<br />
B. Consumption<br />
The Company manufactures several Drug Formulations in different pack sizes. In view of this, it is impracticable<br />
to apportion the consumption and cost of utilities to each Product/Formulation.<br />
B. TECHNOLOGY ABSORPTION, RESEARCH<br />
& DEVELOPMENT (R&D)<br />
1. Specific areas in which R & D is carried out by<br />
the Company & its subsidiaries and benefits<br />
derived as a result of the same.<br />
Formulation Development:<br />
Pharmaceutical Formulation Development:<br />
Development of formulations as immediate<br />
release, delayed release, enteric release,<br />
sustained release, metered dose inhalers,<br />
dry powder inhalers, nasal sprays, topical,<br />
liquid orals, injectables and various platform<br />
technologies. Formulation development includes<br />
literature survey, pre formulation studies,<br />
formulation development and standardization<br />
of dosage forms for selected drug molecules on<br />
laboratory scale.<br />
R & D has developed the formulations for new<br />
and existing molecules and drug combinations<br />
which includes its standardization and execution<br />
at production site, evaluation of these batches<br />
against reference samples for safety, efficacy<br />
and bio-equivalence.<br />
The following products have been developed during<br />
the financial year 2010-2011.<br />
Dietary supplement<br />
1. Calcium Citrate Malate, Calcitriol and Vitamin<br />
K2- 7 Tablets<br />
Antiviral, Analgesic, Antipyretic, Decongestant,<br />
Antihistamine, Antiasthamatic<br />
1. Levocetirizine Tablets 5 mg<br />
2. Montelukast Sodium Tablets 4/5/10 mg<br />
3. Montelukast Sodium + Levocetirizine<br />
dihydrochloride Tablets (10 + 5) mg<br />
4. Levosalbutamol + Ambroxol Hydrochloride +<br />
Guaifenesin Tables (1+30+100) mg<br />
Anti Diabetic<br />
1. Pioglitazone, Glimepiride and Metformin ER<br />
Tablets (7.5 + 1 + 500) mg<br />
2. Pioglitazone, Glimepiride and Metformin ER<br />
Tablets (7.5 + 2 + 500) mg<br />
34<br />
GLENMARK PHARMACEUTICALS LIMITED





![Formulations [India] â Product List - Glenmark](https://img.yumpu.com/46601329/1/190x245/formulations-india-a-product-list-glenmark.jpg?quality=85)
![Formulation [India] â Product List - Glenmark](https://img.yumpu.com/44013338/1/190x245/formulation-india-a-product-list-glenmark.jpg?quality=85)



![Formulations [India] â Product List - Glenmark](https://img.yumpu.com/35994839/1/190x245/formulations-india-a-product-list-glenmark.jpg?quality=85)

