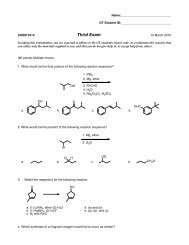
1H 2H 6H 3H
1H 2H 6H 3H
1H 2H 6H 3H
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
TOPIC 7 – REVIEW<br />
Lecture 37<br />
HWeb34<br />
REVIEW<br />
Question 1<br />
Which C 6 H 12 O 2 compound gives the following 1 H NMR spectrum<br />
<strong>6H</strong><br />
<strong>1H</strong><br />
<strong>2H</strong><br />
<strong>3H</strong><br />
6<br />
5<br />
4<br />
3<br />
PPM<br />
2<br />
1<br />
0<br />
O<br />
O<br />
O<br />
O<br />
O<br />
O<br />
O<br />
O<br />
a. 1<br />
b. 2<br />
c. 3<br />
d. 4<br />
1 2 3 4<br />
Question 2<br />
What is the relationship between the following pair of structures<br />
Cl H<br />
H 3 C Cl<br />
H 3 C<br />
C<br />
C<br />
CH 3<br />
H<br />
C<br />
C<br />
H<br />
H Cl<br />
a. They are enantiomers<br />
b. They are diastereomers<br />
c. The are constitutional isomers<br />
d. They are identical<br />
H 3 C<br />
Cl
Question 3<br />
Which one of the following structures represents a different compound from the other three<br />
CH 3<br />
CH 3 H CH 3<br />
CH<br />
H<br />
H<br />
3<br />
H 3 C<br />
CH H 3 C<br />
H<br />
3<br />
H H<br />
CH 3<br />
1 2 3 4<br />
a. 1<br />
b. 2<br />
c. 3<br />
d. 4
Lecture 38<br />
HWeb35<br />
REVIEW<br />
Question 1<br />
Which combination of organic bromide(s) and dicarbonyl compound can be used to prepare the following product (in a<br />
multistep synthesis)<br />
O<br />
O<br />
O<br />
O<br />
O<br />
EtO<br />
OEt EtO<br />
OEt<br />
CH 3 Br PhBr<br />
CH 3 Br PhCH 2 Br<br />
1 2<br />
Ph<br />
O<br />
O<br />
O<br />
O<br />
a. 1<br />
b. 2<br />
c. 3<br />
d. 4<br />
OEt<br />
OEt<br />
CH 3 Br PhBr CH 3 Br PhCH 2 Br<br />
3 4<br />
Question 2<br />
Which combination(s) of organic bromide and carbonyl compound can be used to prepare the following product by reaction<br />
of the Grignard reagent derived from the alkyl bromide with the carbonyl compound<br />
Ph<br />
OH<br />
a. only 1<br />
b. only 1 and 2<br />
c. only 1, 2 and 3<br />
d. 1, 2, 3 and 4<br />
Ph<br />
O<br />
CH 3 Br<br />
O<br />
Ph OCH 3<br />
CH 3 Br<br />
PhBr<br />
1 2 3 4<br />
O<br />
O<br />
PhBr<br />
OCH 3<br />
Question 3<br />
Which combination(s) of alkyl bromide and carbonyl compound can be used to prepare the following product by reaction of<br />
the Wittig reagent derived from the alkyl bromide with the carbonyl compound<br />
CH 3 CH 2 Br<br />
O<br />
CH 3 CH 2 Br<br />
CH 3 CH 2 CH 2 Br CH 3 CH 2 CHBrCH 3<br />
O<br />
O<br />
O<br />
a. only 1 and 2<br />
b. only 2 and 4<br />
c. only 2 and 3<br />
d. only 1, 2 and 3<br />
1 2 3 4
Lecture 39<br />
HWeb36<br />
REVIEW<br />
Question 1<br />
What is the major organic product of the following sequence of reactions<br />
CH 3<br />
O<br />
O<br />
OH<br />
1. NaH<br />
2.<br />
Br<br />
Δ<br />
1. NaH<br />
2. CH 3 I<br />
1 2<br />
CH 3<br />
CH 3<br />
O<br />
a. 1<br />
b. 2<br />
c. 3<br />
d. 4<br />
3 4<br />
Question 2<br />
Which combination of carbonyl compound and amine can be used to prepare the following product by reductive amination<br />
N<br />
(CH 3 ) 2 CO<br />
CHO<br />
CHO<br />
(CH 3 ) 2 CO<br />
NHCH 3<br />
(CH 3 ) 2 CHNHCH 3<br />
(CH 2 ) 2 CHNH 2<br />
NH 2<br />
a. only 1<br />
b. only 3<br />
c. only 1 and 3<br />
d. only 2 and 4<br />
1 2 3 4<br />
Question 3<br />
What is the major organic product obtained from the following sequence of reactions<br />
HNO 3<br />
CH 3<br />
1. NaNO 2<br />
H 2 SO 4<br />
CH 3<br />
Sn, HCl<br />
HCl<br />
2. H 2 O<br />
1. NaH<br />
2. CH 3 I<br />
CH 3 CH 3<br />
CH 3<br />
CH 3<br />
OH<br />
CH 3<br />
OH<br />
a. 1<br />
b. 2<br />
c. 3<br />
d. 4<br />
OCH 3<br />
OH<br />
CH 3<br />
1 2 3 4