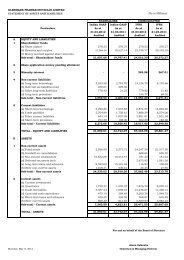
SUMMARY FINANCIAL INFORMATION The selected audited Income Statement and Balance Sheet data for the years ended March 31, 2007, March 31, 2008 and March 31, 2009 set forth below have been derived from the Company’s audited consolidated financial statements and schedules thereto for the years ended March 31, 2007, March 31, 2008 and March 31, 2009, which have been prepared in accordance with Indian GAAP as applicable at the time of their initial preparation and have been audited by Price Waterhouse, Chartered Accountants, the Company’s independent statutory auditor. Consolidated Balance Sheet as at March 31, 2007, March 31, 2008 and March 31, 2009 I. SOURCES OF FUNDS As at March 31, 2007 As at March 31, 2008 As at March 31, 2009 Rs. In ('000s) Rs. In ('000s) Rs. In ('000s) 1. SHAREHOLDERS' FUNDS a) Capital 240,116 248,726 250,520 b) Reserves and Surplus 6,623,534 14,930,003 15,731,044 6,863,650 15,178,729 15,981,564 2. MINORITY INTEREST - 14,796 31,552 3. LOAN FUNDS a) Secured Loans 1,749,306 1,961,282 3,826,548 b) Unsecured Loans 7,617,757 7,948,104 17,116,917 9,367,063 9,909,386 20,943,465 4. Deferred Tax Liability 812,690 1,145,547 1,054,748 Less: Deferred Tax Asset 92,698 200,025 485,489 719,992 945,522 569,259 16,950,705 26,048,433 37,525,840 II. APPLICATION OF FUNDS 1. FIXED ASSETS a) Gross Block 7,095,490 11,241,021 18,385,786 b) Less : Depreciation 1,165,094 2,055,881 2,723,341 c) Net Block 5,930,396 9,185,140 15,662,445 d) Capital Work-in-progress 2,173,888 3,372,287 5,454,080 8,104,284 12,557,427 21,116,525 2. INVESTMENTS 187,237 188,171 181,229 3. CURRENT ASSETS, LOANS AND ADVANCES a) Inventories 2,697,092 4,007,391 6,302,253 b) Sundry Debtors 5,711,645 8,068,517 9,553,428 c) Cash and Bank Balances 1,057,549 1,565,069 714,823 d) Loans and Advances 1,588,046 2,869,032 4,220,877 11,054,332 16,510,009 20,791,381 Less : CURRENT LIABILITIES AND PROVISIONS a) Current Liabilities 2,328,566 3,029,590 4,398,904 b) Provisions 66,582 177,584 164,391 2,395,148 3,207,174 4,563,295 NET CURRENT ASSETS 8,659,184 13,302,835 16,228,086 xv
As at March 31, 2007 As at March 31, 2008 As at March 31, 2009 Rs. In ('000s) Rs. In ('000s) Rs. In ('000s) TOTAL 16,950,705 26,048,433 37,525,840 Consolidated Profit and Loss Account for the year ended March 31, 2007, March 31, 2008 and March 31, 2009 Year ended March 31, 2007 Year ended March 31, 2008 Year ended March 31 ,2009 Rs. In ('000s) Rs. In ('000s) Rs. In ('000s) INCOME Sales & Operating Income 12,515,336 20,092,005 21,160,332 Other income 156,992 458,202 1,740,116 12,672,328 20,550,207 22,900,448 EXPENDITURE Cost of Sales 4,574,712 6,757,996 8,750,997 Selling and Operating Expenses 3,245,159 4,571,018 6,976,794 Depreciation/Amortisation 422,589 716,795 1,026,827 Interest (net) 384,076 631,680 1,404,766 Research and Development Expenses 432,609 757,730 882,703 9,059,145 13,435,219 19,042,087 PROFIT BEFORE TAX AND EXCEPTIONAL ITEMS 3,613,183 7,114,988 3,858,361 Exceptional Item - - 1,169,548 PROFIT BEFORE TAX 3,613,183 7,114,988 2,688,813 Provision for Taxation - Current Year 329,984 857,114 651,299 - Mat Credit (Entitlment)/ Utilisation (181,219) (347,472) 395,278 - Deferred Tax 303,700 199,223 (383,148) - Fringe Benefit Tax 39,731 85,000 81,373 - Prior Period Tax 20,387 - 9,282 NET PROFIT AFTER TAX BEFORE MINORITY 3,100,600 6,321,123 1,934,729 INTEREST Share of (profit)/loss transfer to Minority - 204 (18,092) NET PROFIT AFTER TAX & MINORITY INTEREST 3,100,600 6,321,327 1,916,637 Balance Profit Brought Forward 2,035,295 4,678,920 10,276,665 NET PROFIT AVAILABLE FOR APPROPRIATION 5,135,895 11,000,247 12,193,302 Dividend on Preference Shares 6,942 - - Tax on Dividend on Preference Shares 974 - - Interim Dividend on Equity Shares 95,756 171,545 - Tax on Interim Dividend on Equity Shares 13,430 29,154 - Proposed Dividend on Equity Shares - - 100,208 Tax on Proposed Dividend on Equity Shares - - 17,030 Transfer to Capital Redemption Reserve 200,000 - - Transfer to Foreign Currency Monetary Item Translation - - 366,121 Difference Account Transfer to General Reserve 140,000 522,883 494,490 BALANCE CARRIED TO BALANCE SHEET 4,678,793 10,276,665 11,215,453 Earnings Per Share (Rs.) Basic 25.98 25.84 7.7 Diluted 23.12 24.96 7.5 Face Value per Share 2.00 1.00 1.0 xvi
- Page 1 and 2: Preliminary Placement Document Not
- Page 3 and 4: NOTICE TO INVESTORS The Company acc
- Page 5 and 6: the Issue and are in no way acting
- Page 7 and 8: liable for your decision to partici
- Page 9 and 10: ENFORCEMENT OF CIVIL LIABILITIES Th
- Page 11 and 12: FORWARD LOOKING STATEMENTS All stat
- Page 13 and 14: Term Description Director(s) Direct
- Page 15 and 16: Term NBE NCE NDDS NPPA OTC PCB TRIP
- Page 17 and 18: Use of Proceeds Lock-up Risk Factor
- Page 19 and 20: jurisdictions including Thailand, E
- Page 21 and 22: with commercialisation of Eszopiclo
- Page 23: of the patent and Paragraph IV fili
- Page 27 and 28: If our research and development eff
- Page 29 and 30: consumer acceptance, we may be requ
- Page 31 and 32: Our performance is highly dependent
- Page 33 and 34: portfolio of products that are prom
- Page 35 and 36: esults. Historically, in addition t
- Page 37 and 38: equirements for the above raw mater
- Page 39 and 40: and approvals may result in their e
- Page 41 and 42: plants to our customers located glo
- Page 43 and 44: dividends is subject to the discret
- Page 45 and 46: It may not be possible for you to e
- Page 47 and 48: In addition, the Indian stock marke
- Page 49 and 50: MARKET PRICE INFORMATION The Compan
- Page 51 and 52: (in Rs. million) Period BSE NSE Mar
- Page 53 and 54: The following table shows, as at Ma
- Page 55 and 56: Under current Indian tax laws, divi
- Page 57 and 58: Our Specialty Business is operated
- Page 59 and 60: · devaluation of currencies in key
- Page 61 and 62: Balance Sheet date there is an indi
- Page 63 and 64: Results of Operations The following
- Page 65 and 66: Other Income The total income from
- Page 67 and 68: Provision for Taxation Our provisio
- Page 69 and 70: investments. The Group has financed
- Page 71 and 72: Related Party Transactions The Grou
- Page 73 and 74: Foreign Currency Exchange Rate Risk
- Page 75 and 76:
World Pharmaceutical Market 2008 Sh
- Page 77 and 78:
examined to determine whether any o
- Page 79 and 80:
Further, there are promising opport
- Page 81 and 82:
as patents expire and the shifting
- Page 83 and 84:
jurisdictions including Thailand, E
- Page 85 and 86:
with commercialisation of Eszopiclo
- Page 87 and 88:
of the patent and Paragraph IV fili
- Page 89 and 90:
Our India branded generics business
- Page 91 and 92:
Asia Pacific region, we have filed
- Page 93 and 94:
pursuant to the collaboration. Purs
- Page 95 and 96:
North America / United States Formu
- Page 97 and 98:
other APIs include products ranging
- Page 99 and 100:
iological matrix. These validated a
- Page 101 and 102:
Properties Our properties are used
- Page 103 and 104:
Entity Jurisdiction Shareholding Ac
- Page 105 and 106:
procedures involve obtaining a seri
- Page 107 and 108:
Name, DIN, Designation, Term and Ad
- Page 109 and 110:
Name, DIN, Designation, Term and Ad
- Page 111 and 112:
degrees from Royal Institute of Pub
- Page 113 and 114:
shall not be paid remuneration exce
- Page 115 and 116:
Achin Gupta, 33, is the vice presid
- Page 117 and 118:
Compensation Committee The Compensa
- Page 119 and 120:
PRINCIPAL SHAREHOLDERS The Company
- Page 121 and 122:
S. No Name of the Shareholder No. o
- Page 123 and 124:
Issue Procedure 1. The Company and
- Page 125 and 126:
No Allotment shall be made pursuant
- Page 127 and 128:
Name: Citigroup Global Markets Indi
- Page 129 and 130:
2. In accordance with the SEBI Regu
- Page 131 and 132:
PLACEMENT Memorandum of Understandi
- Page 133 and 134:
SELLING RESTRICTIONS The distributi
- Page 135 and 136:
a) made by an investment firm, bank
- Page 137 and 138:
TRANSFER RESTRICTIONS Allottees are
- Page 139 and 140:
The exceptions to this rule are for
- Page 141 and 142:
company is subject to continuing di
- Page 143 and 144:
case may be. Such company in turn i
- Page 145 and 146:
entering into an agreement for the
- Page 147 and 148:
two depositories that provide elect
- Page 149 and 150:
the shareholders entitled to the pa
- Page 151 and 152:
available for dividends or out of t
- Page 153 and 154:
Annual Report and Financial Results
- Page 155 and 156:
shareholder or repayment of any ter
- Page 157 and 158:
6. Under section 35 of the Income T
- Page 159 and 160:
7. Under section 111A of the Income
- Page 161 and 162:
1. In terms of section 10(34) of th
- Page 163 and 164:
LEGAL PROCEEDINGS Except as disclos
- Page 165 and 166:
dated April 21, 2004, and the Compa
- Page 167 and 168:
2002 in 2048/S/98) dated September
- Page 169 and 170:
6. The Assessing Officer had held t
- Page 171 and 172:
making payment within requisite tim
- Page 173 and 174:
FINANCIAL STATEMENTS GLENMARK PHARM
- Page 175 and 176:
For and on behalf of the Board of D
- Page 177 and 178:
This is the Consolidated Profit and
- Page 179 and 180:
Year ended 31st March,2009 Year end
- Page 181 and 182:
As at 31st March,2009 As at 31st Ma
- Page 183 and 184:
100 each fully paid up of Marksans
- Page 185 and 186:
14. PROVISIONS As at 31st March,200
- Page 187 and 188:
As on 31st March, 2008 Acquisition
- Page 189 and 190:
Year ended 31st March,2009 Year end
- Page 191 and 192:
GLENMARK PHARMACEUTICALS LIMITED SC
- Page 193 and 194:
(a) (b) (c) (d) In respect of Subsi
- Page 195 and 196:
ix) Revenue Recognition The Group r
- Page 197 and 198:
For basic earnings per share 250,02
- Page 199 and 200:
In accordance with the requirements
- Page 201 and 202:
(i) (ii) (iii) Change in Defined Be
- Page 203 and 204:
The Company has opted to follow the
- Page 205 and 206:
GLENMARK PHARMACEUTICALS LIMITED AU
- Page 207 and 208:
This is the Consolidated Balance Sh
- Page 209 and 210:
Partha Ghosh Glenn Saldanha Rajesh
- Page 211 and 212:
Year ended 31st March,2008 Year end
- Page 213 and 214:
As at 31st March,2008 As at 31st Ma
- Page 215 and 216:
(i) (ii) (iii) Convertible at the o
- Page 217 and 218:
As at 31st March,2008 As at 31st Ma
- Page 219 and 220:
GLENMARK PHARMACEUTICALS LIMITED SC
- Page 221 and 222:
GLENMARK PHARMACEUTICALS LIMITED SC
- Page 223 and 224:
Year ended 31st March,2008 Year end
- Page 225 and 226:
2) SIGNIFICANT ACCOUNTING POLICIES
- Page 227 and 228:
v) Foreign Currency Transactions (a
- Page 229 and 230:
4) CONTINGENT LIABILITIES NOT PROVI
- Page 231 and 232:
a) Key management personnel Mr. Gra
- Page 233 and 234:
Rs. In ('000s) 31st Mar, 2008 31st
- Page 235 and 236:
GLENMARK PHARMACEUTICALS LIMITED Au
- Page 237 and 238:
Schedules referred to above and not
- Page 239 and 240:
Schedules referred to above and not
- Page 241 and 242:
Year ended 31st March,2007 Year end
- Page 243 and 244:
2. RESERVES AND SURPLUS Note As at
- Page 245 and 246:
B) The Company had issued 20,000 Ze
- Page 247 and 248:
As at 31st March,2007 Rs. In ('000s
- Page 249 and 250:
GLENMARK PHARMACEUTICALS LIMITED SC
- Page 251 and 252:
GLENMARK PHARMACEUTICALS LIMITED SC
- Page 253 and 254:
Year ended 31st March,2007 Year end
- Page 255 and 256:
Revenues and expenses are translate
- Page 257 and 258:
consumed, labour and manufacturing
- Page 259 and 260:
3) CONTINGENT LIABILITIES NOT PROVI
- Page 261 and 262:
6) RELATED PARTY DISCLOSURES a) Rel
- Page 263 and 264:
(ii) The lease income of Rs.Nil (20
- Page 265 and 266:
7. Glenmark Pharmaceuticals limited
- Page 267 and 268:
ii) To the Foreign Currency Monetar
- Page 269:
ISSUER GLENMARK PHARMACEUTICALS LIM





![Formulations [India] â Product List - Glenmark](https://img.yumpu.com/46601329/1/190x245/formulations-india-a-product-list-glenmark.jpg?quality=85)
![Formulation [India] â Product List - Glenmark](https://img.yumpu.com/44013338/1/190x245/formulation-india-a-product-list-glenmark.jpg?quality=85)




![Formulations [India] â Product List - Glenmark](https://img.yumpu.com/35994839/1/190x245/formulations-india-a-product-list-glenmark.jpg?quality=85)

