PYRUVATE KINASE - Toyobo
PYRUVATE KINASE - Toyobo
PYRUVATE KINASE - Toyobo
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
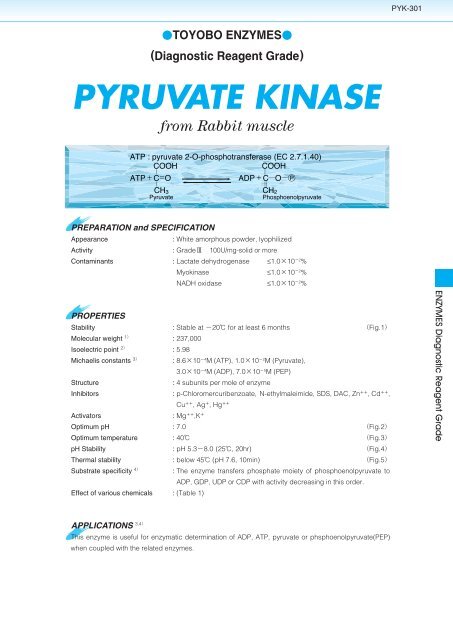
PYK-301<br />
TOYOBO ENZYMES<br />
(Diagnostic Reagent Grade)<br />
<strong>PYRUVATE</strong> <strong>KINASE</strong><br />
from Rabbit muscle<br />
ATP : pyruvate 2-O-phosphotransferase (EC 2.7.1.40)<br />
COOH<br />
ATPCO<br />
<br />
CH 3<br />
Pyruvate<br />
<br />
COOH<br />
ADPCO P<br />
<br />
<br />
CH 2<br />
Phosphoenolpyruvate<br />
PREPARATION and SPECIFICATION<br />
Appearance<br />
: White amorphous powder, lyophilized<br />
Activity<br />
: Grade100U/mg-solid or more<br />
Contaminants : Lactate dehydrogenase ≤1.010 2 %<br />
Myokinase ≤1.010 2 %<br />
NADH oxidase ≤1.010 2 %<br />
PROPERTIES<br />
Stability : Stable at 20 for at least 6 months Fig.1<br />
Molecular weight 1 : 237,000<br />
Isoelectric point 2 : 5.98<br />
Michaelis constants 3 : 8.610 4 M (ATP), 1.010 2 M (Pyruvate),<br />
3.010 4 M (ADP), 7.010 5 M (PEP)<br />
Structure<br />
: 4 subunits per mole of enzyme<br />
Inhibitors : p-Chloromercuribenzoate, N-ethylmaleimide, SDS, DAC, Zn ,Cd ,<br />
Cu ,Ag ,Hg <br />
Activators<br />
: Mg ,K <br />
Optimum pH : 7.0 Fig.2<br />
Optimum temperature : 40 Fig.3<br />
pH Stability : pH 5.38.0 (25, 20hr) Fig.4<br />
Thermal stability : below 45 (pH 7.6, 10min) Fig.5<br />
Substrate specificity 4 : The enzyme transfers phosphate moiety of phosphoenolpyruvate to<br />
ADP, GDP, UDP or CDP with activity decreasing in this order.<br />
Effect of various chemicals : (Table 1)<br />
APPLICATIONS 3,4<br />
This enzyme is useful for enzymatic determination of ADP, ATP, pyruvate or phsphoenolpyruvate(PEP)<br />
when coupled with the related enzymes.
PYK-301<br />
ASSAY<br />
Principle:<br />
PEPADP<br />
pyruvate kinase<br />
PyruvateNADHH <br />
PyruvateATP<br />
lactate dehydrogenase<br />
LactateNAD <br />
The disappearance of NADH is measured at 340nm by spectrophotometry.<br />
Unit definition:<br />
One unit causes the oxidation of one micromole of NADH per minute under the conditions described below.<br />
Method:<br />
Reagents<br />
A. Triethanolamine buffer, pH 7.60.1M containing 11.5mM KCl and 3.0mM MgCl 2<br />
B. ADP solution<br />
140mMDissolve 264mg of ADPNa 2 (MW471.2) in ca.2ml of H 2 O, adjust pH<br />
to ca.7.0 with 1N KOH and fill up to 4.0ml with H 2 O(Should be prepared fresh)<br />
C. PEP solution<br />
16mMDissolve 3.3mg of PEPK (MW206.2) in 1.0ml of H 2 O(Should be<br />
prepared fresh)<br />
D. NADH solution<br />
6.0mMDissolve 4.25mg of NADHNa 2 (MW709.4) in 1.0ml of H 2 O<br />
(Should be prepared fresh)<br />
E. LDH solution<br />
ca.45U/mlDilute lactate dehydrogenase (ammonium sulfate suspension,<br />
Grade from <strong>Toyobo</strong>) to ca.45U/ml with 0.1M triethanolamine buffer, pH 7.6<br />
containing 0.3% of BSA<br />
F. Enzyme diluent<br />
0.1M Triethanolamine buffer, pH 7.6 containing 0.3% of BSA<br />
Procedure<br />
1. Prepare the following reaction mixture in a cuvette (d1.0cm)<br />
Concentration in assay mixture<br />
and equilibrate at 25 for about 5 minutes.<br />
Triethanolamine buffer 90 mM<br />
2.60ml Buffer solution (A) MgCl 2<br />
2.6 mM<br />
0.10ml ADP solution (B) KCl<br />
9.8 mM<br />
ADP<br />
4.6 mM<br />
0.10ml PEP solution (C)<br />
PEP<br />
0.52mM<br />
0.10ml NADH solution (D)<br />
NADH<br />
0.20mM<br />
0.10ml LDH solution (E)<br />
LDH<br />
ca.1.5 U/ml<br />
2. Add 0.05ml of the enzyme solution and mix by gentle inversion.<br />
3. Record the decrease in optical density at 340nm against water for 3 to 4 minutes in a spectrophotometer<br />
thermostated at 25, and calculate the OD per minute from the initial linear portion of the curve (OD<br />
test).<br />
At the same time, measure the blank rate (OD blank) by using the same method as the test except that<br />
the enzyme diluent (F) is added instead of the enzyme solution.<br />
<br />
Dissolve the enzyme preparation in ice-cold enzyme diluent (F) and dilute to 0.251.0U/ml with the same<br />
buffer and store on ice.<br />
Calculation<br />
Activity can be calculated by using the following formula<br />
OD/min (OD testOD blank)Vtdf<br />
Volume activity (U/ml) <br />
6.221.0Vs<br />
Weight activity (U/mg)(U/ml)1/C<br />
OD/min9.807df<br />
Vt<br />
Vs<br />
Total volume (3.05ml)<br />
Sample volume (0.05ml)<br />
6.22 Millimolar extinction coefficient of NADH at 340nm (F/micromole)<br />
1.0 Light path length (cm)<br />
df<br />
C<br />
Dilution factor<br />
Enzyme concentration in dissolution (c mg/ml)<br />
REFERENCES<br />
1) G.L.Cotton, P.F.Hollenberg and M.J.Coon; J.Biol.Chem,244, 1481 (1969)<br />
2) A.Morawiecki; Arch.Immunl.Terap.Doswiadczalnej, 8, 243 (1960)<br />
3) J.T.McQuate and M.F.Utter; J.Biol.Chem.,234, 2151 (1959)<br />
4) F.J.Kayne; The Enzyme, A, 3rd Ed.,p353,Academic Press,New York (1973)
PYK-301<br />
Table 1. Effect of Various Chemicals on Pyruvate kinase<br />
[The enzyme dissolved in 0.1M triethanolamine buffer, pH 7.6 (25U/ml) containing 0.3% of BSA was incubated<br />
with each chemical at 25 for 1hr.<br />
Chemical<br />
Concn.(mM)<br />
Residual<br />
activity<br />
None 100%<br />
Metal salt 2.0<br />
CaCl 2 97<br />
Ba(OAc) 2 98<br />
FeCl 2 87<br />
CoCl 2 73<br />
MnCl 2 100<br />
ZnSO 4<br />
0.8<br />
Cd(OAc) 2 0<br />
NiCl 2 90<br />
CuSO 4<br />
Pb(OAc) 2<br />
0.4<br />
27<br />
AgNO 3 0<br />
HgCl 2 0<br />
2-Mercaptoethanol 2.0 94<br />
PCMB 0.1 12<br />
MIA 2.0 56<br />
Chemical<br />
Concn.(mM)<br />
Residual<br />
activity<br />
NEM 2.0 0.2<br />
IAA 2.0 24<br />
Hydroxylamine 2.0 94<br />
EDTA 5.0 100<br />
,-Dipyridyl 2.0 89<br />
o-Phenanthroline 2.0 94<br />
Borate 50 102<br />
NaF 2.0 100<br />
NaN 3 20 95<br />
TritonX-100 0.10% 97<br />
Brij 35 0.10% 95<br />
Tween 20 0.10% 95<br />
Span 20 0.10% 95<br />
Na-cholate 0.10% 90<br />
SDS 0.05%<br />
0.08<br />
DAC 0.05% 8<br />
Ac, CH3CO; PCMB, p-Chloromercuribenzoate; MIA, Monoiodoacetate; EDTA, Ethylenediaminetetraacetate; IAA,<br />
Iodoacetamide; NEM, N-Ethylmaleimide; SDS, Sodium dodecyl sulfate; DAC, Dimethyl-benzyl-alkyl-ammonium<br />
chloride.<br />
100<br />
100 100<br />
Residual Activity,%<br />
50<br />
-20<br />
4<br />
25<br />
35<br />
Relative Activity<br />
50<br />
Residual Activity,%<br />
50<br />
0 1 2<br />
3<br />
6<br />
0<br />
5<br />
6<br />
7<br />
8<br />
9<br />
0<br />
4<br />
5<br />
6<br />
7<br />
8 9<br />
(weeks) Period(months)<br />
Fig.1. Stability (Powder form)<br />
kept under dry conditions<br />
pH<br />
Fig.2. pH-Activity<br />
25 in the following buffer solution:<br />
,Britton-Robinson buffer;<br />
,0.1M triethanolamine buffrer<br />
pH<br />
Fig.4. pH-Stability<br />
25 20hr-treatment with the follwing<br />
buffer solution:,Britton-<br />
Robinson buffer;,0.1M<br />
triethanolamine buffrer<br />
100<br />
100<br />
Relative Activity<br />
50<br />
Residual Activity, %<br />
50<br />
0<br />
20<br />
30<br />
40<br />
50<br />
60<br />
0<br />
20<br />
30<br />
40<br />
50<br />
60<br />
Temperature, <br />
Fig.3. Temperature activity<br />
in 0.1M triethanolamine buffer,<br />
pH7.6<br />
Temperature, <br />
Fig.5. Thermal stability<br />
10min-treatment with 0.1M triethanolamine<br />
buffer, pH7.6
PYK-301<br />
<br />
<br />
pyruvate kinase<br />
<br />
<br />
lactate dehydrogenase<br />
<br />
<br />
<br />
<br />
<br />
<br />
<br />
<br />
2 <br />
<br />
2<br />
<br />
P <br />
P<br />
<br />
<br />
P<br />
<br />
<br />
2<br />
P<br />
<br />
<br />
<br />
P<br />
<br />
<br />
P<br />
<br />
<br />
<br />
<br />
P <br />
P <br />
P <br />
P <br />
P <br />
P<br />
<br />
<br />
<br />
<br />
<br />
P<br />
<br />
<br />
P<br />
P<br />
P <br />
<br />
P <br />
<br />
<br />
<br />
P