Glenmark cover28.06.04.qxd
Glenmark cover28.06.04.qxd
Glenmark cover28.06.04.qxd
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
Global.<br />
Research focused.<br />
Integrated.<br />
<strong>Glenmark</strong> Pharmaceuticals Limited Annual Report 2003-04
Our vision<br />
To emerge as a leading<br />
integrated research-based global<br />
pharmaceutical company.
Financial highlights<br />
Rs/million<br />
Year March 2000 March 2001 March 2002 March 2003 March 2004<br />
Turnover 1453.40 1907.80 2607.50 3336.40 3806.60<br />
Other income 154.50 36.50 60.70 42.80 34.57<br />
PBIDT 308.20 320.30 502.10 645.40 734.88<br />
Interest 10.40 83.20 129.10 106.90 100.57<br />
Depreciation 11.10 51.00 88.20 103.30 108.89<br />
PBT 286.70 186.10 284.80 435.20 525.42<br />
Tax 69.90 15.20 57.00 103.30 105.38<br />
PAT 216.80 170.70 227.80 331.90 420.04<br />
Turnover 2003-04<br />
International<br />
business 12.3%<br />
Active<br />
Pharmaceutical<br />
ingrediants 11.7%<br />
Domestic<br />
formulations<br />
76%
02 Global. Research-focused. Integrated.<br />
10 Highlights, 2003-04<br />
11 Objectives, 2004-05<br />
12 Interview with the CEO<br />
16 Business divisional analysis<br />
18 Domestic formulations<br />
22 Active pharmaceuticals ingredients<br />
26 International operations<br />
Contents<br />
30 Research and development<br />
37 Management’s Discussion and Analysis<br />
43 Risk management<br />
45 Four-year financial summary<br />
47 Ratios<br />
49 Profile of Directors<br />
50 Directors’ Report<br />
61 Report on Corporate Governance<br />
71 Auditor’s Report<br />
74 The financial statements<br />
1
2<br />
Global. Research-focused. Integrated.
Objective<br />
To make the company<br />
more competitive and<br />
valuable in the global<br />
pharmaceutical industry.<br />
Tenure<br />
The long-term<br />
and post-GATT<br />
(post-2005)<br />
scenario.<br />
Driver<br />
A global market<br />
strategy, a strong<br />
research focus and an<br />
integrated business<br />
model for risk-mitigating<br />
delivery.<br />
3
global<br />
Intensifying competition is the<br />
principal characteristic of the<br />
evolving international<br />
pharmaceutical industry.<br />
This is the nature of the industry’s<br />
transition:<br />
• Companies are becoming more<br />
global in their factor and product<br />
market focus even as geographies are<br />
shrinking,<br />
• The competitive landscape in India<br />
is changing with an impending<br />
respect for product patents forcing<br />
companies to look externally for<br />
avenues of growth, and<br />
• A large number of companies are<br />
pursuing product innovation in the<br />
pursuit of long-term competitiveness.<br />
There is an underlying reason why<br />
globalisation is inevitable for<br />
sustainable growth. The large volume<br />
generic molecules going off-patent<br />
are based in the developed markets.<br />
This is a vast segment: the estimated<br />
size of the US market alone is US $<br />
40 billion while Europe and Japan are<br />
several times the size of the Indian<br />
market. Even the developing markets<br />
of Asia, Africa and Latin America are<br />
worth several billion US dollars.<br />
These markets – large or rapidly<br />
growing – don’t just offer the<br />
opportunity of increasing revenue.<br />
They provide the prospect of<br />
enhanced margins and sustainable<br />
profit growth to all those companies<br />
that intend to leverage the low-cost<br />
Indian manufacturing advantage.<br />
The benefit is indeed reciprocal.<br />
Global players stationed in regulated<br />
international markets also need to<br />
explore win-win alliances with Indian<br />
players. For a good reason: they need<br />
to replace their pipeline of patentprotected<br />
drugs and / or build a<br />
basket of off-patent drugs for the<br />
generic market by outsourcing from<br />
low-cost destinations like India. Going<br />
further, they need to prospect for<br />
potential partners in the area of<br />
formulation manufacture, bulk drug<br />
4
production and contract research. Of<br />
late, some of these pharmaceutical<br />
majors have also begun to look<br />
towards India for collaborators in joint<br />
IPR development with the objective<br />
to seize a competitive advantage in<br />
their existing markets at a low cost.<br />
Over the years, <strong>Glenmark</strong> has<br />
strengthened its proactive<br />
international business strategy to<br />
capitalise on these emerging<br />
opportunities through the following<br />
initiatives:<br />
• Its regulated market strategy is<br />
being addressed through its US and<br />
UK presence with the latter being<br />
viewed as the entry point into the EU<br />
region.<br />
• It has entered into an alliance with<br />
KV Pharmaceuticals to market its<br />
products in the US markets.<br />
• It has developed its brand in the<br />
Caribbean Islands, Central America,<br />
Africa and South East Asia.<br />
• A recent acquisition of Klinger Labs<br />
in Brazil will help the company jumpstart<br />
its initiatives in the Latin<br />
American markets.<br />
• It will continue to build front-end<br />
and customer-facing distribution<br />
systems in the international<br />
geographies where it intends to<br />
market products under its own brand<br />
name.<br />
<strong>Glenmark</strong>’s global-focused business<br />
model has been directed to fulfil a<br />
number of corporate objectives:<br />
increasing revenue and enhanced<br />
profitability are among them. As<br />
these strategies translate into<br />
revenues, the company will<br />
progressively substitute its<br />
dependence on the Indian geography<br />
as its principal growth driver with a<br />
more diverse international presence,<br />
leading to a sustainable increase in<br />
shareholder value.<br />
5
Research<br />
focused<br />
An ongoing research and development<br />
focus is becoming an increasingly<br />
important driver of success in the<br />
pharmaceutical industry.<br />
A cutting-edge R&D initiative is<br />
helping companies identify niches and<br />
build differentiated products. This is<br />
helping future-proof their presence in<br />
an increasingly competitive global<br />
environment.<br />
To understand the growing<br />
importance of research, it is<br />
important to understand the two<br />
levels at which change is<br />
transpiring in the pharmaceutical<br />
industry:<br />
Firstly, innovation and new product<br />
development are becoming<br />
increasingly important in the creation<br />
of new revenue streams.<br />
Secondly, they are leading to the<br />
creation of IPR-rich products. The<br />
more the patented products with<br />
attractive commercial value, the<br />
stronger the long-term competitive<br />
edge, exactly in line with the<br />
requirements of a post-GATT<br />
environment in which companies will<br />
need to possess commercially-viable<br />
proprietary products and technologies<br />
for success.<br />
In response to these changes,<br />
<strong>Glenmark</strong> has drawn out a threepronged<br />
proactive R&D initiative that<br />
covers the following:<br />
• New chemical entity (NCE)<br />
research: The company is targeting<br />
the development of new molecules in<br />
the diabetes, obesity and asthma<br />
6
therapeutic segments. It is working<br />
on baskets of targets and families of<br />
new molecules to enhance the<br />
possibility of success and reduce the<br />
lead-time to market. This research has<br />
shown exciting results and some of<br />
the drug candidates are expected to<br />
move into the clinical trial stage in<br />
2004-05.<br />
• Formulations and new drug delivery<br />
systems: The company is extending<br />
the value of its existing formulation<br />
therapies through the research and<br />
development of patentable drug<br />
delivery systems that would help<br />
create strong differentiated products.<br />
The company has a patented<br />
controlled release technology through<br />
which it is planning to commercialise<br />
2-3 products in 2004-05. In addition,<br />
the company also has teams of<br />
research scientists developing<br />
formulations for all its markets.<br />
These teams are housed across its two<br />
research facilities at Sinnar and<br />
Mahape.<br />
• Strong process chemistry research:<br />
The company reverse engineers bulk<br />
drugs for marketing across various<br />
geographies. The company has filed<br />
patents on ten non-infringing<br />
processes till now.<br />
Over time, these R&D initiatives at<br />
<strong>Glenmark</strong> are expected to translate<br />
into a more valuable product basket,<br />
stronger revenues, enhanced margins<br />
and higher profits.<br />
7
Integrated<br />
Success in a competitive global environment will be<br />
derived from a mature understanding of the diverse<br />
business risks and the creation of a corresponding<br />
risk-mitigated model. <strong>Glenmark</strong>’s integrated business<br />
model allows it to address several markets and<br />
revenue opportunities in parallel and risk-mitigates<br />
the overall delivery of results.<br />
A committed risk management<br />
approach is integral to <strong>Glenmark</strong>: its<br />
senior management is engaged in the<br />
identification and mitigation of<br />
business risk in the pursuit of<br />
enhanced shareholder value.<br />
The risk-management at <strong>Glenmark</strong> is<br />
also comprehensive: it is conducted<br />
from the top of the management<br />
pyramid (de-risking the business<br />
model) to the bottom (de-risking the<br />
individual product baskets). The<br />
approach encompasses an incisive<br />
R&D focus, broad geographic<br />
presence, wider product basket,<br />
superior manufacturing facilities,<br />
quicker regulatory approvals and an<br />
ongoing investment in human<br />
competencies. As a responsible<br />
organisation, the company actively<br />
studies the risk inherent in each of<br />
these diverse aspects, leading to the<br />
formulation of counter-strategies.<br />
The integrated business model of the<br />
company represents its principal risk<br />
management initiative, leading to a<br />
lower cost structure and a direct<br />
control over an increasing number of<br />
points in the value-chain. The<br />
integration translates into revenues<br />
being derived from every point in this<br />
value-chain: from potential licensing<br />
opportunities for new drugs to a<br />
growing presence as a bulk supplier<br />
and formulations player in multiple<br />
markets. Over time, the various<br />
8
constituents of the integration have<br />
dovetailed into an effective riskmitigation<br />
structure:<br />
• The quality demands of the<br />
international markets are being targeted<br />
through world-class manufacturing<br />
facilities that will bear scrutiny under<br />
strict regulated market standards.<br />
• A primary dependence on the<br />
Indian market is being gradually<br />
rationalised (from 90 per cent of<br />
turnover in 2002 to a projected 60 per<br />
cent by 2005).<br />
• There is a concentrated focus on<br />
strengthening the company’s product<br />
basket with new value-added<br />
therapies and product introductions<br />
across more geographies.<br />
• Enhanced quality systems and a<br />
strong regulatory focus are<br />
accelerating product introduction in<br />
various markets and accelerating<br />
DMF and ANDA filings in the<br />
developed markets like the US,<br />
driving revenues.<br />
• The recruitment of local<br />
management in several of its key<br />
markets has resulted in a sound<br />
understanding of the local market<br />
conditions, regulatory requirements<br />
and legal system.<br />
This active management of risk,<br />
combined with rapid revenue<br />
and profit growth, is expected to<br />
enhance shareholder value over the<br />
foreseeable future.<br />
9
Highlights, 2003-04<br />
Revenue 2003-04<br />
11.7<br />
12.3<br />
76<br />
Exports<br />
India, formulations<br />
India, APIs<br />
• Overall revenue and bottom line<br />
grew 14.09 and 26.56 per cent<br />
respectively.<br />
• NCE research gained significant<br />
momentum with positive preclinical<br />
results for two lead<br />
molecules in the Asthma and<br />
Diabetes areas.<br />
• The company entered into a tieup<br />
with Quintiles, a global CRO, to<br />
take its Asthma lead molecule into<br />
clinical trials in UK in July 2004.<br />
• Domestic formulations exhibited<br />
a volume and value growth of 16<br />
per cent and 7.9 per cent<br />
respectively.<br />
• Formulation exports to semiregulated<br />
markets registered<br />
strong value growth and<br />
operations were expanded to<br />
cover 59 semi-regulated country<br />
markets.<br />
• The USA subsidiary entered into a<br />
landmark development and<br />
marketing arrangement for eight<br />
products with KV Pharmaceuticals<br />
in the US, valued at over USD<br />
80 million.<br />
• Steps were initiated to set up<br />
operations in UK and Philippines.<br />
• Steps were initiated to file<br />
formulation dossiers (ANDAs) in the<br />
US in 2004-05.<br />
• A new formulations plant was<br />
commissioned at Goa to meet the<br />
stringent USFDA standards.<br />
• The company’s Brazilian subsidiary<br />
acquired Laboratories Klinger in<br />
Brazil that includes a manufacturing<br />
plant with ANVISA approval.<br />
• Four DMFs were filed (till April<br />
2004).<br />
• The API infrastructure was<br />
strengthened with the expansion<br />
and modernisation at Ankleshwar<br />
and the acquisition of a plant at<br />
Solapur.<br />
• The company tied-up with Apotex<br />
and Eon Labs for the supply of<br />
APIs to the regulated markets.<br />
• The IP portfolio was<br />
strengthened and the company<br />
built a basket of 12 patents on NCE<br />
molecules and 10 non-infringing<br />
process patents.<br />
10
Objectives, 2004-05<br />
• Revenue growth of approximately 35 per cent and a net<br />
profit growth of 80 per cent over 2003-04.<br />
• Commencing commercial operations at the new Goa<br />
facility and obtaining USFDA approval.<br />
• Obtaining USFDA approvals for the Ankhleshwar API<br />
facility and 100 per cent growth in API revenues.<br />
• Developing a robust front-end sales and marketing<br />
network for marketing own-label generics in the USA and<br />
initiating business in countries of the European Union.<br />
Projected 2004-05<br />
35<br />
• Completing Phase I clinical studies for the Asthma lead<br />
molecule and initiating Phase I for the Diabetes lead<br />
molecule.<br />
• An enhanced presence in the semi-regulated markets<br />
(including Latin America) and a 100 per cent growth in<br />
revenues.<br />
• Registering 10-12 ANDAs and 8-10 DMFs.<br />
• The launch of strong molecules in the India formulations<br />
business and the strengthening of flagship brands.<br />
8<br />
Exports<br />
India Formulation<br />
API India<br />
57<br />
11
Interview with the<br />
CEO and MD<br />
“A number of initiatives<br />
that we embarked upon in<br />
2002-03 gained<br />
momentum in 2003-04.”<br />
Glenn Saldanha, Managing Director and<br />
CEO, reviews the company’s<br />
performance in 2003-04.<br />
12
Q<br />
HOW WOULD YOU<br />
RATE THE COMPANY’S<br />
PERFORMANCE IN 2003-04?<br />
The financial year under review was a<br />
favourable one for <strong>Glenmark</strong> not only<br />
because we reported an all-round<br />
increase in our numbers, but because<br />
we strengthened the competitive<br />
ability of the business for the longterm.<br />
A number of initiatives that we<br />
embarked upon in 2002-03 gained<br />
momentum in 2003-04. For instance,<br />
the year represented a step forward<br />
for our international business which<br />
was manifested in our export revenue<br />
growth in excess of 50 per cent. Our<br />
USA subsidiary commenced<br />
operations and we signed a landmark<br />
contract with KV Pharmaceuticals to<br />
market our products in the US. Our<br />
sales in the African, South Asian,<br />
Russian, CIS and Latin American<br />
markets gained momentum. A wider<br />
presence of offices in UK, Philippines,<br />
Malayasia and Vietnam helped us<br />
entrench our international presence.<br />
What was particularly heartening was<br />
that even as we were increasingly<br />
global-focused, our domestic<br />
formulations business grew by about<br />
7.9 percent in value terms,<br />
outperforming the sluggish growth of<br />
the Indian pharmaceutical industry.<br />
The volume growth was even higher<br />
at about 16 per cent. The strong<br />
growth across our businesses helped<br />
our bottom-line grow by 26.56 per<br />
cent to Rs. 420.04 million in 2003-04.<br />
To strengthen our vision of emerging<br />
as an integrated global player, we<br />
commissioned a new USFDAapprovable<br />
facility in addition to the<br />
significant completion of expansions<br />
and upgrades at our other<br />
manufacturing facilities. The benefits<br />
of these activities will be visible over<br />
the foreseeable future.<br />
Q<br />
WHAT WERE THE OTHER<br />
ACHIEVEMENTS?<br />
We embarked on a number of<br />
initiatives to migrate up the value<br />
chain. For instance, we<br />
commissioned a state-of-art<br />
manufacturing facility at Goa to cater<br />
to the stringent regulatory<br />
requirements of the developed<br />
markets. We completed various<br />
modules of expansion and upgrade<br />
programmes at our other<br />
manufacturing facilities. We filed<br />
three DMFs in 2003-04 and have<br />
already filed an additional DMF in<br />
April 2004. We have also filed patents<br />
through non-infringing processes for<br />
manufacturing ten bulk drugs. We are<br />
currently working on 8-10 more DMFs<br />
to be filed during 2004-2005. We<br />
introduced new drugs to counter<br />
declining prices in the Indian market<br />
with enhanced revenues. We<br />
consolidated our domestic formulation<br />
business by strengthening key brands<br />
and deepening our product basket. In<br />
the API business, we enhanced the<br />
capacity of our bulk drug<br />
manufacturing facility at Ankleshwar<br />
(acquired in 2002-03 from GSK<br />
Pharma) to address the growing list of<br />
clients in our developed and<br />
developing markets. We also<br />
completed upgrades at the facility to<br />
comply with USFDA requirements.<br />
Several new products were<br />
introduced to strengthen our API<br />
product and revenue baskets.<br />
Q<br />
HOW DID THE<br />
MANAGEMENT<br />
STRENGTHEN THE<br />
COMPANY’S R&D THRUST?<br />
<strong>Glenmark</strong> continued to focus on two<br />
disease targets – for Asthma/COPD<br />
and Diabetes. The new drug research<br />
initiative has yielded extremely<br />
promising results: we are on track to<br />
enter Phase I clinical trials in the UK<br />
with two new chemical entities<br />
(NCEs) in 2004-05, one in each<br />
therapeutic segment. A tie-up is in<br />
place with a global CRO, Quintiles to<br />
take our Asthma molecule into<br />
Phase I clinical trials in the UK by July<br />
2004. Even though the molecules will<br />
be commercialised only over the next<br />
five years, subject to their success in<br />
the clinical trials, the company will be<br />
able to outlicense or co-develop them<br />
with a global pharmaceutical major<br />
immediately after the early clinical<br />
trials have been completed. The<br />
company has already filed over 12<br />
patents, capitalising on the work done<br />
by the new drug research team.<br />
It would be relevant to explain how<br />
our R&D strategy will de-risk the<br />
13
Risk vs. Reward Value Chain<br />
R&D Investments<br />
Reward<br />
New Drugs<br />
New Drug Research<br />
<strong>Glenmark</strong>'s<br />
Evolution<br />
Regulated<br />
Markets<br />
Semi-regulated Markets<br />
Branded Generics<br />
Specialty<br />
Products<br />
Regulated<br />
Markets<br />
Generics<br />
Novel Delivery Systems<br />
Formulation Development<br />
And<br />
API Process Reverse<br />
Engineering<br />
Semi-regulated<br />
Markets APIs<br />
Risk<br />
organisation: we are concentrating on<br />
more than one therapeutic segment,<br />
multiple targets and a pipeline of drug<br />
compounds with a view to strengthen<br />
our success prospects. We are<br />
actively pursuing research in the area<br />
of novel drug delivery systems<br />
(NDDS) and the ownership of several<br />
patents will constitute an integral part<br />
of our endeavour to differentiate our<br />
products across the domestic and<br />
international markets. Separate teams<br />
are working on formulation<br />
development and API process<br />
research for launch across our various<br />
markets. Therefore, our research<br />
efforts span the entire risk-reward<br />
spectrum in the pharmaceutical chain<br />
and will drive growth as <strong>Glenmark</strong><br />
evolves along the spectrum.<br />
Q<br />
HOW DID THE COMPANY<br />
RESPOND TO THE SLOW<br />
DOWN IN THE INDIAN<br />
FORMULATIONS BUSINESS?<br />
It would be relevant to understand<br />
the reasons behind a slowdown in the<br />
Indian formulations business:<br />
• The Indian market is extremely<br />
competitive with several ‘me-too’<br />
launches in each segment.<br />
• The market has seen severe price<br />
competition; four key products of<br />
<strong>Glenmark</strong> witnessed a severe price<br />
erosion owing to competitive<br />
pressures.<br />
• Opportunities for launching new<br />
products are drying up as India<br />
prepares to usher in a regime with a<br />
greater respect for product patents in<br />
2005.<br />
As a result, while the Indian market<br />
grew by approximately 8 per cent in<br />
volume terms, it translated into a<br />
meagre 5 per cent growth in value.<br />
We expect this sluggish value growth<br />
to sustain for the next 2-3 years as<br />
companies expand their therapeutic<br />
segment coverage in response to the<br />
challenge of 2005 and diminishing<br />
new product options, thereby<br />
increasing competition.<br />
As a future-focused organisation,<br />
<strong>Glenmark</strong> has followed a multipronged<br />
strategy to counter this<br />
impact. Aggressive new product<br />
development is one: we have focused<br />
on the rapid launch of powerful<br />
products like Etoricoxib, Tacrolimus<br />
and Rosuvastatin to replace older<br />
therapies and we are also constantly<br />
evaluating newer/under-leveraged<br />
therapeutic segments to launch new<br />
products. Existing products are seeing<br />
value-addition in the form of<br />
combination therapies and new<br />
delivery platforms. We are confident<br />
that these initiatives will translate into<br />
robust growth across the foreseeable<br />
future.<br />
Concurrent with this, we are<br />
strengthening our existing brands and<br />
marketing divisions and also helping<br />
our sales forces specialise with the<br />
objective of maximising our presence.<br />
We are optimistic that this approach<br />
will create a superior product basket<br />
14
einforced by a stronger market<br />
presence to help us outperform<br />
industry growth consistently across<br />
the long-term.<br />
Q<br />
HOW DOES THE COMPANY<br />
EXPECT TO STRENGTHEN<br />
ITS PRESENCE IN THE API<br />
BUSINESS?<br />
Our presence in the API business will<br />
be strengthened through an ongoing<br />
investment in our manufacturing<br />
facilities, research capabilities and<br />
expansion into newer markets. For<br />
instance, we are expanding and<br />
upgrading our plants for API<br />
manufacture; we are strengthening<br />
our in-house research team with a<br />
view to engineer drugs and accelerate<br />
their introduction; we are filing DMFs<br />
in the US and EU to supply APIs to<br />
companies in these markets.<br />
At <strong>Glenmark</strong>, we view the API<br />
business from three perspectives.<br />
Firstly, API manufacture serves as a<br />
backward integration in our<br />
formulation-centric value-chain, which<br />
helps us rationalise cost and become<br />
increasingly competitive. Secondly,<br />
API products are not intended only for<br />
captive use but are marketed to users<br />
in India and abroad so that they<br />
represent a standalone revenue-driven<br />
business for the company. Thirdly, the<br />
company expects to enter into<br />
partnerships in its API business with<br />
manufacturers of generic formulations<br />
in the developed markets.<br />
Q<br />
WHAT ARE THE<br />
SIGNIFICANT STRENGTHS<br />
OF THE COMPANY?<br />
Our responsiveness to the external<br />
environment, which is reflected in our<br />
ability to understand the transitions in<br />
international markets and implement<br />
a corresponding business strategy<br />
with speed. This has actually<br />
transpired: a few years ago, we were<br />
a formulations company but in<br />
response to a shift in the global<br />
dynamics, we are today adequately<br />
diversified with a growing presence in<br />
APIs, a focused and balanced R&D<br />
initiative supported by a strong, low<br />
cost manufacturing base.<br />
This response has also been evident<br />
in our India-centric business: for<br />
instance, when we noticed the first<br />
signs of increasing competition,<br />
sluggish growth and margin erosion in<br />
the domestic market, we migrated<br />
towards new products, value-added<br />
segments and an ongoing cost<br />
rationalisation. I am pleased that<br />
despite a challenging marketplace, we<br />
reported attractive financial growth<br />
and enhanced shareholder value in<br />
2003-04.<br />
Q<br />
WHAT ARE THE<br />
SIGNIFICANT<br />
OPPORTUNITIES AHEAD OF<br />
THE COMPANY?<br />
The answer is quite simple – a deeper<br />
penetration of the developing and<br />
developed international markets. For<br />
instance, the developed market<br />
opportunity lies largely in the generic<br />
segment in US and the European<br />
Union. The size of this segment in<br />
those geographies has been<br />
estimated at several US billion dollars,<br />
offering significant opportunities for<br />
low cost manufacturers from<br />
countries such as India. As these<br />
markets commoditise, the low cost<br />
advantage will provide us with staying<br />
power and profitability.<br />
We expect to address a large<br />
opportunity in the developing markets<br />
through generic products and branded<br />
products. For instance, we will invest<br />
in a marketing infrastructure across<br />
several important developed and<br />
developing markets so that we may<br />
strengthen financial growth, brand<br />
recognition and enhanced margins.<br />
<strong>Glenmark</strong> faces another significant<br />
opportunity: bringing successful new<br />
chemical entities to markets as patent<br />
protected drugs in combination with<br />
an international partner.<br />
Q<br />
HOW DOES THE COMPANY<br />
EXPECT TO SIGNIFICANTLY<br />
ENHANCE SHAREHOLDER VALUE?<br />
In my mind, value is emerging from<br />
our brand performance, strong R&D<br />
thrust, global diversification and a<br />
robust cash flow. Sustainability will<br />
come from our efforts to build a<br />
deeper presence in our chosen<br />
therapeutic segments as well as an<br />
ongoing expansion in newer<br />
segments and markets. It is the<br />
combination of these two approaches<br />
that will help us create a sustainable<br />
and higher margin business. We are<br />
of the opinion that the GATT<br />
challenge of 2005 is not going to pose<br />
a threat until 2008-10 by which time<br />
our NCE strategy will start delivering<br />
results. As a result, our various<br />
initiatives, our integration and our<br />
active risk management will not only<br />
help us grow our revenues but also<br />
risk-mitigate our profits, resulting in<br />
superior returns to our shareholders.<br />
15
16<br />
Business divisional analysis
Over the years, <strong>Glenmark</strong> has responded to an increasing brand clutter in a<br />
competitive marketplace as well as the challenge of addressing emerging<br />
opportunities by segregating its business into three groups with a distinctive<br />
product-market focus. In 2003-04, these groups comprised:<br />
Domestic market formulations:<br />
This group handles all formulation<br />
sales within India.<br />
Active pharmaceutical<br />
ingredients (APIs):<br />
This group caters to the growing API<br />
needs within the organisation and<br />
diverse customer needs in the Indian<br />
and international geographies. It comarkets<br />
value-added formulations<br />
along with APIs to other<br />
pharmaceutical companies.<br />
International operations:<br />
This group focuses on sales to the<br />
semi-regulated/developing country<br />
markets. It also comprises<br />
subsidiaries that are focussed on the<br />
developed markets.<br />
<strong>Glenmark</strong> Pharmaceuticals Limited<br />
Domestic market<br />
formulations – 76 per<br />
cent of revenues<br />
Active Pharmaceutical<br />
Ingredients – 11.7 per<br />
cent of revenues<br />
International operations<br />
– 12.3 per cent of<br />
revenues<br />
A value-added business model<br />
Over the years, <strong>Glenmark</strong> has created<br />
an integrated value-added business<br />
model. This model extends from API<br />
development at one end to the<br />
marketing of formulations in India and<br />
several international markets at the<br />
other.<br />
This business model is leveraging on<br />
the opportunities by climbing a valuepyramid<br />
spanning API, Generics and<br />
Specialities. As each element within<br />
this pyramid has been prudently<br />
monetised across an increasing<br />
number of markets, the company has<br />
succeeded in progressively improving<br />
performance and potential growth<br />
targets.<br />
Over the last four years, <strong>Glenmark</strong><br />
has also succeeded in transferring its<br />
rich and longstanding Indian<br />
experience to semi-regulated<br />
markets with nearly similar<br />
demographic and regulatory profiles:<br />
Africa, South East Asia and Latin<br />
America. Besides, the company has<br />
created subsidiaries to enter the<br />
high-margin developed markets of<br />
USA and European Union, a<br />
decisive future-proofing initiative.<br />
Recently the company also<br />
acquired a company in Brazil<br />
that will help strengthen<br />
presence in the Latin American<br />
markets.<br />
The following section reviews the<br />
operations and the growth strategy of<br />
each of the company’s marketfocussed<br />
divisions.<br />
17
Group<br />
Domestic<br />
formulations<br />
18
GLENMARK’S DOMESTIC<br />
FORMULATIONS BUSINESS IS ITS<br />
LARGEST REVENUE GENERATOR:<br />
WITH REVENUES OF RS 2892.0<br />
MILLION IN 2003-04 (RS 2743.97<br />
MILLION IN 2002-03). While the<br />
Indian pharmaceutical industry grew<br />
at about 5 per cent, the company’s<br />
domestic formulation revenues grew<br />
7.9 per cent by value and 16 per cent<br />
by volume. <strong>Glenmark</strong> countered a<br />
decline in realisations through the<br />
introduction of new and improved<br />
drugs.<br />
THERAPEUTIC SEGMENTS<br />
The company has segregated its<br />
formulation business into three<br />
divisions - <strong>Glenmark</strong>, Gracewell and<br />
Healtheon - to strengthen its focus on<br />
defined therapeutic segments as well<br />
as associated practitioners/specialists.<br />
<strong>Glenmark</strong> division: It is the<br />
oldest division focused on<br />
gynaecology, physicians and<br />
orthopaedics, handling several<br />
flagship brands of the company.<br />
Gracewell division: Focused on<br />
the dermatology, pain, antibiotics and<br />
respiratory segments.<br />
Healtheon division: This is the<br />
newest division focused on lifestylerelated<br />
segments like diabetes,<br />
cardiovascular and lipid-lowering<br />
drugs.<br />
While dermatology, internal medicine<br />
and paediatrics accounted for the<br />
largest revenue in 2003-04, the<br />
company is also present in several<br />
other segments that present<br />
significant growth opportunities.<br />
Therapeutic segment share in revenues<br />
Per cent<br />
Therapeutic segment 2000 2001 2002 2003 2004<br />
Dermatological 40 36 39 35 33.0<br />
Gynaecological 10 9 10 6 6.4<br />
Internal medicine 24 25 25 26 20.5<br />
Paediatric 21 25 18 19 17.9<br />
ENT 5 4 4 3 3.4<br />
Diabetics 0 1 4 7 7.1<br />
Pain Management 0 0 0 5 9.7<br />
Cardiovascular 0 0 0 0 2.0<br />
Source : MAT ORG Data<br />
Dermatological<br />
Gynaecological<br />
Internal medicine<br />
Paediatric<br />
ENT<br />
Diabetics<br />
Pain Management<br />
Cardiovascular<br />
9.7 2.0<br />
7.1<br />
3.4<br />
17.9<br />
33<br />
6.4<br />
20.5<br />
PERFORMANCE HIGHLIGHTS,<br />
2003-04<br />
• The formulations group reported a<br />
value growth of 7.9 per cent,<br />
outperforming the Indian market<br />
growth rate of five per cent.<br />
• The volume growth of the<br />
formulations group was higher, but its<br />
impact was stunted by a sharp price<br />
decline.<br />
• The domestic formulations business<br />
was ranked number one in its<br />
operating market (comprising<br />
therapeutic segments that<br />
account for 95 per cent of revenues –<br />
as per ORG-MAT March 2004<br />
figures).<br />
• The group established its<br />
dominance in new segments like pain<br />
management through products like<br />
Valedecoxib and brand extensions<br />
(rated by ORG as the best product<br />
launch in the last two years in the<br />
IPM) and also improved its position in<br />
relatively new segments like<br />
diabetology.<br />
• The group strengthened its<br />
presence in the dermatology segment<br />
and launched new molecules in the<br />
anti-platelet, anti-hypertensive and<br />
lipid-lowering segments.<br />
• The group strengthened its field<br />
force to more than 900 market<br />
representatives.<br />
• An improved planning and design of<br />
marketing activities helped rationalise<br />
the group’s marketing spend.<br />
19
DIVISIONAL STRATEGY<br />
<strong>Glenmark</strong>’s attractive growth in the formulations segment<br />
is a result of the sustained introduction of improved drugs<br />
and an entry into new growth segments.<br />
Focus: Even as the group is focused on enhancing its<br />
presence in well-researched therapeutic segments, there is<br />
a continuous endeavour to expand to newer segments<br />
with attractive potential. This has lead to a gradual<br />
diversification of the company’s therapeutic basket over<br />
the last few years. The divisional approach promotes this<br />
focus and helps <strong>Glenmark</strong>’s domestic formulation business<br />
drive rapid recognition in a competitive market.<br />
Domestic formulations<br />
Diversified therapeutic segment portfolio<br />
Internal medicine<br />
Respiratory<br />
Dermatology<br />
Gynaecology<br />
49<br />
5<br />
21<br />
25<br />
33<br />
26<br />
6<br />
7<br />
18<br />
10<br />
Respiratory<br />
Pain management<br />
Diabetes<br />
Gynaecology<br />
Dermatology<br />
Int. medicine<br />
Source : MAT ORG Data<br />
Establishing first-mover advantage: At the company,<br />
the focus is not just in establishing a therapeutic presence;<br />
it is to be the first to launch latest generation drugs and<br />
capture a first-mover’s advantage in these segments.<br />
Some of these first-to-launch products include Telmesartan<br />
(anti-hypertensives), Rosuvastatin, Ezetimibe (cholesterol<br />
lowering), Tacrolimus, Tazoretene and Cilastazol (lipidlowering).<br />
Replacing older therapies: <strong>Glenmark</strong> also focuses on<br />
the replacement of older and inferior molecules with newer<br />
therapies and combination drugs, a strategy more<br />
profitable than the launch of me-too brands. These new<br />
molecules/combination therapies have redefined the<br />
dominant therapy in their segments and led to an<br />
increase in sales.<br />
Market size of molecules launched by <strong>Glenmark</strong><br />
Molecules FY 2004 FY 2003<br />
Rs. Mn Growth per cent Rs. Mn<br />
Valdecoxib oral Solids 85.9 219.6 26.9<br />
Levofloxacin 56.3 40.0 40.2<br />
Esomeprazole 31.4 19.5 26.3<br />
Rosuvastatin 5.8 NA 0.0<br />
Note: ORG Marg MAT March data<br />
20
The company’s revenues in 2003-04 from new molecules (launched in the preceding two years in each case) are detailed<br />
in the table below:<br />
Revenue contribution from new products (
Group<br />
Active<br />
pharmaceutical<br />
ingredients<br />
22
ACTIVE PHARMACEUTICAL<br />
INGREDIENTS, WHICH ARE USED<br />
AS RAW MATERIAL IN THE<br />
MANUFACTURE OF<br />
FORMULATIONS, HAVE OFTEN<br />
BEEN REFERRED AS THE BUILDING<br />
BLOCKS IN THE PHARMACEUTICAL<br />
INDUSTRY.<br />
<strong>Glenmark</strong>’s API business was<br />
developed four years ago in keeping<br />
with its vision of emerging as an<br />
integrated pharmaceutical player. As a<br />
result, the company’s manufacture of<br />
API not only serves its captive<br />
requirement but is also marketed to<br />
third parties within India and abroad.<br />
<strong>Glenmark</strong>’s non-captive API<br />
operations recorded revenues of<br />
Rs 445.3 million in 2003-04 compared<br />
with Rs 286.1 million in 2002-03, a<br />
growth of 55.6 per cent. The<br />
company is leveraging its in-house<br />
capability to enhance value in this<br />
business over the foreseeable future.<br />
Therapeutic segment contribution<br />
Per cent<br />
Therapeutic segment Per cent of Sales in 2003-04<br />
Hypertensives 39<br />
Gastroenterology 21<br />
Pain management 20<br />
Diabetology 6<br />
Dermatology 5<br />
Anti-Viral 3<br />
Anti-Depressant 1<br />
Other 5<br />
Hypertensives<br />
Gastroenterology<br />
Pain management<br />
Diabetology<br />
Dermatology<br />
Anti-Viral<br />
Anti-Depressant<br />
Other<br />
20<br />
31<br />
6<br />
3<br />
5 1 5<br />
39<br />
HIGHLIGHTS, 2003-04<br />
• The company upgraded and<br />
increased the capacity of the API<br />
facility acquired from GSK to address<br />
the growing needs of customers in<br />
the developed and developing<br />
markets. The plant, which is CGMP<br />
and WHO-compliant, is expected to<br />
be awarded a USFDA certification in<br />
the first quarter of 2004-05.<br />
• The company has filed 4 DMFs by<br />
April 2004 and is working on another 8-<br />
10 DMFs to be launched in 2004-05.<br />
• The company’s co-marketing<br />
arrangements contributed 30 per cent<br />
of revenues with a list of leading<br />
pharmaceutical clients.<br />
• 10 new products were introduced<br />
across four therapeutic segments<br />
including Dermatology,<br />
Cardiovascular, Pain Management and<br />
CNS segments.<br />
• New markets for API exports were<br />
identified and pursued with product<br />
registrations. A significant contribution<br />
to sales turnover came from the Asia<br />
Pacific, South America, Middle East<br />
and Canada.<br />
• Several patents were filed on<br />
non-infringing processes for APIs that<br />
have been developed by the research<br />
team.<br />
THE API ADVANTAGE<br />
As several products come to the end<br />
of their patent-protected lifecycle in<br />
the regulated markets, generic<br />
opportunities and API growth<br />
potential will increase manifold.<br />
The India advantage is three-fold: a<br />
significant wage cost advantage,<br />
high-quality certified manufacturing<br />
facilities and strong process<br />
chemistry skills.<br />
23
Strategically, a backward integration into the synthesis and<br />
manufacture of APIs helps manufacturers in three ways:<br />
• The potential to diversify revenues and profits by<br />
addressing the fast-growing API demand.<br />
• A significant cost and pricing advantage in the<br />
manufacture of formulations especially when captively<br />
manufactured APIs are used.<br />
• An ability to rapidly launch new formulations due to the<br />
captive availability of required APIs, resulting in a firstmover’s<br />
advantage and higher-than-normal initial revenues.<br />
DIVISIONAL GROWTH STRATEGY<br />
Even though <strong>Glenmark</strong> commenced the manufacture of<br />
APIs several years after it entered the production of<br />
formulations, it has built significant strengths over the last<br />
few years. These comprise:<br />
• Economies of scale.<br />
• An ability to manufacture complex multi-step molecules<br />
and build value-added therapies for higher and sustainable<br />
growth.<br />
• An ability to leverage non-infringing processes and<br />
manufacture quality generic products that are delivered on<br />
time and at competitive prices in target markets.<br />
• A comprehensive understanding of regulatory<br />
requirements across various geographies.<br />
Over the years, the company strengthened its API<br />
presence through a four-pronged strategy:<br />
The exploration of opportunities in the growing<br />
lifestyle segment: As lifestyles have become increasingly<br />
sedentary and food habits have trended towards the subnutritious,<br />
new ailments with long-standing relevance have<br />
emerged. As a future-directed organisation, <strong>Glenmark</strong><br />
focuses on the growing opportunities offered by this<br />
lifestyle segment (even in formulations and international<br />
operations).<br />
Rapid launch of new molecules: To counter the<br />
industry feature of shortening product life cycles and<br />
declining realisations, <strong>Glenmark</strong> introduces new<br />
molecules with an increasing frequency to climb the valuechain.<br />
Over the years, new product launches have<br />
contributed to an increasing proportion of revenue. The<br />
accelerated launch of new products across diverse<br />
therapeutic groups has also served to effectively de-risk<br />
the API business from price declines across its existing<br />
basket of products.<br />
Building strong process research capabilities: The<br />
company’s API division possesses strong, dedicated<br />
process research capabilities across its six laboratories.<br />
24
The accelerated launch of new<br />
products across diverse<br />
therapeutic groups has also<br />
served to effectively de-risk the<br />
API business from price<br />
declines across its existing<br />
basket of products.<br />
These laboratories are complete with state-of-the-art<br />
equipment and analytical tools at the company’s R&D<br />
centre at Mahape, New Mumbai. The laboratories are<br />
manned by a qualified 50 member team of scientists and<br />
process chemists. The research strength has also been<br />
reinforced by a sophisticated information centre that<br />
provides relevant scientific data that guides the company in<br />
the development of a pipeline of high opportunity drugs.<br />
The research facilities specialise in chiral chemistry, heterocyclic<br />
chemistry, resolution chemistry and carbohydrate<br />
chemistry. The research team is currently working on 18<br />
projects and has commercialised 35 products over the last<br />
three years. Its capabilities are reflected in the fact that<br />
product development time and cost are significantly lower<br />
compared to competition. The company has filed patents<br />
on non-infringing processes for ten products and has also<br />
filed three DMFs in 2003-04. A fourth DMF has been filed<br />
in April 2004.<br />
Low cost, high quality manufacturing: The division<br />
has three plants – one at Kurkumbh, a recently acquired<br />
plant at Solapur and another at Ankhleshwar. Ankhleshwar<br />
was acquired from GlaxoSmithKline and has been<br />
upgraded to comply with USFDA certification<br />
requirements. The plant’s 75 KL reaction volume capability<br />
is best suited for the launch of new products and also<br />
provide a good platform for versatile chemical reactions<br />
such as Friedel Craft, Grignard and Suzuki.<br />
OUTLOOK<br />
The API division expects to drive business revenue growth<br />
(non-captive) at about 100 per cent in 2004-05 through the<br />
following initiatives:<br />
• A tie-up with formulators in various countries including<br />
India. The division will also market value-added formulation<br />
products in the domestic market through co-marketing<br />
arrangements. The target number of products on offer are<br />
expected to grow from the current 35 to 53.<br />
• The development of 8-10 Drug Master Files (DMFs) for<br />
the US market to stimulate the API and generic formulation<br />
businesses.<br />
• A focus on the Asia-Pacific, South America, Eastern<br />
Europe and parts of Western Europe.<br />
• Additional product launches for the domestic market to<br />
serve the anti-hypertensive, anti-diabetes and internal<br />
medicine segments.<br />
• A capacity upgrade for the Ankleshwar facility.<br />
END NOTE<br />
<strong>Glenmark</strong> will continue to focus on strengthening its API<br />
operations and transplanting its experience into the<br />
advanced generic markets of USA and Europe.<br />
25
Group<br />
International<br />
operations<br />
26
The company’s US subsidiary<br />
concluded a landmark deal<br />
with KV Pharmaceuticals, a<br />
mid-cap US speciality and<br />
generics firm, to develop and<br />
market generics.<br />
THIS FASTEST GROWING SEGMENT<br />
IN GLENMARK CURRENTLY<br />
OPERATES IN THE REGULATED<br />
MARKETS ALONG WITH THE<br />
RAPIDLY GROWING MARKETS OF<br />
ASIA, AFRICA, CARIBBEAN<br />
ISLANDS, LATIN AMERICA, RUSSIA<br />
AND THE CIS STATES. THE<br />
POTENTIAL IS REFLECTED IN THE<br />
FACT THAT THE DIVISION’S<br />
TURNOVER OF RS. 469.3 MILLION<br />
IS GROWING AT MORE THAN 50<br />
PER CENT PER ANNUM.<br />
PERFORMANCE HIGHLIGHTS,<br />
2003-04<br />
• The company’s international<br />
operations recorded revenues of<br />
Rs. 469.3 million in 2003-04, a growth<br />
of 58 per cent over the previous year<br />
(Rs 296.9 million in 2002-03) at<br />
margins that were significantly higher<br />
than the rest of the company’s<br />
businesses.<br />
• Revenues from Africa more than<br />
doubled, Asian revenues were close<br />
to double and the Russian and CIS<br />
revenues grew 35 per cent.<br />
• The company expanded operations<br />
to cover 59 semi-regulated country<br />
markets by March 2004.<br />
• Four countries were added to the<br />
portfolio in 2004.<br />
• The marketing and sales network<br />
was expanded to several countries<br />
and the company appointed<br />
country managers across the semiregulated<br />
markets to directly market<br />
the <strong>Glenmark</strong> brand and its<br />
formulations.<br />
• The company’s US subsidiary<br />
concluded a landmark deal with KV<br />
Pharmaceuticals, a mid-cap US<br />
speciality and generics firm to<br />
develop and market generics.<br />
• A subsidiary was registered in UK to<br />
explore opportunities in the European<br />
Union.<br />
REGULATED MARKETS: THE US<br />
INITIATIVE<br />
During 2003-04, <strong>Glenmark</strong><br />
Pharmaceutical Inc., the company’s<br />
US subsidiary, strengthened its<br />
management team with the objective<br />
to accelerate the filing of ANDAs and<br />
market finished formulations in the<br />
US market.<br />
This initiative was as timely as it is<br />
critical: the generic opportunity<br />
represents a significant revenue<br />
potential for <strong>Glenmark</strong>.<br />
Apart from various other initiatives,<br />
the US subsidiary signed a landmark<br />
agreement with KV Pharmaceuticals<br />
Inc, a US $ 1.4 billion marketcapitalisation<br />
pharmaceutical<br />
company. Under the agreement, the<br />
two companies share responsibilities<br />
for the development, registration,<br />
supply and marketing of eight<br />
identified drugs that are expected to<br />
go off-patent in the near future.<br />
27
The company is also<br />
building its own sales and<br />
marketing team to offer<br />
products to distributors and<br />
stockists in the US under<br />
the <strong>Glenmark</strong> label.<br />
<strong>Glenmark</strong> will be entitled to significant upfront and<br />
milestone payments in the development / registration<br />
stages and royalties from sales within US. The agreement<br />
has the option to cover an additional ten generic products<br />
and three branded products, leveraging the patented<br />
platform technologies developed by <strong>Glenmark</strong>. The<br />
additional products would be jointly identified by <strong>Glenmark</strong><br />
and KV Pharmaceuticals at a later date.<br />
The products to feature in the KV Pharmaceuticals Inc.<br />
agreement will span five therapeutic segments including<br />
Cardiovascular, Central Nervous System, Dermatology,<br />
Immunology and Diabetology with their launch dates<br />
spanning the next three years.<br />
The company is also building its own sales and marketing<br />
team to offer products to distributors and stockists in the<br />
US under the <strong>Glenmark</strong> label. In the long run, the<br />
company’s wider product basket will be leveraged to build<br />
a robust product pipeline, helping build the company’s own<br />
franchise in the US generic drugs business. The company<br />
has already identified 15 products for which ANDAs will be<br />
filed over the next three years; of these the company is in<br />
an advanced stage of development for eight products.<br />
Some of these products will also leverage patentprotected<br />
New Drug Delivery Systems that have been<br />
developed through in-house research.<br />
<strong>Glenmark</strong> enjoys a distinct competitive edge that will drive<br />
its success in the US markets:<br />
• Its status as a speciality company with deep skills in select<br />
therapeutic segments will help it leverage opportunities in<br />
those areas (dermatology is an example) better.<br />
• The strong local management team enjoys a prior<br />
experience in establishing the US operations for other<br />
Indian pharmaceutical companies; this will ensure speedy<br />
execution.<br />
• <strong>Glenmark</strong>’s vertical integration will enable it to serve the<br />
US markets with APIs and formulations at competitive<br />
costs.<br />
• The company’s strong generic and formulation<br />
development competencies will translate into the<br />
rapid deployment of target products and a stronger<br />
revenue growth.<br />
28
<strong>Glenmark</strong> Pharmaceuticals’ wholly owned Brazilian<br />
subsidiary <strong>Glenmark</strong> Farmaceutica Ltda. acquired a<br />
private pharmaceutical Brazilian company called<br />
‘Laboratorios Klinger’ in the last quarter of 2003-04.<br />
Klinger has 21 products under registration in Brazil<br />
spanning several therapeutic segments including some<br />
OTC products. The company has a workforce of 176<br />
employees including 91 market representatives. Apart<br />
from offering an entry route into the competitive<br />
Brazilian market, the acquisition will also allow<br />
<strong>Glenmark</strong> to address other Latin American markets.<br />
Apart from continuing the lucrative products in<br />
Klinger’s portfolio, <strong>Glenmark</strong> will also consider<br />
transferring several products from its global portfolio to<br />
Brazil. Significant synergies with <strong>Glenmark</strong> in product<br />
development, bulk sourcing and local formulation<br />
manufacture will drive value extraction in the Latin<br />
American market.<br />
<strong>Glenmark</strong>’s revenues from the US market will commence<br />
in 2005 as a part of the KV deal and additional revenues<br />
from marketing products under the <strong>Glenmark</strong> label are<br />
expected to commence by 2006.<br />
SEMI-REGULATED/DEVELOPING MARKETS<br />
<strong>Glenmark</strong> has focused on select markets for four years,<br />
building a significant presence and brand image in them.<br />
These markets, at an early stage of evolution, are<br />
served by an earlier generation of drugs though this is<br />
undergoing rapid change in recent years. Since a number<br />
of these markets are price-sensitive, they offer<br />
significant generic opportunities for low-cost<br />
players like <strong>Glenmark</strong> to fill therapy gaps and capture<br />
market share.<br />
<strong>Glenmark</strong>’s international expansion has benefited from an<br />
ongoing knowledge management: it has leveraged the<br />
experience gained in one geography to enter another,<br />
thereby shrinking the time-to-market and rapidly<br />
establishing a strong market presence. <strong>Glenmark</strong>’s edge in<br />
the semi-regulated markets has been derived from its<br />
reputation as a responsible manufacturer with a visible<br />
long-term commitment: it has not relied on revenue<br />
building from a one-time opportunistic perspective but<br />
invested in a sustainable brand and marketing presence in<br />
its focus markets.<br />
The company’s international presence has been<br />
reinforced through a judicious mix of strategies, including<br />
the engagement of local sales teams to promote its<br />
products in these markets. The company promotes its<br />
brands directly to the healthcare fraternity, building a<br />
strong franchise and generating a consequent<br />
demand-pull. Its strong and deep product pipeline<br />
also fills gaps in existing therapies in the markets of its<br />
presence. It is the company’s ability to adapt to<br />
varying international conditions that has<br />
enhanced its image as a customer-centric pharmaceutical<br />
player.<br />
The share of revenue from these markets as a proportion<br />
of <strong>Glenmark</strong>’s revenue is expected to increase significantly<br />
over the near future. The company expects to achieve over<br />
100 per cent growth (including Latin America) in revenues<br />
from semi-regulated markets in 2004-05.<br />
29
30<br />
Research and<br />
development
HISTORICALLY THE INDIAN<br />
PHARMACEUTICAL INDUSTRY<br />
POSSESSED DEEP SYNTHETIC AND<br />
PROCESS CHEMISTRY<br />
CAPABILITIES.<br />
The origin of this limited focus was<br />
derived from India’s longstanding<br />
process patent regime, which allowed<br />
companies to reverse-engineer and<br />
sell in India patent-protected drugs of<br />
the regulated markets. In view of the<br />
assured protection and easy access to<br />
cutting-edge research, Indian<br />
companies did not make investments<br />
in costly original research with any<br />
seriousness.<br />
A decision to respect product patents<br />
from 2005 will however change the<br />
scenario. As reverse-engineering will<br />
be statutorily banned, the initial<br />
hesitation in research investments is<br />
leading to a mild optimism: there is a<br />
growing realisation that India presents<br />
significant cost and skill advantages<br />
for the original research required to<br />
build IPR assets. A combination of the<br />
need to differentiate products in<br />
target markets, respect product<br />
patents and leverage India’s research<br />
strengths have translated into a<br />
growing investment in IPR<br />
development.<br />
In view of this, a number of Indian<br />
companies, including <strong>Glenmark</strong>, are<br />
building capabilities in original<br />
research, targeting new chemical<br />
entities (patentable drug options) and<br />
novel drug delivery systems.<br />
NEW CHEMICAL ENTITIES<br />
(NCEs)<br />
New chemical entities represent the<br />
highest end of the research value<br />
chain; typically, they also offer the<br />
highest reward. A successful NCE,<br />
following the relevant approvals, can<br />
be marketed with a 12-14 year<br />
product exclusivity period during<br />
which it can recover its investment a<br />
number of times over. Their collective<br />
revenue potential is reflected in the<br />
fact that blockbuster drugs such as<br />
Viagra and Statins, among others,<br />
enjoy annual revenues in excess of<br />
US$ 5-6 billion, a little under the size<br />
of the entire Indian pharmaceutical<br />
market.<br />
While the opportunity is significant,<br />
the development of NCEs involves a<br />
multitude of skill sets, a development<br />
time of 6-8 years and considerable<br />
risk. NCE research focuses on<br />
developing chemicals / molecules that<br />
can selectively address molecular<br />
targets of certain specific diseases<br />
and provide a cure with minimal side<br />
effects. As such, successful NCE<br />
research has almost become the<br />
prerogative of large and well-funded<br />
MNCs and institutions, supported by<br />
heavy research budgets. Indian firms<br />
do not invest in the target<br />
identification exercise; instead, they<br />
adopt an approach called ‘analogue<br />
research’, which entails working on<br />
certain pre-identified targets for<br />
specific diseases to develop<br />
molecules that alter the target’s<br />
mechanism in the diseased person.<br />
Analogue NCE research is timeconsuming,<br />
risky and expensive. The<br />
entire process can span 6-8 years<br />
before a drug can be taken to the<br />
market and it runs the risk of the<br />
molecule needing to be dropped at<br />
any intervening stage for its inability<br />
to clear trials.<br />
THE LICENSING OPPORTUNITY<br />
The Indian advantage is not limited to<br />
manufacturing; the country offers an<br />
arbitrage opportunity even when it<br />
comes to the cost of intellectual<br />
capital and facility overheads. The<br />
nation also possesses a deep<br />
inventory of requisite competencies<br />
required to engage in original<br />
research. In the area of drug<br />
discovery, for instance, India enjoys a<br />
staggering 80-90 per cent cost<br />
advantage compared to developed<br />
countries like the US. Additionally, as<br />
global majors intensify their research<br />
activities to fill-in a fast depleting<br />
product pipeline, their failure rates are<br />
expected to increase, widening this<br />
differential further.<br />
However, Indian companies face a<br />
resource crunch to fund even a tenth<br />
of the absolute original research cost<br />
of USD 800-1000 million. Besides,<br />
regulators in developed countries are<br />
reluctant to accept Indian clinical trial<br />
data. As a result, the Indian research<br />
industry is inadequately equipped to<br />
sustain the time and cost implications<br />
of a start-to-finish research model.<br />
In the light of the high research costs,<br />
an alternative collaborative route in<br />
NCE research is being increasingly<br />
favoured. In this arrangement, the<br />
lead compounds are licensed to global<br />
majors. A successful drug candidate<br />
for such a licensing deal falls within<br />
certain categories:<br />
• Sunrise therapies: Sunrise sectors<br />
are those therapy areas that are new<br />
and retain the promise of generating<br />
high revenues.<br />
31
• Niche therapies: Niche areas are<br />
more valuable for Indian companies<br />
who aim to build partnerships and<br />
enter into co-marketing arrangements<br />
with mid-cap and smaller overseas<br />
firms.<br />
• Chronic therapies: Certain ailments<br />
like diabetes and cardio-vascular<br />
ailments are chronic diseases.<br />
Patients are prescribed the lifelong<br />
use of drugs that, in turn, present<br />
higher value to potential license<br />
partners due to a higher sustainability<br />
of sales.<br />
GLENMARK’S VISION<br />
Along with process chemistry and<br />
reverse-engineering generics, original<br />
research represents a key pillar in<br />
<strong>Glenmark</strong>’s business vision. <strong>Glenmark</strong><br />
judiciously invests cash flows from<br />
the generic business into generating<br />
long-term intellectual property assets<br />
through its research initiatives.<br />
<strong>Glenmark</strong> recognises the value of<br />
investing in original research,<br />
generating valuable IPR assets that<br />
will sustain its revenues and earnings<br />
in a post-GATT, product patent<br />
regime. These IPR assets will also<br />
allow the company to establish its<br />
brand name in regulated overseas<br />
markets and thereby become a truly<br />
global company.<br />
GLENMARK NCE R&D<br />
HIGHLIGHTS<br />
In view of the growing importance of<br />
captive access to cutting-edge<br />
research, the company embarked on<br />
investing in a state-of-the-art R&D<br />
infrastructure and competencies<br />
about four years ago. The research<br />
division has yielded dramatic results<br />
in a short period, justifying the<br />
investments.<br />
Considerable progress has been<br />
made in developing a drug targeting<br />
the asthma/COPD segment; the<br />
company is now tying up with<br />
international contract research labs to<br />
commence early clinical trials in July<br />
2004. The company has also<br />
succeeded in developing a set of lead<br />
compounds that are in the late<br />
pre-clinical stage of the diabetes<br />
therapy areas.<br />
The selection of the therapeutic areas<br />
for research was based on a distinct<br />
ground reality:<br />
• Global growth potential: The<br />
incidence of asthma/COPD, diabetes<br />
and obesity are growing rapidly;<br />
collectively their incidence is<br />
expected to be more than USD 44<br />
billion by 2007.<br />
• Potential for pioneering research:<br />
Since the existing therapies for these<br />
ailments have undesirable sideeffects,<br />
they offer attractive<br />
replacement opportunities. For<br />
example, steroids used currently in<br />
treating asthma are non-selective and<br />
report significant side-effects on the<br />
human body.<br />
• Licensing opportunity: Since<br />
<strong>Glenmark</strong>’s research areas are of<br />
interest to several international majors<br />
because of their high revenue potential,<br />
they present significant partnership<br />
opportunities in licensing at the late<br />
pre-clinical or early clinical stages.<br />
• Research skill-sets: <strong>Glenmark</strong>’s<br />
senior scientists, responsible for<br />
directing its research programme,<br />
enjoy significant exposure, interest<br />
and experience in the selected areas<br />
for NCE focus. To reinforce their<br />
effort, the company has appointed an<br />
advisory board of eminent scientists,<br />
who possess experience in the<br />
molecular targets selected for<br />
research.<br />
PROGRESS IN NCE R&D<br />
Asthma/COPD<br />
Asthma is a chronic debilitating<br />
disease characterised by ‘airway<br />
32
hyperactivity’ and inflammation. The<br />
incidence of asthma is growing rapidly<br />
worldwide and the available<br />
medications are unable to meet the<br />
growing needs and incidence. As it is<br />
a chronic ailment, majority of asthma<br />
patients require therapy over a long<br />
period, sometimes spanning a lifetime<br />
and the global market for asthma<br />
medication is estimated at US $ 8-12<br />
billion. Current medications include<br />
inhaled ß- agonists, bronchial steroids<br />
and antileukotriene antagonists.<br />
These current therapies aim to treat<br />
asthma by producing antiinflammatory<br />
and/or bronchodialatory<br />
effect. However, none of the existing<br />
therapies are able to effectively<br />
control the disease and some of the<br />
treatments such as steroids have<br />
potential side-effects.<br />
PDE-4 inhibitors are a recent focus<br />
area for scientists; research has<br />
commenced over the last ten years.<br />
The research aims to develop<br />
compounds that selectively inhibit<br />
Phospodiesterase Enzyme (PDE-4), an<br />
enzyme that catalyses metabolism of<br />
cyclic-AMP, a secondary messenger in<br />
cellular functions. Elevation of<br />
intracellular cAMP levels is known to<br />
have both bronchodialatory and antiinflammatory<br />
activities. Hence,<br />
selective PDE-4 inhibitors present an<br />
attractive alternative for asthma<br />
therapy. However, some of the PDE-4<br />
inhibitors entered into clinical<br />
development namely Rolipram<br />
(Schering Plough), Ariflo (Glaxo) etc.<br />
During the last decade have shown<br />
side-effects that include nausea and<br />
vomiting and hence, limit the potential<br />
dosage that can be delivered and also<br />
the efficacy. The most encouraging<br />
results have been demonstrated<br />
recently by Roflumilast (Altana), which<br />
has completed Phase-III development<br />
in Europe and has been shown to<br />
increase lung function in asthmatic<br />
patients. However, Roflumilast has<br />
also shown emetic side-effects at<br />
elevated dosages. Hence, though the<br />
drug candidate appears promising, a<br />
need for safer medication continues<br />
to exist.<br />
<strong>Glenmark</strong> has been researching PDE-<br />
4 inhibitors for more than three years.<br />
The research has shown promising<br />
results and the latest lead molecule<br />
for <strong>Glenmark</strong> is GRC-3886, a highly<br />
selective PDE-4 inhibitor with no<br />
emetic side-effects demonstrated in<br />
animal models. It has excellent in-vitro<br />
and in-vivo pharmacological activity<br />
against the target. As per the<br />
preclinical studies conducted by<br />
<strong>Glenmark</strong>, GRC-3886 has also shown<br />
equivalent activity with that of<br />
Roflumilast. Based on its high<br />
selectivity, low side-effect profile and<br />
favourable pharmacokinetic profile,<br />
the molecule has been selected as a<br />
lead candidate for clinical<br />
development.<br />
Currently GRC-3886 is in late phase<br />
toxicity studies in animal models at a<br />
leading European contract research<br />
organisation (CRO) and is expected to<br />
enter into human Phase – I clinical<br />
trials by July-2004. <strong>Glenmark</strong> has<br />
entered into a tie-up with Quintiles, a<br />
leading global CRO, for the clinical<br />
testing of GRC-3886 in UK.<br />
The preclinical profile of GRC-3886 is<br />
being communicated at the following<br />
world scientific forums:<br />
• Experimental Biology Meet<br />
involving American Association of<br />
Immunologists in April 2004 at<br />
Washington USA.<br />
• Inflammatory diseases, clinical<br />
updates, new targets and novel<br />
theory methods for anti-inflammatory<br />
therapeutics in June 2004, Hilton<br />
Logon, Boston.<br />
• 14th Annual Congress of European<br />
Respiratory Society in September<br />
2004 at Glasgow, U.K.<br />
• 11th International Congress of<br />
Immunology, July 18-23 2004 in<br />
Montreal, Canada.<br />
Diabetes/Obesity<br />
Globally, Type II Diabetes is one of<br />
the most common chronic diseases.<br />
The ‘Metabolic Syndrome’ of the<br />
disease includes obesity,<br />
hypertension, hyperlipidemia and<br />
cardiovascular diseases. Presently<br />
diabetes causes significant morbidity<br />
and mortality due to long-term micro<br />
and macro vascular complications.<br />
The current incidence of Type II<br />
Diabetes in US is estimated to be 7<br />
per cent of the population. Spend on<br />
related treatment accounts for as<br />
much as 10 per cent of all healthcare<br />
dollars in the US. Furthermore, the<br />
incidence of Type II Diabetes is<br />
increasing globally at a rapid rate in<br />
Africa, South America and Asia<br />
leading to the disease now being<br />
considered a worldwide ‘epidemic’. In<br />
India, environmental and genetic<br />
factors, influenced by changing socioeconomic<br />
scenario, have made an<br />
increasing number of Indians<br />
susceptible to this deadly epidemic.<br />
Based on recent epidemiological data,<br />
the growth rate of Type II diabetes is<br />
estimated to be 12-16 per cent in India,<br />
much higher than the global rate.<br />
Type II Diabetes is associated with<br />
insulin resistance in peripheral tissues<br />
such as muscles and fat, impaired<br />
glucose stimulated insulin secretion<br />
from pancreatic beta-cells and<br />
elevated hepatic gluconeogenesis.<br />
Though certain drugs are available to<br />
treat the pathological conditions<br />
associated with Type II Diabetes,<br />
33
current pharmacotherapy does not<br />
adequately address the metabolic<br />
defects underlying this disease.<br />
<strong>Glenmark</strong> Research Centre was<br />
developing new drug candidates in<br />
the Beta-3 Agonist area. In parallel,<br />
the <strong>Glenmark</strong> Research Centre is also<br />
focusing on two other novel targets,<br />
hitherto undisclosed, associated with<br />
glucose dependent insulin secretion<br />
and peripheral insulin resistance.<br />
These additional targets were<br />
selected because they contribute<br />
alternative pharmacological<br />
approaches to treat the distinct<br />
deficits existing in the therapy of Type<br />
II Diabetes. The targets have<br />
established proof of concept and are<br />
currently being pursued by various<br />
multinational pharmaceutical majors.<br />
Several of the drug candidates in<br />
these novel target areas are being<br />
developed by global competitors<br />
and are at various stages of<br />
development from preclinical to<br />
clinical testing.<br />
Due to exciting leads being developed<br />
for the undisclosed targets, the Beta-<br />
3 project stream has currently been<br />
put on hold. <strong>Glenmark</strong> Research<br />
Centre has identified preclinical leads<br />
in the area of diabetes and expects to<br />
conduct human phase I clinical trials<br />
by October 2004. The research centre<br />
plans to communicate the preclinical<br />
profiles of these lead molecules at<br />
the following world scientific<br />
forums:<br />
• 64th Scientific Session of American<br />
Diabetes Association to be held in<br />
June 2004 at Florida, USA.<br />
• 40th Annual Meeting of EASD<br />
(European Association for study of<br />
Diabetes) to be held in September<br />
2004 at Munich, Germany.<br />
Risk management in NCE<br />
research<br />
While <strong>Glenmark</strong> has very promising<br />
lead compounds in both areas, a<br />
parallel research initiative continues to<br />
identify back-up molecules. This acts<br />
as a risk-mitigating strategy and is<br />
important till such time the drug<br />
candidate advances to late-clinical<br />
trials. Thus, apart from the lead<br />
compound in both therapy areas,<br />
<strong>Glenmark</strong> has developed a back-up<br />
pre-clinical pipeline of several drug<br />
candidate molecules for<br />
asthma/COPD and diabetes/obesity<br />
respectively.<br />
In addition to these therapy areas, the<br />
company is also working with leading<br />
government laboratories like the<br />
Institute of Chemical Biology, CDRI<br />
(Lucknow) and National Chemical<br />
Laboratories (Pune) in the areas of<br />
obesity, anti-inflammation and pain<br />
management.<br />
NOVEL DRUG DELIVERY<br />
SYSTEMS (NDDS)<br />
NDDS products are those that offer a<br />
technological advantage over simple<br />
therapeutic solutions. These products<br />
achieve their desired effects by<br />
altering the release profile, availability<br />
and safety profile of the drug. Even<br />
though the risk profile and the cost of<br />
development are less than that of<br />
new chemical entities, these products<br />
require a new drug application<br />
process and are protected through<br />
longer patent exclusivity periods.<br />
These products provide<br />
pharmaceutical companies<br />
with a pipeline of products<br />
that can be marketed at a premium<br />
over simple therapies or generic<br />
products.<br />
<strong>Glenmark</strong>’s NDDS<br />
R&D highlights<br />
Over the years, <strong>Glenmark</strong> has<br />
emphasised the development of a<br />
host of technologies that possess IPR<br />
value. These technologies are related<br />
to drug delivery in the human body<br />
and are adaptable to several drugs<br />
especially in the oral and dermal<br />
range. Even if the underlying drug is a<br />
generic, packaging it with a patentprotected<br />
delivery system offers an<br />
attractive opportunity to counter<br />
declines in margins and realisations<br />
that generics usually face. These<br />
delivery technologies also help in<br />
branding the company as a speciality<br />
organisation in domestic and<br />
international markets.<br />
<strong>Glenmark</strong> is presently working on a<br />
host of technologies with significant<br />
IPR value. The company possesses<br />
several patents spanning areas such<br />
as controlled release and matrix<br />
technologies; it plans to develop six<br />
sustained release products over the<br />
next 18-24 months. This NDDS effort<br />
is a key element in the company’s<br />
strategy to enter the US market<br />
through ANDA filings.<br />
INFRASTRUCTURE AND SKILL<br />
ASSETS<br />
<strong>Glenmark</strong> Research Centre presently<br />
employs 250 scientific staff engaged<br />
in drug discovery, drug delivery<br />
systems, process development and<br />
analytical research. This talented pool<br />
of dedicated scientists includes 35<br />
PhDs, several of them with postdoctoral<br />
experience from universities<br />
in US/ Europe. The individual research<br />
divisions are headed by people with<br />
rich experience in the respective<br />
research areas. <strong>Glenmark</strong> has also<br />
made significant investments in<br />
equipping the research team with<br />
requisite modern scientific<br />
34
equipments to carry out drug<br />
discovery and development as per<br />
national/ international standards and<br />
regulations, which include:<br />
1. LC/MS/MS (Liquid<br />
Chromatography/ Mass<br />
Spectrometer).<br />
2. Nuclear Magnetic Resonance<br />
Spectrometer-NMR.<br />
3. 35 HPLCs.<br />
4. Fluid Bed Dryer-GLATT.<br />
5. Formulation equipment for<br />
lab-scale development.<br />
6. Gas Chromatographs and other<br />
analytical equipment.<br />
Apart from being engaged in<br />
research, the scientists represent<br />
<strong>Glenmark</strong> Research Centre in various<br />
national / international seminars and<br />
meetings to present the results of<br />
in-house research efforts. Some of the<br />
representations related to asthma and<br />
diabetes research have already been<br />
highlighted in the concerned sections.<br />
Publication of the results of in-house<br />
research in peer-reviewed international<br />
journals is a continuous activity at the<br />
<strong>Glenmark</strong> Research Centre.<br />
NCE: EXCITING PRODUCT PIPELINE, ENTERING CLINICAL STAGE<br />
Stages of Development<br />
Phase I Phase II A Estimated<br />
Therapeutic Target Compound (6-8 (3-6 Phase I<br />
Pre Clinical mths) mths.) start date<br />
Other In<br />
Synthesis In Vitro Pharmaco- Taxicity Vivo IND<br />
Screening kinetics animal Filing<br />
models<br />
Asthma/<br />
COPD<br />
Asthma/<br />
COPD<br />
PDE4 GRC 3566<br />
PDE4 GRC 3886<br />
GRC 3886 will<br />
enter Phase I<br />
with a leading<br />
CRO in UK by<br />
July-August<br />
2004<br />
Obesity/ Beta 3<br />
diabetes agonist<br />
Diabetes Undisclosed<br />
GRC 1087<br />
GRC 8087<br />
One of the<br />
compounds<br />
expected<br />
to enter<br />
Phase I by<br />
Oct 2004<br />
35
Global Management Team<br />
Name<br />
Responsibility<br />
Glenn Saldanha<br />
CEO<br />
A. S. Mohanty Dir (Domestic formulations)<br />
Rajesh Desai<br />
Dir (Fin/IT/Legal)<br />
Jeff Weiss<br />
CEO (US Subsidiary)<br />
Ailton Wiliczinski<br />
CEO (Brazil Subsidiary)<br />
Dr. Shekar Bhirud<br />
Sr. VP (API)<br />
Avadhut Sukhtankar<br />
Sr. VP (Operations)<br />
K. Anand VP (Regulatory Affairs and QA)<br />
V. S. Mani VP (Finance)<br />
Jeff Glazer<br />
Exec. VP (IP in US)<br />
Alind Sharma<br />
VP (HR)<br />
Doug Sunday<br />
VP (API in US)<br />
Vithal Dhamankar<br />
VP (Semi Regulated Markets - Formulations)<br />
36
Management’s<br />
discussion and analysis<br />
INDUSTRY STRUCTURE<br />
The Indian pharmaceutical market of USD 4.8 bn accounts<br />
for only 1.6 per cent of the world market. In spite of its<br />
small size, the sector represents an attractive opportunity<br />
for growth as it moves from its prevalent respect for<br />
process patents to a regime that respects product patents<br />
following 2005.<br />
While the new regime is still some months away from<br />
becoming a legal reality, certain changes have already<br />
transpired:<br />
• Competition has intensified; formulation companies are<br />
accelerating new product launch.<br />
• The launch of each new product category is followed by<br />
the rapid introduction of a number of me-too brands,<br />
leading to sharp price declines.<br />
• The combination of existing price controls and price<br />
declines have eroded corporate profitability.<br />
• As brands have resisted the price declines better, brand<br />
buy-outs have been initiated by companies striving to<br />
consolidate their position in select therapeutic segments,<br />
and<br />
• A slow industry growth within India has prompted<br />
pharmaceutical companies to extend their reach to<br />
international geographies.<br />
Over the last few years, India’s pharmaceutical industry has<br />
also seen an increase in the incidence of mergers and<br />
acquisitions, driven by a growing need among<br />
manufacturers to strengthen their product portfolio,<br />
minimise investment duplication and widen their marketing<br />
coverage.<br />
OPERATIONAL OVERVIEW<br />
<strong>Glenmark</strong> constantly reviews its product-market portfolio<br />
with a view to strengthen sustainable growth. Internally,<br />
the three areas that the company focused on to drive<br />
financial improvement were:<br />
• An investment in revenue generating assets,<br />
• A stronger manufacturing and supply chain management,<br />
• A superior system to improve the information quality<br />
available to the management.<br />
Over the last five years since the IPO, the company has<br />
progressively invested in long-term value assets that would<br />
strengthen its competitive position. Investments made in<br />
acquiring the API plant at Ankleshwar from GSK have since<br />
paid off. The company has also commissioned a plant at<br />
Goa that has been built to meet the stringent USFDA<br />
certification standards. The plant will allow <strong>Glenmark</strong> to<br />
serve the regulated markets overseas in the coming years.<br />
The company also continues to support a sizeable revenue<br />
expenditure and capital investments in the R&D facility at<br />
New Bombay.<br />
The company is also spending on upgrading and expanding<br />
the manufacturing operations across all locations. These<br />
investments will enhance manufacturing efficiencies<br />
through increased throughput and lower costs in the near<br />
to medium term. The company is actively working on<br />
superior vendor management practices that are likely to<br />
improve quality and timeliness of supplies and lower costs<br />
over the longer-term. Along with an ongoing drop in prices<br />
of bulk and packing material these initiatives are expected<br />
to improve the company’s bottom line in the future.<br />
37
To ensure superior control of operations, the company has<br />
instituted a superior managerial and executive information<br />
system. These systems are beginning to deliver the<br />
desired results and as a result, the company is able to<br />
monitor its operations and costs much better.<br />
As a result of all the above initiatives, <strong>Glenmark</strong>’s operating<br />
margins in 2003-04 was 18.40 per cent compared to 18.06<br />
per cent in 2002-03.<br />
REVENUES<br />
The company generated a turnover of Rs. 3806.61 million<br />
in 2003-04 compared to Rs. 3336.37 million in 2002-03, an<br />
increase of 14.09 %, generated on the strength of an<br />
improved performance across all its geographic markets.<br />
DOMESTIC SALES<br />
The company’s domestic sales in 2003-04 amounted to<br />
Rs. 2892 million compared to Rs. 2743.97 million in 2002-<br />
03, an increase of 5.39%. The higher income was achieved<br />
in spite of realisation declines across a number of products.<br />
A strong performance by flagship brands, new product<br />
introduction and the consolidation of franchises in<br />
segments of strength accounted for the revenue growth.<br />
EXPORT FORMULATIONS<br />
Exports, derived from the company’s presence in semiregulated<br />
markets, amounted to Rs. 469.28 million in 2003-<br />
04 compared to Rs. 296.92 million in 2002-03.<br />
Figures in Rs. mn<br />
Geography Mar 2004 Mar 2003 % growth<br />
Asia 139.93 80.80 73.18<br />
Africa 162.73 76.80 111.89<br />
Russia and others 166.62 139.32 21.14<br />
To drive growth sustainably over the coming years, the<br />
company made investments in the Latin American market<br />
and concluded a deal with KV Pharma, a mid-cap (USD 1.4<br />
bn) US speciality and generics player. The latter<br />
arrangement involves the development and marketing of<br />
eight products that are nearing patent expiry in the US<br />
market. Revenues from these initiatives are expected to<br />
commence in the next two years.<br />
API AND CO-MARKETING<br />
The API and co-marketing division recorded revenues of<br />
Rs. 445.32 million in 2003-04 compared to Rs. 286.10<br />
million in 2002-03 an increase of 55.65 per cent. Besides,<br />
the division also addressed a part of the captive<br />
requirement for bulk actives within the formulation division.<br />
COST OF SALES<br />
The cost of sales declined from 49.28 per cent in 2002-03<br />
to 47.11 per cent in 2003-04 mainly on account of a<br />
changed product mix, various cost reduction measures and<br />
a general decline in the realisations of bulk actives.<br />
SELLING AND OPERATING EXPENSES<br />
Selling and operating expenses were Rs 1065.03 million in<br />
2003-04, an increase of 13.01 per cent against Rs.942.42<br />
million in 2002-03. Selling and operating expenses as a<br />
percentage of sales in 2003-04 was 27.98 per cent<br />
compared with 28.25 per cent in 2002-03, a decline of 0.27<br />
per cent.<br />
Promotional costs, including incentives and commission,<br />
increased by 2.63 per cent compared to the previous year<br />
mainly on account of an investment in <strong>Glenmark</strong>’s brand in<br />
the export markets. The increase in salaries, travelling and<br />
other expenses occurred mainly due to recruitments, salary<br />
38
evisions and other expenses that were made to achieve<br />
the targeted revenue growth. Freight and distribution costs<br />
decreased by 40.37 per cent.<br />
As a prudent accounting practice, the Company made a<br />
provision for doubtful debts of Rs. 11.80 million. The other<br />
expenses including administrative costs like telephone,<br />
rent, repairs and insurance have shown a marginal increase<br />
due to a rise in the overall activities.<br />
DEPRECIATION<br />
The provision for depreciation was Rs 108.89 million in<br />
2003-04 compared with Rs.103.33 million in 2002-03. This<br />
rise is mainly due to the increase in fixed assets of<br />
company.<br />
INTEREST<br />
The expenditure on account of interest was Rs 100.58<br />
million in 2003-04 compared with Rs.106.88 million in<br />
2003-04. The company was able to reduce its cost of debt<br />
by replacing high cost debt with low cost debt. Moreover,<br />
the conversion of debentures and repayment of other loans<br />
lowered the debt burden and consequently the interest.<br />
Figures in Rs. mn<br />
Head 2003-04 2002-03 2001-02 2000-01<br />
Interest 100.58 106.88 129.13 83.23<br />
EBITDA 734.89 645.43 502.13 320.33<br />
Interest cover (times) 7.31 6.04 3.89 3.85<br />
In the future, stronger cash flows and superior working<br />
capital management is likely to further lower interest costs<br />
and improve interest cover.<br />
RESEARCH AND DEVELOPMENT EXPENSES<br />
The Research and Development expenditure was<br />
Rs 248.07 million in 2003-04 compared with Rs.147.26<br />
million in 2002-03, a rise of 68.45 per cent. The R&D<br />
revenue expenditure was 6.52 per cent of the turnover in<br />
2003-04 compared with 4.41 per cent in 2002-03. The<br />
increase was in line with the growth of the R&D staff<br />
strength and increase in research activity. The detailed<br />
break up of the R&D expenditure is as below:<br />
Figures in Rs. mn<br />
Head Mar 2004 Mar 2003<br />
Salaries and other benefits 78.48 45.30<br />
Chemicals and consumables 80.56 52.97<br />
Other expenses 89.03 48.99<br />
PROVISION FOR TAXATION<br />
The taxation charge for the year was Rs 90.30 million<br />
compared with Rs.35.30 million in 2002-03.<br />
PROVISION FOR DEFERRED TAXATION<br />
A provision for deferred tax of Rs. 15.08 million was made<br />
as per Accounting Standard 22 ‘Accounting for Taxes on<br />
Income’ issued by the Institute of Chartered Accountants<br />
of India. The deferred tax provision for the future tax<br />
consequences was attributable to timing differences<br />
between the financial statement, determination of income,<br />
and its recognition for tax purposes.<br />
DIVIDEND<br />
The company paid an interim dividend of 65 per cent on<br />
the equity capital for 2003-04 and a dividend of 10.50 per<br />
cent on preference shares.<br />
EQUITY CAPITAL AND SHAREHOLDER FUNDS<br />
Equity capital and share premium is increased to<br />
39
Rs.1110.45 million from Rs.602.23 million due to the ESOP<br />
and conversion of debenture issued to CDC and SARF. The<br />
General Reserve and profit and loss increased to<br />
Rs. 1107.71 million from Rs.786.65 million due to the<br />
retained earnings.<br />
SECURED LOANS<br />
Secured loans declined to Rs 1029.43 million in 2003-04<br />
compared with Rs.1193.57 million in 2002-03, a fall of 13.75<br />
per cent. This was largely on account of the conversion of<br />
debentures into equity (mentioned above). The other major<br />
component of the secured loan was the working capital loan<br />
from Bank of India that was secured by hypothecation of<br />
stocks and receivables and mortgage of land, building,<br />
equipment, machinery at Nashik and Sinnar. Increased<br />
turnover resulted in higher working capital utilisation from<br />
banks.<br />
UNSECURED LOANS<br />
Unsecured loans increased to 119.31 million in 2003-04<br />
compared with Rs. 53.86 million in 2002-03.<br />
FIXED ASSETS<br />
The company’s gross asset block increased from<br />
Rs.1246.52 million as at 31st March, 2003 to Rs 1723.54<br />
million as at 31st March 2004 on account of modernisation<br />
of the Nashik and Ankleshwar plants, a part capitalisation<br />
of the Goa plant and additions made in the R&D division.<br />
Capital work-in-progress and capital advances worth Rs.<br />
244.97 million were made towards the Ankleshwar and<br />
Goa plant.<br />
RECEIVABLES<br />
Receivables declined from Rs.1236.69 million in 2002-03 to<br />
Rs 1304.81 million in 2003-04, a drop in number of days<br />
from 135 to 125.<br />
NET WORKING CAPITAL<br />
Net working capital levels remained almost unchanged, a<br />
feature that is likely to continue in 2004-05 as the<br />
Company’s export business grows.<br />
LOANS AND ADVANCES<br />
Loans and advances declined from Rs 502.70 million in<br />
2002-03 to Rs 392.26 million in 2003-04 due to receipts<br />
from <strong>Glenmark</strong> Laboratories on account of the sale of the<br />
Verna plant.<br />
CURRENT LIABILITIES<br />
Current liabilities and provisions increased from<br />
Rs. 617.48 million in 2002-03 to Rs 745.12 million in<br />
2003-04 comprising of an increase in creditors.<br />
This was mainly due to the increased working capital<br />
requirements to support revenue growth. During the year,<br />
the company paid an interim dividend of 65 per cent,<br />
which resulted in an additional dividend tax liability of<br />
Rs 9.86 million.<br />
Figures in Rs. mn<br />
Head Mar 2004 Mar 2003<br />
Creditors 464.60 388.20<br />
Other Liabilities 192.29 154.70<br />
Dividend and Dividend Tax 88.23 74.58<br />
Total 745.12 617.48<br />
INVESTMENT<br />
Investments rose from Rs 161.12 million in 2002-03 to<br />
Rs 299.65 million in 2003-04 due to an investment in the<br />
company’s subsidiaries located in various international<br />
geographies.<br />
INVENTORY<br />
Raw materials inventory increased from Rs.150.78 million<br />
in 2002-03 to Rs 310.10 million in 2003-04, mainly on<br />
account of the need to hold a larger quantity of material to<br />
service a larger product basket for the domestic, export<br />
and API businesses. The company is actively working<br />
towards rationalising this inventory through superior supply<br />
chain and vendor management processes. Finished goods<br />
and work-in-process inventory increased from Rs. 290.17<br />
million in 2002- 03 to Rs 508.93 million in 2003-04.<br />
RETURN ON CAPITAL EMPLOYED<br />
To prepare itself for a post-GATT environment, the<br />
company made significant investments in its research &<br />
development and manufacturing facilities, brand acquisition<br />
and marketing. All these initiatives are expected to yield<br />
returns in the years to come, strengthening the company’s<br />
return on employed capital.<br />
Figures in Rs. Mn<br />
Head Mar 2004 Mar 2003<br />
Net Fixed Assets (incl. WIP) 1621.64 1235.65<br />
Net Current Assets 1841.25 1606.81<br />
Sum: Capital employed 3462.89 2842.46<br />
EBITDA 734.89 645.43<br />
ROCE (%) 21.22 22.70<br />
40
OPPORTUNITIES<br />
Low per capita expenditure on pharmaceuticals<br />
India has one of the lowest per capita health care<br />
expenditures in the world, which is likely to correct over<br />
the coming years. For instance, India’s per capita<br />
expenditure on pharmaceuticals is only USD 4, well below<br />
USA (USD 1992), Canada (USD 1483), Germany (USD<br />
1819) and United Kingdom (USD 1415).<br />
Privatisation of insurance<br />
Presently, only two million Indians - 0.2 per cent of the<br />
population - are medically insured even as a recent study<br />
indicates that 75 per cent are potentially insurable.<br />
Insurance companies have estimated that household<br />
healthcare spending will rise from 2 per cent to 6 per cent<br />
in the coming years, translating into attractive growth for<br />
India’s pharmaceutical industry.<br />
Rising income levels<br />
Rising incomes and a growing number of the elderly,<br />
sustained by advances in hygiene and medicine,<br />
are driving a shift in the market away from vitamins,<br />
anti-infectives and gastrointestinal treatments towards<br />
products that treat cardiovascular problems, central<br />
nervous system disorders, diabetes and other complex<br />
ailments. By 2010, cardiovascular and central nervous<br />
system treatments will account for a higher share of<br />
remedies provided. This is expected to result in a faster<br />
growth for companies like <strong>Glenmark</strong> that specialise in<br />
related niches.<br />
Rural opportunity<br />
Presently, 76 per cent of the Indian pharmaceutical off take<br />
transpires in urban centres. The four metros namely Delhi,<br />
Mumbai, Kolkata and Chennai account for about a fourth of<br />
the entire Indian pharmaceutical market. Within rural India,<br />
the market is concentrated in areas where the level of<br />
infrastructure development is relatively high. According to<br />
the World Development Report 2000, only 50 per cent of<br />
the population in India has access to healthcare facilities. In<br />
rural areas, this percentage is lower. As penetration levels<br />
improve, a broader growth for India’s pharmaceutical<br />
industry is expected.<br />
Generics opportunity<br />
This has been discussed in detail earlier.<br />
THREATS<br />
The implementation of GATT from 2005 represents the<br />
biggest threat facing Indian pharmaceutical industry. India will<br />
recognise product patents, thus reducing process reverse<br />
engineering opportunities. Indian companies that have not<br />
prepared for this reality will face intense competition and<br />
perhaps even de-grow over the coming years.<br />
OUTLOOK<br />
<strong>Glenmark</strong>’s short-term and long-term outlook appears<br />
encouraging for the following reasons:<br />
• An integrated approach to markets – a presence in R&D,<br />
bulk actives and formulations with an increasing coverage<br />
of markets with its own sales force.<br />
• A strong focus on monetising each element in its<br />
integrated chain.<br />
• A focus on expanding to new global geographic markets<br />
with customised strategies.<br />
• A horizontal and vertical expansion in therapeutic<br />
segments in all target markets.<br />
• Established skills and a track record of engineering and<br />
launching drugs in different therapeutic segments with low<br />
cycle times – often as the first-mover in the target<br />
markets.<br />
• Wider doctor reach through aggressive marketing.<br />
• Investments in upgrading capabilities to service a higher<br />
revenue target including asset creation, investments in<br />
understanding regulatory requirements, alliances etc.<br />
• Investments in building IPR assets – new drugs and<br />
delivery systems for the future.<br />
HUMAN RESOURCES DEVELOPMENT<br />
The Company’s HR policy is to attract and retain the best<br />
talent and emerge as an employer of choice. The HR<br />
activities at <strong>Glenmark</strong> assume significance when viewed<br />
from the perspective of growing operational complexity<br />
and resultant diversity in human resource skill-sets<br />
required:<br />
• The marketing executives in pharmaceutical companies<br />
need to possess a good medical knowledge in diverse<br />
therapeutic areas and need to build and manage<br />
relationships with the doctors. As the company has<br />
diversified into different country markets, local marketing<br />
41
executives have been recruited. The resultant pool of sales<br />
and marketing staff has grown more complex and diverse –<br />
encompassing employees in different socio-cultural and<br />
regulatory settings.<br />
• Though pharmaceutical manufacturing is an area of<br />
Indian strength, the stringent compliance of manufacturing<br />
facilities and systems to the needs of diverse regulatory<br />
agencies mandates improved skill-sets for the employees<br />
running these plants.<br />
• The company requires a growing skill range as it grows<br />
diverse businesses like APIs, exports to semi-regulated<br />
markets, etc. Similarly as the company expands the scope<br />
and intensity of its research activities, the company needs<br />
to recruit and retain the best scientists and Ph.D talent to<br />
drive its R&D agenda.<br />
• Supporting and managing the resultant complexity calls<br />
for broader skill-sets in diverse support areas such as<br />
finance, HR, IT, etc.<br />
During the year under review, the company embarked on<br />
several initiatives targeted at attracting, retaining and<br />
motivating a diverse employee set to manage the<br />
increasing complexity in the business. These included: a<br />
cadre-based employee classification method to devise<br />
relevant remuneration policies and plan career<br />
development requirements. The company also installed a<br />
balanced-scorecard system to set and distribute<br />
performance targets aligned to the company’s goals and<br />
measure/control their achievement. These first-wave<br />
initiatives and other complementary systems that were<br />
implemented in the year under review will be further<br />
strengthened in the following year.<br />
REGULATORY COMPLIANCE SYSTEMS<br />
Since pharmaceutical products affect human lives in a<br />
direct way, the corresponding raw material quality is<br />
stringently regulated by the health authorities of the<br />
various countries. These authorities govern each aspect of<br />
bringing a drug to the market; moreover, these regulatory<br />
agencies keep raising their quality benchmarks in response<br />
to consumer concerns. In view of this, a manufacturer<br />
seeking to serve these countries requires to invest in,<br />
closely track and comply with this evolving regulatory<br />
environment. Any delay in compliance could potentially<br />
lead to a staggered market entry and lost business<br />
opportunities.<br />
Over the years, <strong>Glenmark</strong> has built a dedicated Regulatory<br />
Affairs team engaged in tracking and building protocols to<br />
comply with the stringent regulatory requirements across<br />
geographies. It has also extended this ability into a<br />
confidence-building documentation system, which proves<br />
the quality of the company’s products as safe for<br />
consumption and customised to the precise requirements<br />
of these geographies. Recent sales to regulated markets<br />
like Canada as well as the rapid development of product<br />
dossiers for registration in the European Union and US are<br />
examples of how the company has translated its capability<br />
into a growing market presence.<br />
The company has also initiated several steps to make its<br />
plants ready for global inspections and certification by<br />
USFDA, MHRA (UK), MCC (South Africa), ANVISA (Brazil)<br />
etc.<br />
INTERNAL CONTROL AND SYSTEMS<br />
<strong>Glenmark</strong>’s adequate controls cover a comprehensive<br />
definition of individual roles and responsibilities, an<br />
effective feedback flow to facilitate effective monitoring<br />
and a responsible internal audit process.<br />
SEGMENTAL ANALYSIS<br />
<strong>Glenmark</strong> has only one segment – pharmaceuticals.<br />
CAUTIONARY STATEMENT<br />
Statements in the Management Discussion and Analysis<br />
Report describing the Company’s objective,<br />
projections and estimates are forward looking statements<br />
and progressive within the meaning of applicable<br />
Security Laws and Regulations. Actual results may vary<br />
from those expressed or implied depending upon<br />
economic conditions, government policies and other<br />
incidental factors.<br />
42
Risk management<br />
STRATEGY RISK<br />
A strategic error could result in poor<br />
profits.<br />
Risk management<br />
The management at <strong>Glenmark</strong> is<br />
convinced that its strategy -<br />
dovetailed with the emerging industry<br />
environment whereby India will<br />
recognise product patents (as<br />
opposed to process patents) - is<br />
prudent with regard to the<br />
circumstances. It will enable the<br />
company to enhance shareholder<br />
value on a sustainable basis through a<br />
number of initiatives.<br />
• For instance, the company expects<br />
to emerge as a cost-effective,<br />
integrated manufacturer of products<br />
directed at the advanced markets in<br />
growing therapeutic areas.<br />
• It has made researched choices in<br />
its target markets reinforced through<br />
meaningful alliances and acquisitions<br />
and the presence of an adequate<br />
marketing team.<br />
• It has commissioned state-of-the-art<br />
plants with an eye to obtaining<br />
important regulatory approvals.<br />
• In its NCE endeavours, <strong>Glenmark</strong> is<br />
looking to license successful products<br />
following early clinical trials, resulting<br />
in a quicker inflow of revenues.<br />
This comprehensive strategy, in line<br />
with <strong>Glenmark</strong>’s vision to migrate<br />
from being an India-centric<br />
pharmaceutical company into a global<br />
research-led organisation, is being coordinated<br />
by a committed senior<br />
management team with a deep<br />
industry experience.<br />
BUSINESS PORTFOLIO RISK<br />
<strong>Glenmark</strong> runs the risk of spreading<br />
itself thin over a large portfolio of<br />
businesses rather than concentrating<br />
its resources on a specific segment.<br />
Risk management<br />
<strong>Glenmark</strong> has paid a considerable<br />
attention to the selection of its<br />
business mix. It has recognised that<br />
domestic revenues and profits could<br />
come under increasing pressure. To<br />
counter this anticipated impact, it has<br />
proactively consolidated its<br />
longstanding leadership in key<br />
segments like dermatology and<br />
entered new/growing segments.<br />
The revenue slowdown from within<br />
India is also being replaced by the<br />
company’s growing presence in<br />
international markets. In each of the<br />
key overseas markets, the company<br />
has built a strong local management<br />
team who can customise decisionmaking<br />
in line with the terrain<br />
requirements. The company has also<br />
strengthened its internal systems to<br />
respond with a greater speed to the<br />
market complexities that will arise as<br />
a result of the geographic<br />
diversification. Besides, <strong>Glenmark</strong><br />
possesses a strong organisational<br />
critical mass that will enable it to<br />
allocate adequate resources to these<br />
segments to mitigate any possibility<br />
of a dilution in focus and priority.<br />
RESEARCH RISK<br />
Original research is expensive, time<br />
consuming and faces a low probability<br />
of commercial viability. Since there is<br />
an uncertainty with regard to when<br />
commercialisation could indeed be<br />
possible, a sustained delay could lead<br />
43
to significant losses.<br />
Risk management<br />
Following 2005, when India begins to<br />
respect product patents, companies<br />
that historically process-reengineered<br />
drugs as their core focus will need to<br />
look at alternative income streams. In<br />
our opinion, investments in original<br />
drug research will be critical for<br />
survival: companies will need to<br />
discover new processes or create<br />
new products.<br />
<strong>Glenmark</strong> is doing just that: the<br />
company is engaged in original drug<br />
research in the asthma/COPD and<br />
diabetes/obesity segments. These<br />
segments were selected after an<br />
exhaustive study across the following<br />
parameters: the existing and<br />
projected size of the therapeutic<br />
segment, prospective competition in<br />
an unprotected patent environment,<br />
the size of the potential demand as<br />
well as the scope latent in the<br />
products for attractive value-addition.<br />
<strong>Glenmark</strong>’s NCE research team<br />
possesses deep and broad-based skill<br />
sets. It is working on parallel targets<br />
to maximise its success prospects.<br />
The company’s optimism is derived<br />
from promising results already<br />
achieved within a short period: some<br />
drug candidates are scheduled to<br />
enter Phase I clinical trials in the<br />
coming year. The company’s R&D<br />
initiative also translated into the<br />
development of several patentprotected<br />
platform technologies for<br />
Drug Delivery Systems, which augurs<br />
favourably for the long-term.<br />
COMPETITION RISK<br />
The Indian pharmaceutical market is<br />
highly competitive. This can impact<br />
the company’s revenues and profits.<br />
Risk management<br />
Despite the recent price decline, the<br />
Indian market appears attractive from<br />
a long-term perspective for a number<br />
of reasons:<br />
The pharmaceutical consumption per<br />
capita was US $ 412 in Japan and<br />
only US $ 3 in India. As incomes rise,<br />
we expect that India’s pharmaceutical<br />
industry’s CAGR will accelerate,<br />
which is an adequate incentive to be<br />
present within the industry. Besides,<br />
India possesses attractive potential to<br />
grow its role within the international<br />
space as well: a vast population of<br />
graduates with a technical<br />
background, skill maturity in synthetic<br />
chemistry, product re-engineering<br />
expertise and a low cost base for<br />
manufacture. It is our conviction that<br />
the evolving global market will be<br />
compatible with this Indian profile:<br />
drugs that accounted for US $90<br />
billion sales in 1995 are expected to<br />
go off-patent by 2005. In response to<br />
this, <strong>Glenmark</strong> is leveraging its India<br />
advantage to cater to the global<br />
generics markets, which will help it<br />
more than compensates for any<br />
revenue slowdown in India.<br />
CURRENCY RISK<br />
The company may suffer losses from<br />
arising from currency fluctuations.<br />
Risk management<br />
To contain the risk arising from<br />
volatile forex movements, the<br />
company selectively hedged its forex<br />
positions. The company has instituted<br />
a competent forex management<br />
system.<br />
ENVIRONMENT RISK<br />
Pharmaceutical companies generate<br />
gaseous, solid and liquid emissions,<br />
which could be a risk if these exceed<br />
the mandatory limits stipulated by the<br />
regulatory authorities.<br />
Risk management<br />
<strong>Glenmark</strong> is committed to sound and<br />
responsible environment<br />
management. This is reflected in the<br />
company’s impeccable record: it has<br />
never been censured by regulatory<br />
authorities for environmental noncompliance.<br />
Over the years, <strong>Glenmark</strong><br />
has invested in world-class pollutionmitigating<br />
plants and practices, which<br />
have helped treat its solid, liquid and<br />
gaseous waste effectively.<br />
44
Four year financial summary<br />
Balance Sheet<br />
(Rs. in '000)<br />
2000-01 2001-02 2002-03 2003-04<br />
Share Capital<br />
Equity capital 101105 101404 101811 118546<br />
Preference share application money 100000 – – –<br />
Preference capital – 100000 100000 100000<br />
Share capital 201105 201404 201811 218546<br />
Total reserves 1093374 1081872 1323944 2132109<br />
Capital and reserves 1294479 1283276 1525755 2350655<br />
Secured Loans 525390 697711 1193564 1029429<br />
Unsecured Loans 196002 73385 53864 119314<br />
Total loans 721392 771096 1247428 1148743<br />
Deferred Tax Liability – 204873 269856 293181<br />
Total 2015871 2259245 3043039 3792579<br />
Gross Block 1315325 1416755 1246518 1723534<br />
Less: Depreciation 88170 175581 243340 346864<br />
Net Block 1227155 1241174 1003178 1376670<br />
Capital work-in-progress 61784 232471 244966<br />
Investments 17339 26158 161126 299647<br />
Current Assets<br />
Inventories 348938 322280 444371 821601<br />
Sundry Debtors 468612 742102 1236691 1304813<br />
Cash & bank 71702 45342 40518 67694<br />
Loans & advances 112343 246545 502704 392263<br />
Total current assets 1001595 1356269 2224284 2586371<br />
Current liabilities<br />
Sundry Creditors 196816 191620 325585 371758<br />
Provisions 48566 64872 11717 39772<br />
Others 53295 228657 280177 333591<br />
Total current liabilities 298677 485149 617479 745121<br />
Net Current Assets (Working Capital) 702918 871120 1606805 1841250<br />
Misc Expenses 68459 59009 24076 6419<br />
Deferred tax assets – – 15383 23627<br />
Total 2015871 2259245 3043039 3792579<br />
45
Profit and Loss Account<br />
(Rs. in '000)<br />
2000-01 2001-02 2002-03 2003-04<br />
Turnover 1907820 2607555 3336365 3806606<br />
Less: Sales tax 137022 177928 251600 255771<br />
Less: Excise duty paid 251473 250824 327169 308398<br />
Net Sales 1519325 2178803 2757596 3242437<br />
Other Income 36510 60695 42823 34573<br />
Total Income 1555835 2239498 2800419 3277010<br />
Salary, wages and labour charges 55515 51808 90698 120547<br />
Consumption of raw materials 579265 605091 712886 966088<br />
Purchase of trading goods 123196 224898 306895 286013<br />
Power and fuel 8316 6059 7462 31365<br />
(Increase)/ decrease in inventory (129572) 64757 (77942) (218758)<br />
Others 37248 20435 25312 43767<br />
Cost of sales 673968 973048 1065311 1229022<br />
Employee Cost 155332 214195 269585 349932<br />
Promotion expenses 120525 112714 184292 184386<br />
Other selling and operating expenses 263610 359303 488540 530708<br />
Total selling and operating expenses 539467 686212 942417 1065026<br />
Research and development 22070 78110 147257 248073<br />
Total expenses 1235505 1737370 2154985 2542121<br />
EBIDT 320330 502128 645434 734889<br />
Interest 83231 129134 106884 100578<br />
EBDT 237099 372994 538550 634311<br />
Depreciation 50951.00 88226 103326 108891<br />
PBT 186148 284768 435224 525420<br />
Current tax 15260 22607 35300 90298<br />
Deferred tax – 34396 49600 15081<br />
PAT before extraordinary items 170888 227765 350324 420041<br />
Prior period and extraordinary items – – 18429 –<br />
PAT 170,888 227765 331895 420041<br />
46
Ratios<br />
Debt Equity<br />
Debtors cycle (Turnover)<br />
1.0<br />
0.96<br />
140<br />
135.29<br />
0.8<br />
0.77<br />
120<br />
125.11<br />
0.64<br />
103.88<br />
0.6<br />
0.56<br />
100<br />
89.65<br />
0.4<br />
2000-01 2001-02 2002-03 2003-04<br />
80<br />
2000-01 2001-02 2002-03 2003-04<br />
Balance Sheet<br />
(Rs. in '000)<br />
2000-01 2001-02 2002-03 2003-04<br />
Return on Networth 15.18 20.26 23.68 18.72<br />
Return on Capital Employed (avg cap emp) 15.89 24.67 26.74 23.43<br />
Debt Equity 0.64 0.77 0.96 0.56<br />
Debtors cycle (Turnover) 89.65 103.88 135.29 125.11<br />
Creditors cycle (Cost of goods sold) 106.59 71.88 111.55 110.41<br />
Inventory Cycle (Net sale) 83.83 53.99 58.82 92.49<br />
Inventory turnover (Net Turnover) 4.35 6.76 6.21 3.95<br />
Gross Turnover/Capital employed 0.95 1.27 1.20 1.09<br />
Gross Turnover/Gross Block 145.05 184.05 267.65 220.86<br />
Net Block/Cap Emp. 60.87 60.42 36.17 39.34<br />
W.Cap/Cap Emp. 34.87 42.40 57.94 52.62<br />
47
PAT Margin<br />
Research and development/Net sales<br />
13<br />
13.00<br />
8.00<br />
7.14<br />
12<br />
12.04<br />
6.25<br />
5.34<br />
11<br />
11.25<br />
4.50<br />
2.75<br />
3.58<br />
10.45<br />
1.45<br />
10<br />
2000-01 2001-02 2002-03 2003-04<br />
1.00<br />
2000-01 2001-02 2002-03 2003-04<br />
Profit and Loss Account<br />
(Rs. in '000)<br />
2000-01 2001-02 2002-03 2003-04<br />
Revenue<br />
Domestic sales/Turnover 94.74 95.76 94.62 87.22<br />
Export sales/Turnover 5.26 4.24 5.38 12.78<br />
Excise/Turnover 13.18 9.62 9.81 8.10<br />
Margins - (basis – turnover)<br />
EBDITA margin 21.08 23.05 23.41 22.66<br />
Cash profit margin 14.59 14.50 15.77 16.30<br />
Pre tax profit margin 12.25 13.07 15.78 16.20<br />
PAT Margin 11.25 10.45 12.04 12.95<br />
Expenses<br />
Cost of sales/ Total expenses 54.55 56.01 49.43 48.35<br />
Selling and operative expenses/Total expenses 43.66 39.50 43.73 41.90<br />
Research and development/Total expenses 1.79 4.50 6.83 9.76<br />
Cost of sales/ Net sales 44.36 44.66 38.63 37.90<br />
Selling and operative expenses/Net sales 35.51 31.49 34.18 32.85<br />
Research and development/Net sales 1.45 3.58 5.34 7.65<br />
Interest cover 3.85 3.89 6.04 7.31<br />
Cost of debt 11.54 17.30 10.59 8.39<br />
Shareholder Value *<br />
EPS (Face value Rs.10/-) 16.90 22.46 34.41 7.09<br />
EPS (Net of extraordinary income) 16.90 22.46 32.60 7.09<br />
CEPS (Face value Rs.10/-) 21.94 31.16 42.75 8.92<br />
CEPS (Net of extraordinary income) 21.94 31.16 40.94 8.92<br />
Book value 111.37 110.87 137.67 37.86<br />
* Notes: During the financial year 2003-04, each paid up share of Rs. 10 has been sub-divided into a paid up share of Rs. 2 each.<br />
48
Profiles of the Directors<br />
Mr. Gracias Saldanha (Chairman)<br />
Mr. Gracias Saldhana, 67, is the founder of the Company. He has<br />
over 31 years experience in the industry. His educational<br />
qualifications include an M. Sc. from Bombay University with a<br />
Diploma in Management studies from Jamnalal Bajaj Institute of<br />
Management Studies, Mumbai. He has worked with leading<br />
pharmaceutical companies like Abbott Laboratories and E. Merck.<br />
Mrs. B.E. Saldanha (Director – Exports)<br />
Mrs B. E. Saldanha, 64, has done her B.Sc., B.Ed. from Bombay<br />
University and has been Working Director since 1982. She has<br />
taken a keen interest in developing the Company’s export<br />
business.<br />
Mr. Glenn Saldanha (Managing Director & CEO)<br />
Mr. Glenn Saldanha, 34, is a B. Pharma from Bombay University<br />
and was awarded the Watumall Foundation Award for overall<br />
excellence. His other educational qualifications include an MBA<br />
form New York University’s Leonard N. Stern School of Business<br />
(US). He has worked for Eli Lilly in the US and was a Management<br />
Consultant with Price Waterhouse Coopers. His Services have<br />
been used by Smithkline Beecham, Rhorer, Astra, Merck and<br />
Johnson and Johnson, among others.<br />
Mrs. Cheryl Pinto (Director – Corporate Affairs)<br />
Mrs. Cheryl Pinto, 37, is a graduate in Pharmacy from the<br />
University of Bombay. She has over 11 years experience in the<br />
pharmaceuticals business.<br />
Mr. Abhinna Sundar Mohanty (Director – Domestic<br />
Formulations)<br />
Mr. Abhinna Sundar Mohanty, 50, is an M.Sc. In-charge of<br />
domestic Formulations, he has over 26 years experience in<br />
pharmaceutical sales and marketing as well as healthcare sectors.<br />
Mr. R. V. Desai (Director – Finance/IT/Legal)<br />
Mr. R.V. Desai, 46, is a Science Graduate and a Chartered<br />
Accountant. In-charge of Finance /IT/Legal, he has over 20 years<br />
experience.<br />
Mr. Steven Bates<br />
Mr. Steven Bates is a CFA from UK with over 20 years experience<br />
in the financial industry. He has worked with top firms viz. James<br />
Capel, JP Morgan Fleming Asset Management, as head of Global<br />
emergence market. Mr. Bates is at present Director, Zephyr<br />
Management UK Ltd, an FSA regulated investment business<br />
investing in emerging markets. He is also an Investment Advisor<br />
to Cardiff & Vale of Glamorgan Pension Fund, Member of the<br />
Governing Board of Kosovo Pensions Savings Trust (UN<br />
appointment) and Consultant on pension reform to Russian<br />
Foundation for Social Reform (World Bank).<br />
Mr. Julio F. Ribeiro (Non–Executive)<br />
Mr. Julio Ribeiro, 74, is a retired government official and has<br />
served the country under various assignments. Amongst the major<br />
positions held, he has been the Ex-commissioner of Police,<br />
Mumbai, Former Special Secretary to Government of India,<br />
Ministry of Home Affairs, Former Director General of Police,<br />
Punjab, Ex-adviser to the Governor of Punjab, Ex-Ambassador of<br />
India to Romania and is currently a Director in IIT Corporate<br />
Services Ltd.<br />
Dr. Prasanna R. Gore (Non-Executive)<br />
Dr. Prasanna R. Gore, 39, is a Ph.D and M. S. in Pharmaceutical<br />
Marketing from University of West Virginia, Morgantown, WV and<br />
a B.Sc. from University of Bombay, India. He has over 11 years<br />
experience in the pharmaceutical and healthcare sectors ranging<br />
from pharmaceutical sales, academic faulty and strategic<br />
consulting. He was elected Vice-Chairman of the American<br />
Association of Pharmaceutical Scientists – Economic,<br />
Management, and Marketing section for the year 2000.<br />
Mr. J. M. Trivedi (Non-Executive)<br />
Mr. J. M. Trivedi, 50, is a B.Tech (Chemical Engg.) form Indian<br />
Institute of Technology, Mumbai and has a Diploma in Basic<br />
finance form ICFAI. He is Director of CDC’s Mumbai office and has<br />
more than 21 years experience in the financial sector including 13<br />
years in venture capital/private equity.<br />
Mr. Sridhar Gorthi (Non-Executive Alternate Director)<br />
Mr. Sridhar Gorthi, 32 is a BA. LLB (Hons) form the National Law<br />
School of India University. Mr. Sridhar Gorthi is presently a partner<br />
in Trilegal and has worked with Arthur Anderson and Lex Inde,<br />
Mumbai. He was involved in legal advisory services to various<br />
multinational and domestic corporations on restructuring, debt<br />
finance, joint ventures, acquisition/mergers etc.<br />
Mr. Natvarlal B. Desai (Non-Executive)<br />
Mr. Natvarlal B. Desai, 77, is a retired General Manager of Bank of<br />
Baroda. He has over 45 years experience in the Banking Sector.<br />
He has worked in India and overseas. He was Chairman of Bank of<br />
Baroda Uganda Ltd. He was the Founder and Managing Director of<br />
Equitorial Bank PLC, UK form which he retired in 1992.<br />
Mr. M. Gopal Krishnan (Non-Executive)<br />
Mr. M. Gopal Krishnan, 69, worked with Bank of India for over 25<br />
years, most of which were spent overseas in Kenya and UK.<br />
Thereafter, he worked with Equitorial Bank PLC, UK as Founder<br />
Director until he retired.<br />
49
Directors' Report<br />
Your Directors have pleasure in presenting their 26th Annual Report and Audited Accounts<br />
of the Company for the year ended March 31, 2004.<br />
FINANCIAL RESULTS<br />
Rs. in million<br />
2003-2004 2002-2003<br />
Profit before Interest, Depreciation & Tax 734.89 645.43<br />
Less: Interest 100.58 106.88<br />
Less: Depreciation 108.89 103.32<br />
Less: Tax (Current Year, Deferred Tax & for earlier years) 105.38 103.33<br />
Profit after Tax 420.04 331.90<br />
Surplus brought forward from earlier years 235.88 29.07<br />
Profit available for appropriations 655.92 360.97<br />
APPROPRIATIONS<br />
Interim Dividend on Preference Shares 10.50 10.50<br />
Interim Dividend on Equity Shares 77.02 66.11<br />
Dividend Tax 11.21 8.47<br />
Transfer to Debenture Redemption Reserve 0.25 –<br />
Transfer to General Reserves 60.00 40.00<br />
Balance carried to Balance Sheet 496.94 235.88<br />
655.92 360.97<br />
DIVIDEND<br />
Your Company has paid interim dividend @ 65 per cent<br />
on the Paid-up Equity Share Capital of the Company and @<br />
10.5 per cent on the paid up Preference Share Capital of<br />
the Company. The total outflow on account of Dividend<br />
inclusive of Dividend Tax, is Rs. 98.73 million (Rs. 85.08<br />
million). Your Directors recommend that the interim<br />
dividend already paid be taken as final dividend for the year<br />
ended 31 March 2004.<br />
RESULTS OF OPERATIONS<br />
Your Directors are pleased to report satisfactory<br />
performance for the year under review. The Company<br />
achieved gross revenue of Rs.3841.18 million<br />
(Rs. 3,379.19 million), registering an increase of 13.67 per<br />
cent over the previous year. The increase in revenue is due<br />
to the launching of new products, growth in the existing<br />
products, expansion of field staff and<br />
new initiatives taken in terms of co-marketing and<br />
bulk drugs.<br />
PROFITS<br />
The operating profit before interest, depreciation and<br />
other income increased to Rs.734.89 million from<br />
Rs. 645.43 million, an increase of 13.86 per cent over the<br />
previous year.<br />
50
OPERATIONS:<br />
PHARMA DIVISION<br />
Domestic sales at Rs.3167.38 million (Rs. 3023.24 million)<br />
registered an increase of 4.76 per cent over the previous<br />
year. The growth in the domestic segment during the<br />
period was affected due to price reductions in various new<br />
products introduced by the Company, in response to<br />
industry-wide price pressures. As per ORG report, our<br />
ranking has improved to 24 among the top 50<br />
pharmaceutical companies. Your Company introduced new<br />
brands like Tacroz, Tazret and Razel, which were well<br />
received in the market. Other brand extensions made<br />
during the year like Telma – H Tabs, Glimilin - 2 MF Tabs,<br />
Vorth Insta Gel, Valus - 20 XT, and Valus Insta Gel are<br />
expected to do well in the future.<br />
EXPORTS<br />
Exports income at Rs. 471.27 million for the year 2003-<br />
2004 showed a healthy increase as compared to<br />
Rs. 296.92 million in the previous year, showing a growth<br />
of 58.71 per cent.<br />
INTERNATIONAL BUSINESS:<br />
• Product Development and Licence Agreement with<br />
K. V. Pharmaceuticals Company, USA.<br />
<strong>Glenmark</strong> Pharmaceuticals Inc., USA (GPI), a wholly<br />
owned subsidiary of your Company has entered into a<br />
product development and marketing license agreement<br />
with the USA-based K. V. Pharmaceuticals Company<br />
(KV). <strong>Glenmark</strong>, under the agreement, will initially<br />
develop and license eight generic products to KV for<br />
regulatory approval and marketing in the North<br />
American market. These will include both Paragraph III<br />
and Paragraph IV ANDA (Abbreviated New Drug<br />
Application) filing opportunities. The alliance will help<br />
your Company to use KV’s vehicle to get its products<br />
into the US market thereby helping your Company to<br />
build its US business and get positive cash flows in the<br />
US almost immediately. The companies will also<br />
collaborate to secure US regulatory approval of the<br />
products. The two companies expect to launch the first<br />
product under the agreement during the last half of<br />
2005.<br />
• Marketing arrangement with Lannett Company Inc.,<br />
USA:<br />
Your Company through its wholly owned subsidiary in<br />
the USA viz. <strong>Glenmark</strong> Pharmaceuticals Inc., USA, has<br />
entered into a marketing arrangement with the<br />
Philadelphia-based Lannett Company Inc., USA, to<br />
market the ANDAs (Abbreviated New Drug Application)<br />
which <strong>Glenmark</strong>, USA would file over the next two<br />
years.<br />
API AND CO-MARKETING<br />
The sales of API and the Co-marketing segment of your<br />
Company increased from Rs. 286 million to Rs. 445.32<br />
million, registering a growth of 55.85 per cent over the<br />
previous year. Exports were made to nearly 20 countries.<br />
During the year, your Company developed and launched 20<br />
technologically advanced products in the global market and<br />
initiated tie-ups with two major generic players in USA and<br />
Canada. Apart from this, co-marketing arrangements were<br />
also entered into with major pharma companies in the<br />
domestic market including Ranbaxy, Dr. Reddy’s, Cadila<br />
and Torrent.<br />
FILING OF DMFs (DRUG MASTER FILE)<br />
During the year, your Company has filed an initial set of<br />
DMFs on Amiodarone, Cilastazol and Flucanozole. In April<br />
2004, your Company filed a fourth DMF for Glimiperide.<br />
DRUG DISCOVERY RESEARCH AND DEVELOPMENT<br />
CENTRE AT MAHAPE, NAVI MUMBAI<br />
The working of the R&D centre is satisfactory. As stated<br />
earlier, the Company's research activities are focussed in<br />
the areas of diabetes, asthma, obesity, pain and<br />
inflammation.<br />
Your Company has developed GRC 3886, a highly selective<br />
PDE 4 inhibitor targeting the treatment of asthma. The<br />
molecule is completing its pre-clinical studies with a<br />
contract research organisation in Europe and is expected to<br />
enter Phase-I clinical trials in UK in July-August 2004. Your<br />
51
Company is in the final stages of closing a deal with<br />
Quintiles, a leading global CRO, to conduct the Phase-I<br />
clinical trials.<br />
Your Company is also working on an undisclosed target for<br />
diabetes and has developed several promising candidates,<br />
one of which will be selected for Phase-I clinical trials later<br />
in 2004-05. On account of the expected superiority of<br />
these compounds over those developed for the Beta-3<br />
agonist area, the erstwhile project researching Beta-3<br />
agonists for diabetes has been put on hold.<br />
Your Company has several back up compounds in the PDE-<br />
4, Beta-3 agonist and the undisclosed diabetes target<br />
space which can be studied further, subject to the<br />
performance of the lead compounds in Phase-I clinicals.<br />
FIRE AT COMPANY’S MANUFACTURING FACILITY AT<br />
KURKUMBH<br />
The manufacturing operations at the leased factory of the<br />
Company at Kurkumbh were suspended temporarily due to<br />
fire in the factory. The Company did not suffer significant<br />
losses/ loss of sales as the production was moved to<br />
Ankleshwar factory, and also because the plant was fully<br />
insured.<br />
PLANT AT GOA<br />
The company has commissioned a plant at Goa that has<br />
been built to meet the stringent USFDA certification<br />
standards. The plant will allow <strong>Glenmark</strong> to serve the<br />
regulated markets overseas in the coming years.<br />
Formulation research centre at Sinnar (Nasik)<br />
The centre works on developing formulations for sale in<br />
India and semi-regulated export markets. Several products<br />
have been developed at this centre and they are being<br />
launched in the market.<br />
EMPLOYEE STOCK OPTION SCHEME<br />
During the year, your Company formulated a new<br />
Employees Stock Option Scheme, ESOS 2003, pursuant to<br />
which options have been issued to the employees of the<br />
Company. On exercising the convertible options so<br />
granted, the paid-up equity share capital of the company<br />
will increase by a like number of shares.<br />
The details of stock options granted by the Company are<br />
disclosed in compliance with clause 12 of the Securities<br />
Exchange Board of India (Employee Stock Options Scheme<br />
and Employee Stock Purchase Scheme), 1999 and set out<br />
in the Annexure to this Report.<br />
ACQUISITIONS<br />
During the year your Company has acquired a local<br />
manufacturing Company having an ANVISA approved plant<br />
in Brazil, for a consideration of US $ 5.25 million. The<br />
acquisition will facilitate entry in the Brazilian and other<br />
Latin American markets.<br />
Your Company has also acquired a bulk drugs intermediate<br />
manufacturing plant at Solapur for a consideration of<br />
Rs. 17.80 million. This will help the Company in increasing its<br />
capacity for manufacturing bulk drugs.<br />
SUBSIDIARY COMPANIES<br />
Pursuant to the provision of Section 212 of the Companies<br />
Act, 1956 the Audited Accounts as of 31 March 2004<br />
together with the reports of the Directors and Auditors of<br />
the subsidiary companies, viz. <strong>Glenmark</strong> Exports Limited,<br />
GM Pharma Limited, <strong>Glenmark</strong> Pharmaceuticals Inc, USA,<br />
<strong>Glenmark</strong> Farmaceutica Ltda, <strong>Glenmark</strong> Philippines Inc. and<br />
<strong>Glenmark</strong> Impex LLC are attached.<br />
Your Company has incorporated wholly owned subsidiaries<br />
in Philippines, Brazil, Nigeria, United Kingdom, the United<br />
States of America and Switzerland.<br />
SUB-DIVISION OF EQUITY SHARES<br />
The face value of the shares of the Company have been<br />
split from Rs.10/- per share to Rs.2/- per share.<br />
FINANCE<br />
Your Company was able to reduce its cost of borrowings<br />
from Rs. 106.90 million in 2002-2003 to Rs. 100.58 million<br />
in 2003-2004, by prepaying /repaying high cost debt and<br />
replacing the same with low cost borrowings. The<br />
Company was able to source funds for short term<br />
requirements at lower rates, due to its rating of AA- by<br />
52
CRISIL. The 5 per cent FCDs of Rs.50 crore issued to CDC<br />
Financial Services (Mauritius) Ltd and South Asia Regional<br />
Fund were converted into equity shares of the Company at<br />
a price of Rs. 305.42 per share of face value of Rs. 10/-<br />
(current face value of equity shares of the Company is<br />
Rs. 2/-per share).<br />
DIRECTORS<br />
Mr. Glenn Saldanha, Mrs. Cheryl Pinto and Mr. J.F. Ribeiro<br />
retire by rotation and are eligible for re-appointment. Mr.<br />
Steven Bates was appointed as Additional Director of the<br />
Company on 22 April 2004. Mr. Bates holds office upto the<br />
date of the ensuing Annual General Meeting. The Company<br />
has received a notice in writing pursuant to the provisions<br />
of Section 257 of the Act from a member of the Company<br />
proposing his appointment as Director of the Company.<br />
During the year Mr. Sameer Paigankar resigned from the<br />
Board of the Company with effect from 29 March 2004.<br />
Your Directors wish to place on record their appreciation of<br />
the valuable services rendered by him during his tenure as<br />
Director on the Board.<br />
CORPORATE GOVERNANCE<br />
The Corporate Governance and Management’s Discussion<br />
and Analysis Report form an integral part of this Report and<br />
are set out as separate Annexures to this Report. The<br />
Certificate of the Auditors of the Company certifying<br />
compliance with the conditions of Corporate Governance<br />
as stipulated in clause 49 of the Listing Agreement with<br />
Stock Exchanges is annexed with the report on Corporate<br />
Governance.<br />
AUDITORS<br />
M/s Price Waterhouse, Chartered Accountants, Auditors of<br />
the Company, retire at the conclusion of the ensuing<br />
Annual General Meeting and being eligible, offer<br />
themselves for re-appointment.<br />
HUMAN RESOURCES<br />
Industrial relations continued to be cordial. Information as<br />
per Section 217(2A) of the Companies Act, 1956, read with<br />
the Company’s (Particulars of Employees, Rules, 1975)<br />
forms part of this Report.<br />
COMPANIES (DISCLOSURE OF PARTICULARS IN THE<br />
REPORT OF THE BOARD OF DIRECTORS) RULES 1988.<br />
The particulars as prescribed under the Rules appear in the<br />
Annexures forming part of the Directors Report.<br />
DIRECTORS' RESPONSIBILITY STATEMENT<br />
The Directors confirm that –<br />
(i) in the preparation of the annual accounts, the applicable<br />
accounting standards have been followed along with<br />
proper explanation relating to material departures;<br />
(ii) appropriate accounting policies have been selected and<br />
applied consistently and have made judgments and<br />
estimates that are reasonable and prudent so as to give<br />
a true and fair view of the state of affairs of the<br />
Company as at 31 March 2004 and of the profit of the<br />
Company for the year ended 31 March 2004;<br />
(iii) proper and sufficient care has been taken for<br />
maintenance of adequate accounting records in<br />
accordance with the provisions of the Companies Act,<br />
1956 for safeguarding the assets of the Company and<br />
for preventing and detecting fraud and other<br />
irregularities;<br />
(iv) the annual accounts have been prepared on a going<br />
concern basis.<br />
APPRECIATION<br />
Your Directors express their gratitude to the Company’s<br />
customers, shareholders, business partners viz. distributors<br />
and suppliers for their understanding and support.<br />
Your Directors record their appreciation and gratitude to the<br />
financial institutions and banks for their continued and<br />
timely assistance in meeting the Company’s resource<br />
requirement. Finally your Directors acknowledge the<br />
dedicated services rendered by all the employees of the<br />
Company.<br />
For and on behalf of the Board of Directors<br />
Mumbai<br />
G. Saldanha<br />
Date: July 5, 2004<br />
Chairman<br />
53
Annexure to the<br />
Directors’ Report<br />
ANNEXURE-A<br />
Information under section 217(1)(e) of the Companies Act,1956 read with Companies<br />
(Disclosure of Particulars in the Report of the Board of Directors) Rules, 1988 and<br />
forming part of the Directors’ Report.<br />
A. CONSERVATION OF ENERGY<br />
Energy Generation Measures Taken<br />
A. Power and Fuel Consumption Current Year Previous Year<br />
1. Electricity<br />
(a) Purchased<br />
Unit (in '000 Kwhrs) 4947.07 1591.10<br />
Total Amount (Rs. in '000's) 25510.30 7597.35<br />
Rate/Unit (Rs.) 4.65 4.78<br />
(b) Own Generation<br />
(i) Through Diesel Generator<br />
Unit (in '000 Kwhrs) 136.28 79.20<br />
Units per Ltr. of Diesel Oil 3.19 1.56<br />
Cost/Unit (Rs) 7.05 6.60<br />
(ii) Through Steam Turbine/Generator Nil Nil<br />
2. Coal Nil Nil<br />
Qty.<br />
Total Cost<br />
Avg. Rate<br />
3. Furnace Oil<br />
Qty. (K. Ltr.) 183.20 40.00<br />
Total Amount (Rs. in '000's) 3297.60 720.00<br />
Avg. Rate (Rs. /K. Ltr.) 18.00 18.00<br />
4. i. Internal generation<br />
Light Diesel Oil<br />
Qty. (In Ltr. '000's) 101.41 132.01<br />
Total Cost (Rs. in '000's) 2218.80 2247.00<br />
Rate/Unit (Rs.) 21.88 17.03<br />
ii. Natural Gas<br />
Qty. (M 3 '000's) 476.79 Nil<br />
Total Cost (Rs. in '000's) 4767.87 Nil<br />
Rate/Unit (Rs.) 10.00 Nil<br />
B<br />
Consumption<br />
The Company manufactures several Drug Formulations in different pack sizes. In view of this, it is impracticable to<br />
apportion the consumption and cost of utilities to each Product/Formulation.<br />
54
B. TECHNOLOGY ABSORPTION, RESEARCH &<br />
DEVELOPMENT (R&D)<br />
1. Specific areas in which R & D is carried out by the<br />
Company<br />
a) Pharmaceutical Formulation Development: Formulation<br />
of various pharmaceutical dosage forms and processes<br />
for new & existing molecules. Development of<br />
formulations as immediate release, delayed release,<br />
sustain release and various platform technologies. This<br />
includes literature survey, preformulation studies,<br />
formulation and standardisation of dosage forms for<br />
selected drug molecules on laboratory scale.<br />
b) Development of technology based products like<br />
Carbonyl iron floating tablets, Clotrimazole<br />
Mucoadhesive vaginal tablets, Esomeprazole plus<br />
Domperidone SR, Rosiglitazone plus Metformin SR, line<br />
extensions of X-Met SR for launch in the domestic<br />
segment.<br />
c) Development of ANDA for the US markets: Successful<br />
technology transfer to sites in the US and Canada.<br />
Development of 7 more ANDA’s with successful pilot<br />
BE data and ready for transfer to manufacturing site by<br />
September – October 2004.<br />
2. Analytical Method Development :<br />
a) Development of new analytical test processes and their<br />
evaluation for dosage forms. This includes stability<br />
indicating methods, validation and standardisation of<br />
analytical processes. Accelerated and time lapse<br />
stability studies of Research and Development<br />
formulations under various climatic conditions.<br />
b) Development of technology based patentable platform<br />
technology. Filing of 8 patents in the US and PCT for<br />
various dosage form including oral controlled release<br />
and topical products.<br />
3. Packaging Material Development :<br />
Development of packaging forms and their improvements<br />
for new as well as existing products.<br />
4. Technology Transfer:<br />
As per the technology transfer protocols for various<br />
formulation, scale up for production / commercial batches.<br />
Monitoring of first three to five commercial batches.<br />
Setting-up of pelletisation facility and transfer of technology<br />
for Esomeprazole pellets and Itraconazole pellets at<br />
Ankleshwar. Esomeprazole pellets are under regular<br />
manufacture for exports as well as domestic market.<br />
5. Benefits derived as a result of the above R&D<br />
R&D has developed the new formulations for new &<br />
existing molecules & drug combinations. Developed<br />
technology is transferred to commercial production for<br />
following products;<br />
<strong>Glenmark</strong> Gracewell Healtheon<br />
Glevo 750 Tablets Tacroz Ointment Ezzicad Tablets<br />
(Levofloxacin) (Tacrolimus) (Ezetimibe)<br />
Candid Microspray Tazret Gel Lerez AT Tablets<br />
(Clotrimazole 1%) (Tazarotene) (Lercanidipine Hcl + Atenolol)<br />
Valus Tablets Tazret Forte Cream Telma H Tablets<br />
(Valdecoxib) (Tazarotene) (Telmisartan + Hydrochlorothiazide)<br />
Stiloz Tablets Maclar Dry Syrup Razel Tablets<br />
(Cilostazol) (Clarithromycin for oral suspension) (Rosuvastatin)<br />
Kretos Tablets<br />
Kefpod Suspension<br />
(Etoricoxib)<br />
(Cefpodoxime Proxetil for oral suspension)<br />
Deriva – C Gel<br />
(Adapalene + Clindamycin)<br />
Ebov Tablets (Etoricoxib)<br />
Vorth Tablets (Valdecoxib)<br />
The products launched and sold in the domestic and exports markets would earn revenues for your Company. The IPR<br />
generated can lead to profitable licensing opportunities.<br />
55
6. Future plan of action<br />
R&D is working on new molecules in the following<br />
segment;<br />
- Antihypertensive molecules<br />
- Antifungal molecules<br />
- Antibacterial molecules<br />
- Antiasthmatic molecules<br />
- Antidiabetic products<br />
- Technology – such as microspheres & aerosols.<br />
- Technology – to replace solvents used in film coating by<br />
water.<br />
- Development of formulations for Semi regulatory market.<br />
- Development of formulations for Latin American market.<br />
Your Company is targeting development and technology<br />
transfer and ANDA filing for 10-12 products for the coming<br />
year. These products will be transferred to the Goa plant.<br />
7. Expenditure on R & D:<br />
(Rs. in million)<br />
2003-04 2002-03<br />
a) Capital Expenditure 123.55 158.61<br />
b) Revenue Expenditure 248.07 147.26<br />
c) Total 371.62 305.87<br />
d) R & D Expenditure as a 9.67% 9.16%<br />
percentage of total turnover<br />
TECHNOLOGY ABSORPTION, ADOPTION AND<br />
INNOVATION:<br />
1. Efforts in brief towards technology absorption,<br />
adoption and innovation.<br />
Most of our efforts in the area of technology absorption,<br />
adoption and innovation are based on our own efforts in R<br />
& D. They include improvement in yield and quality,<br />
improvement of processes and development of new<br />
processes with validation studies.<br />
2. Benefits derived :<br />
Benefits derived are enhanced production of our products,<br />
improvement in the yield and quality of products and<br />
introduction of new products, cost reduction of products<br />
and processes without affecting the quality of the products<br />
and process efficacy.<br />
Our R&D Centre is recognised by D.S.I.R., Ministry of<br />
Science and Technology, Government of India.<br />
3. Information regarding technology imported during the<br />
last five years - Nil<br />
C. FOREIGN EXCHANGE EARNINGS AND OUTGO<br />
Total foreign exchange earned was Rs. 489.54 Million and<br />
outflow was Rs. 533.05 Million.<br />
For and on behalf of the Board of Directors<br />
Mumbai<br />
G. Saldanha<br />
Date: July 5, 2004<br />
Chairman<br />
56
ANNEXURE-B<br />
Information of employees under Section 217 (2A) of the Companies Act, 1956 read with Companies (Particulars of<br />
Employees) Rules, 1975.<br />
Particulars of employees :<br />
Sl Name Age Designation Remuneration Qualification Experience Date of Last<br />
No Yrs Received (Rs.) Yrs Commencement Employment<br />
of Employment<br />
1. B. E. Saldanha 64 Director-Exports 7,177,090 B. Sc, B. Ed., 33 01.01.1982 Walsingham House School<br />
2. Glenn Saldanha 34 Managing Director 14,559,510 B. Pharm., 10 01.08.1998 Price Waterhouse<br />
& Chief Executive Officer MBA (USA) Coopers, USA<br />
3. Cheryl Pinto 37 Director-Corporate Affairs 7,410,921 B. Pharm. 11. 01.08.2000 Cheryl Laboratories (Pvt.)<br />
Limited<br />
4. R. V. Desai 46 Director-Finance 3,124,062 B. Sc. , F.C.A 22 02.01.1984 Progressive Consultants<br />
IT & Legal Limited<br />
5. A. S. Mohanty 50 Director-Formulations 3,186,542 M. Sc. 27 25.03.1983 Alembic Limited<br />
6. S. A. Paigankar* 34 Director- Strategic 3,106,677 B. Pharm., MBA 12 24.05.1999 Smith & Nephew<br />
Planning Healthcare Limited<br />
7. K. Anand* 48 Sr.V.P.- QA & Regulatory 13,09,495 M. Sc. 28 17.10.2003 Nicholas Piramal India Ltd.<br />
Affairs<br />
*Employed for part of the year.<br />
Notes:<br />
i) Remuneration includes Salary, Allowances, Bonus, Company’s Contribution to Provident Fund and Superannuation Fund, Medical Benefits, LTA and perquisites.<br />
ii) Mrs. B. E. Saldanha, Mr. Glenn Saldanha and Ms. Cheryl Pinto are related to each other as defined under Section 6 of the Companies Act, 1956.<br />
iii) The nature of employment in all cases is contractual.<br />
For and on behalf of the Board of Directors<br />
Mumbai G. Saldanha<br />
Date: July 5, 2004 Chairman<br />
57
Disclosure pursuant to the provisions of Securities and Exchange Board of India (Employee<br />
Stock Option Scheme and Employee Stock Purchase Scheme) Guidelines, 1999.<br />
A. <strong>Glenmark</strong> Employee Stock Option Scheme ‘1999<br />
a) Options granted 6,70,000<br />
b) The Pricing Formula • Book Value as on 31st March, 1999 in<br />
respect of 2,85,000 options<br />
• Market price as on 1st January, 2001 in<br />
respect of 3,85,000.<br />
c) Options vested 5,85,000<br />
d) Option exercised 5,85,000<br />
e) The total number of shares arising as a result of exercise of option. 5,85,000<br />
f) Options lapsed NIL<br />
g) Variation of terms of options NIL<br />
h) Money realised by exercise of option Rs.1,36,47,200/-<br />
i) Total number of options in force as on 31st March’2004 85,000<br />
j) Employee wise details of options granted to:<br />
i) Senior Managerial personnel i) Mr. Sameer Paigankar<br />
ii) Dr. B. Gopalan<br />
iii) Dr. A. Lakdawala<br />
iv) Mr. A.S. Mohanty<br />
v) Mr. Rajesh V. Desai<br />
vi) Mr. Dereck Lobo<br />
vii) Mr. J. A. Mograwala<br />
viii) Dr. Shekhar Bhaskar Bhirud<br />
ix) Dr. Subramaniyam Duvuuri<br />
ii) Any other employee who receives a grant in any one year of<br />
option amount to 5% or more of options granted during the year. i.) Mr. Sameer Paigankar<br />
ii.) Dr. B. Gopalan<br />
iii.) Dr. A. Lakdawala<br />
iv.) Mr. A.S. Mohanty<br />
v.) Mr. Rajesh V. Desai<br />
vi.) Mr. Dereck Lobo<br />
vii.) Mr. J. A. Mograwala<br />
viii.)Dr. Shekhar Bhaskar Bhirud<br />
ix.) Dr. Subramaniyam Duvuuri<br />
x.) Mr. Arloph John Vieira<br />
iii) Identified employees who were granted option during any<br />
NIL<br />
one year, equal to or exceeding 1% of the issued capital<br />
(excluding outstanding warrants and conversions) of the<br />
Company at the time of grant<br />
k) Diluted Earnings Per Share (EPS) pursuant to issue of shares on<br />
exercise of option calculated in accordance with International<br />
Accounting Standard (IAS) 33. Rs. 7.37/-<br />
* Figures have been recast pursuant to sub-division of face value of equity share from Rs. 10 each to Rs. 2 each.<br />
58
B . <strong>Glenmark</strong> Employee Stock Option Scheme ‘2003<br />
a) Options granted 700,725<br />
b) The Pricing Formula Market value as on October 22nd, 2003<br />
c) Options vested NIL<br />
d) Option exercised NIL<br />
e) The total number of shares arising as a result of exercise of option. NIL<br />
f) Options lapsed NIL<br />
g) Variation of terms of options NIL<br />
h) Money realised by exercise of option NIL<br />
i) Total number of options in force as on 31st March’2004 700,725<br />
j) Employee wise details of options granted to:<br />
i) Senior Managerial personnel i) Mr. R. V. Desai<br />
ii) Mr. A.S. Mohanty<br />
iii) Mr. Sameer Paigankar<br />
iv) Dr. B. Gopalan<br />
v) Dr. Shekhar Bhaskar Bhirud<br />
vi) Dr. Anish Desai<br />
vii) Dr. G. N. Choudhary<br />
viii) Dr. Swaroop Kumar<br />
ix) Dr. Krishna K. Solanki<br />
x) Mr. M.J. Mendonza<br />
xi) Mr. V. S. Mani<br />
xii) Mr. Alind Sharma<br />
xiii) Mr. V. Dhamankar<br />
xiv) Mr. J. A. Mograwala<br />
xv) Mr. T. R. Grover<br />
xvi) Mr. A. C. Sukhtankar<br />
xvii) Mr. K. R. Menon<br />
xviii) Mr. Sanjay Mahajan<br />
xix) Mr. M. Anil<br />
xx) Mr. Pritam Roy<br />
xxi) Mr. K. Anand<br />
ii) Any other employee who receives a grant in any one year of NIL<br />
option amount to 5% or more of options granted during the year.<br />
iii) Identified employees who were granted option during any NIL<br />
one year, equal to or exceeding 1% of the issued capital<br />
(excluding outstanding warrants and conversions) of the<br />
Company at the time of grant<br />
k) Diluted Earnings Per Share (EPS) pursuant to issue of shares Rs. 7.37/-<br />
on exercise of option calculated in accordance with International<br />
Accounting Standard (IAS) 33.<br />
l) Method of Calculation of employees compensation cost The company has calculated the employee compensation cost using the intrinsic<br />
value of the stock options.<br />
Difference between the employee compensation cost so<br />
computed at<br />
i) above and the employee compensation cost that shall have been<br />
recognised if it had used the fair value of the options<br />
NIL<br />
The impact at this difference on profits and on EPS of the Company<br />
(In Thousand)<br />
Profit after Tax (PAT) Rs. 408196<br />
Less: Additional employee Compensation cost based on fair value NIL<br />
Adjusted PAT Rs. 408196<br />
Adjusted EPS : Basic Rs. 7.41<br />
Diluted Rs. 7.37<br />
m) Weighted-average exercise price and fair value of<br />
Stock Options granted:<br />
Stock Options Weighted average Weighted average Fair value (in Rs.) Closing market price at NSE<br />
granted on exercise price (in Rs.) * Face Value Rs 10 on the date of grant (in Rs.)<br />
22/10/2003 *376.50 * 144.27 *357.35<br />
n) Description of the method and significant assumptions used The Black Scholes option pricing model was developed for estimating fair value of<br />
during the year to estimate the fair value of the options,<br />
traded options. Since Option pricing model requires use of substantive assumptions,<br />
including the following weighted average information:<br />
changes therein can materially affect Fair Value of options. The option pricing<br />
models do not necessarily provide a reliable measure of fair value of options.<br />
The main assumptions used in the Black-Scholes option pricing model during the year were as follows:<br />
Risk free interest rate 5%<br />
Expected life of options from the date of grant<br />
6 years<br />
Expected Volatility 33.83%<br />
Dividend yield 1.9%<br />
59
Pursuant to amendment in the SEBI (Employees Stock<br />
Option Scheme and Employees Stock Purchase<br />
Scheme), Guidelines, 1999, SEBI vide their letter<br />
No.CDF/DIL/UR/ 23141/2003 dated 8th December, 2003<br />
has advised the Company to make the disclosures<br />
under the said guidelines, as may be applicable to the<br />
Company, for the information of the members. The<br />
following disclosures are made in compliance with the<br />
above requirement.<br />
Total number of options to be granted<br />
Total number of options granted under this scheme<br />
8,75,000 Options convertible into 8,75,000 Equity Shares<br />
of Rs. 2/- each.<br />
Identification of classes of employees entities to participate<br />
under the scheme of the Company<br />
The following categories of employees are entitled to<br />
participate/ be issued options under the scheme:<br />
Permanent employees : √<br />
Permanent employees outside India : √<br />
Permanent employees of subsidiary : –<br />
Permanent employees of holding : √<br />
Whole –time directors : √<br />
Independent directors : –<br />
The Compensation Committee consisting of directors<br />
reviews the performance and identifies the employees<br />
from the above said categories for grant of options. The<br />
number of options is also decided based on the<br />
performance appraisal system of the Company<br />
Employees and directors who are either promoters or<br />
belong to the promoter group as defined in SEBI<br />
Guidelines and any person holding 2% of the outstanding<br />
share capital of the Company’s equity shares at any time<br />
after commencement of this Plan shall not be eligible<br />
under this Plan.<br />
Requirements of vesting, period of vesting and maximum<br />
period within which the options shall be vested<br />
The minimum period of vesting is one year from the date<br />
of grant and may extend upto a maximum period of three<br />
years from the date of the grant, with such lock in period<br />
as may be decided by the Board/Compensation<br />
Committee. The vesting may also happen in one or more<br />
trenches as decided by the Compensation Committee.<br />
Exercise price or Pricing formula<br />
Exercise Price shall be the Book Value as per the last<br />
audited Balance Sheet or as may be decided by the<br />
Compensation Committee.<br />
The exercise price shall be subject to adjustment at the<br />
discretion of the Compensation Committee, in the event of<br />
the Company distributing Bonus Shares/Rights Shares, so<br />
that the holder of any Warrant, who has not opted for<br />
conversion before the record date for the issue of the said<br />
Bonus Shares/Rights Shares shall be entitled to receive the<br />
number of Shares of the Company which he would have<br />
held or have been entitled to receive on such Warrant<br />
being exercised immediately prior to such record date.<br />
Exercise period and process of exercise<br />
The exercise period commences from the date of vesting<br />
of options and the options may at any time till the<br />
employee continues in the services of the Company, or his<br />
leaving the services of the Company due to termination or<br />
resignation whichever is earlier.<br />
In order to exercise the options, employee would give an<br />
exercise notice as per the draft provided in the Scheme to<br />
the designated officer with the full consideration towards<br />
the options to be exercised.<br />
Appraisal process for determining the eligibility of<br />
employees to the Employees Stock Options Scheme<br />
The options are allotted to the Employees based on their<br />
performance appraisal, period of service, the present and<br />
potential contribution of the employee to the growth of the<br />
Company.<br />
The Compensation Committee of directors reviews the<br />
performance appraisal system of the Company and<br />
identifies the employees for grant of options.<br />
Maximum number of options to be issued per<br />
employee and in aggregate.<br />
Company can issue maximum 8,75,000 options under the<br />
scheme & a maximum no. of 2,50,000 options can be<br />
granted per eligible employee.<br />
Accounting methods<br />
The Company shall confirm to the accounting policies<br />
specified in Clause 13.1 of the SEBI (Employees Stock<br />
Option Scheme and Employees Stock Purchase Scheme)<br />
Guidelines, 1999.<br />
60
Report on<br />
Corporate Governance<br />
Pursuant to Clause 49 of the Listing Agreement, a Report<br />
on Corporate Governance is given below.<br />
1. The Company’s philosophy on Code of Governance is<br />
aimed at assisting the top management of the Company in<br />
the efficient conduct of its business and in meeting its<br />
obligations to shareholders. The Company has adopted a<br />
codified Corporate Governance Charter, inter alia, to fulfil<br />
its corporate responsibilities and achieve its financial<br />
objectives.<br />
The Company believes in and has consistently practiced<br />
good corporate governance. The Company creates an<br />
environment for the efficient conduct of the business and<br />
to enable management to meet its obligations to all its<br />
stakeholders, including amongst others, shareholders,<br />
customers, employees and the community in which the<br />
Company operates.<br />
Given below is a Report on Corporate Governance of your<br />
Company.<br />
2. BOARD OF DIRECTORS:<br />
A. Composition:<br />
The Board comprises of eleven Directors, of whom, five<br />
are executive, and six are non-executive Directors. The<br />
Chairman of the Board is a non-executive Director.<br />
The non-executive Directors are professionals with<br />
experience in management, pharma industry, legal, finance,<br />
marketing and general administration who bring in a wide<br />
range of skills and experience to the Board.<br />
a) Details of the Board of Directors<br />
Name of the Director Status No. of Board No. of other No. of Committee<br />
Meetings attended Directorship held # Membership (s)<br />
Chairman Member<br />
Gracias Saldanha Non-Executive -<br />
- Chairman Promoter Group 4 5 – –<br />
B .E. Saldanha Executive -<br />
Promoter Group 4 5 – –<br />
Glenn Saldanha Executive -<br />
Managing Director & CEO Promoter Group 7 2 – 2<br />
Cheryl Pinto Executive -<br />
Promoter Group 6 – – 1<br />
J. F. Ribeiro Non-Executive<br />
- Independent 5 2 3 –<br />
R.V .Desai Executive 7 – – 1<br />
A. S. Mohanty Executive 7 – – –<br />
61
Name of the Director Status No. of Board No. of other No. of Committee<br />
Meetings attended Directorship held # Membership (s)<br />
Chairman Member<br />
Prasanna Gore Non-Executive -<br />
Independent – – – 1<br />
N.B Desai Non-Executive -<br />
Independent 5 – – 2<br />
M. Gopal Krishnan Non-Executive -<br />
Independent 2 – – 1<br />
J. M. Trivedi Non-Executive -<br />
Independent 5 3 – –<br />
Sridhar Gorthi<br />
(Appointed as Alternate<br />
Director to Dr. Prasanna Non-Executive -<br />
Gore on May 12, 2003). Independent 6 1 – 1<br />
S. A. Paigankar<br />
(Resigned on March 29, 2004). Executive 2 – – –<br />
B. S. Mehta Non-Executive -<br />
(Resigned on July 31, 2003) Independent – – – 2<br />
Rohit S. Mehta<br />
(Served as Alternate Director<br />
to B. S. Mehta from May Non-Executive -<br />
12, 2003 to July 31, 2003.) Independent 3 – – 2<br />
# Includes Directorship(s) in Private Companies.<br />
b) During the Financial Year ended March 31, 2004 seven<br />
board meetings were held on the following dates:<br />
May 15, 2003; May 16, 2003; July 31, 2003; September<br />
29, 2003; October 24, 2003; January 31, 2004; March 29,<br />
2004.<br />
B. None of the non-executive Directors of the Company,<br />
have any pecuniary relationship or transactions with the<br />
Company other than sitting fees paid for attending board<br />
meeting/committee meetings and those already disclosed<br />
in the note four of schedule 21 to the Financial Statement<br />
in the Annual Report.<br />
C. Mr. Gracias Saldanha, Mrs. B .E. Saldanha, Mr. Glenn<br />
Saldanha, Mr. R.V. Desai, Mr. A. S. Mohanty, Mr. N.B<br />
Desai & Mr. S. A. Paigankar attended the last Annual<br />
General Meeting of the Company held on September 26,<br />
2003.<br />
3. AUDIT COMMITTEE:<br />
Your Company has a qualified and independent Audit<br />
Committee. During the Financial Year ended March 31,<br />
2004 the Committee was re-constituted on July 31, 2003.<br />
The committee met four times on May 15, 2003; July 31,<br />
2003; October 22, 2003 and January 31, 2004.<br />
62
Name No. of meetings Remarks<br />
attended<br />
1. J. F. Ribeiro 3 Chairman of the Committee.<br />
2. Prasanna Gore – Appointed as member of the Committee on July 31, 2003.<br />
3. Sridhar Gorthi 4 Alternate Director to Dr. Prasanna Gore.<br />
4. N. B. Desai 1 Appointed as a member of the Committee on July 31, 2003<br />
5. M. Gopal Krishnan – Appointed as a member of the Committee on July 31, 2003<br />
6. B. S. Mehta – Resigned from the Board of Directors and the Committee on July 31, 2003.<br />
7. Rohit S. Mehta 2 Served the Committee in the capacity of Alternate Director to Mr. B. S.<br />
Mehta, from May 12, 2003 to July 31, 2003.<br />
Mr. Glenn Saldanha, Managing Director & CEO, Mr. R. V. Desai, Director-Finance, IT & Legal and Mr. Prakash Sevekari,<br />
Cost Auditor are invitees to the Meeting. Mr. M. J. Mendonza, Vice President-Legal and Company Secretary is the<br />
Secretary to the Committee. The terms of reference of this committee are wide enough covering matters specified in the<br />
Companies Act, 1956 read together with Clause 49 of the Listing Agreement of the Stock Exchange. The current Charter<br />
of the Audit Committee is in line with international best practices and the regulatory changes formulated by SEBI and the<br />
listing agreements with the stock exchanges on which your Company is listed.<br />
4. REMUNERATION OF DIRECTORS:<br />
A. The remuneration of the executive and non-executive Directors of your Company is decided by the Board of Directors.<br />
B. Given below are the details of remuneration / fees / commission paid to Directors during the financial year ended March<br />
31, 2004:<br />
Amount in Rs.<br />
Name of Director Salary Commission Perquisites Retirement Sitting Total<br />
benefits /Other Fees<br />
reimbursements<br />
1 Gracias Saldanha 10,084,182 – – 20,000 10,104,182.41<br />
2 B. E. Saldanha 3,960,000 990,000 173,590 2,053,500 – 7,177,089.61<br />
3 Glenn Saldanha 3,180,000 9,887,165 303,743 1,188,602 – 14,559,509.96<br />
4 Cheryl Pinto 1,755,000 5,042,123 224,930 388,869 – 7,410,921.22<br />
5 R. V. Desai 2,308,430 275,016 233,795 306,820 – 3,124,061.71<br />
6 A. S. Mohanty 2,333,430 275,016 264,852 313,244 – 3,186,542.30<br />
7 S. A. Paigankar 2,205,130 252,098 369,678 279,772 – 3,106,677.34<br />
8 J. M. Trivedi – – – – 25,000 25,000.00<br />
9 J. F. Ribeiro – – – – 40,000 40,000.00<br />
10 M. Gopal Krishnan – – – – 10,000 10,000.00<br />
11 N. B. Desai – – – – 30,000 30,000.00<br />
12 Rohit S. Mehta – – – – 25,000 25,000.00<br />
13 Sridhar Gorthi – – – – 50,000 50,000.00<br />
15,741,990 26,805,600 1,570,588 4,530,806 200,000 48,848,984.55<br />
63
Notes:<br />
1 The Executive Directors have been appointed/ re-appointed on May 16, 2002 for the term of five years. The service<br />
contract can be terminated with a notice of six months.<br />
2. Sitting fees in respect of Mr. J. M. Trivedi have been paid to CDC Advisors Pvt. Ltd., of which he is a nominee.<br />
3. During the Year, the following Directors acquired Shares of Company under ESOS-99:-<br />
i) Mr. S. A. Paigankar* 15,000 Equity Shares of Rs. 2/- each.<br />
ii) Mr. A. S. Mohanty<br />
20,000 Equity Shares of Rs. 2/- each.<br />
iii) Mr. R. V. Desai<br />
20,000.Equity Shares of Rs. 2/- each.<br />
* Resigned on March 29, 2004.<br />
The options had a vesting period of three years and were issued at a premium of Rs. 12.34 per equity share.<br />
5. SHAREHOLDERS COMMITTEES:<br />
Share Transfer and Shareholders’ / Investors’ Grievances Committee:<br />
The Committee comprising of the following members who review shareholders’ complaints and resolution thereof.<br />
1. Mr. J. F. Ribeiro – Chairman<br />
2. Mr. Glenn Saldanha – Member<br />
3. Mr. R.V. Desai – Member<br />
The Committee once met on 23rd June, 2003, prior to being merged with the Share Transfer Committee and reconstitution<br />
on July 31, 2003.<br />
Compliance Officer: Mr. M.J. Mendonza-Vice President-Legal & Company Secretary is the Compliance Officer of the<br />
Company.<br />
Name of committee Members No. of Attendance at the meeting<br />
meetings held<br />
Shareholders’ and Investors’ 1) J. F. Ribeiro 1 All Directors were present in the meeting.<br />
Grievance Committee. 2) Glenn Saldanha<br />
3) R. V. Desai<br />
Share Transfer Committee. 1) Glenn Saldanha 10 Mr. R.V. Desai attended all meetings, Mr. Glenn<br />
2) Cheryl Pinto Saldanha and Mrs. Cheryl Pinto attended seven<br />
3) R. V. Desai meetings.<br />
Share Transfer and 1. J. F. Ribeiro 22 Mr. R.V. Desai attended all meetings,<br />
Shareholders’ / Investors’ 2. Glenn Saldanha Mr. J. F. Ribeiro & Mr. Glenn Saldanha attended<br />
Grievance Committee. 3. N. B. Desai 20 meetings each & Mr. N. B. Desai attended<br />
4. R.V. Desai 19 meetings.<br />
• Details of investor’s complaints received during the year ended March 31, 2004:<br />
No. of complaints 2003-2004 2002-2003<br />
Received 172 219<br />
Disposed 172 219<br />
Pending Nil Nil<br />
• The Company's Registrars, Karvy Computershare Private Ltd, had received letters / complaints during the financial year,<br />
all of which were replied / resolved to the satisfaction of the shareholders.<br />
6. COMPENSATION COMMITTEE:<br />
i) The Compensation Committee comprises of following members of the Board:<br />
64
1. Mr. J. F. Ribeiro – Chairman<br />
2. Mr. Glenn Saldanha – Member<br />
3. Mr. N. B. Desai – Member<br />
ii) The Committee was reconstituted on September 29, 2003.<br />
iii) During the year ended March 31, 2004, one meeting of<br />
Compensation Committee was held on 22nd October,<br />
2003 and where except Mr. N. B. Desai all other Directors<br />
were present.<br />
iv) Broad terms of reference of the Compensation<br />
Committee:<br />
• To recommend and review remuneration package of<br />
Executive / Non-Executive Directors.<br />
• To present report to the Board on remuneration package<br />
of Directors and others.<br />
v) Compensation Policy :<br />
The Company follows a market linked remuneration policy,<br />
which is aimed at enabling the Company to attract and<br />
retain the best talent. Compensation is also linked to<br />
individual and team performance as they support the<br />
achievement of Corporate Goals. The Company has<br />
formulated an Employee Stock Option Scheme for<br />
rewarding & retaining performers.<br />
7. DISCLOSURES BY MANAGEMENT :<br />
a) No material, financial and commercial transactions were<br />
reported by the management to the Board, in which the<br />
management had personal interest having a potential<br />
conflict with the interest of the Company at large.<br />
b) There are no transactions with the Director or<br />
Management, their subsidiaries or their relatives etc. that<br />
may have potential conflict with the interest of the<br />
Company at large.<br />
c) There was no non-compliance during the last three years<br />
by the Company on any matter related to capital market.<br />
Consequently, there were no penalties imposed nor<br />
strictures passed on the Company by Stock Exchanges,<br />
SEBI or any statutory authority.<br />
8. SHAREHOLDERS INFORMATION:<br />
a) The relevant information relating to the Directors<br />
appointed after last Annual General Meeting of the<br />
Company and to be re-appointed at the ensuing Annual<br />
General Meeting to be held on September 24, 2004 are<br />
given below:<br />
i. Glenn Saldanha:- 34, is a B.Pharm from Bombay<br />
University and was awarded Watumall Foundation award<br />
for overall excellence. His other educational qualifications<br />
includes an MBA from New York University‘s Leonard N.<br />
Stern School of Business (USA). He has worked for Eli Lilly<br />
in the USA and was a management consultant with<br />
PriceWaterhouse Coopers. His services have been used<br />
by SmithKline Beecham, Rhone Poulenc Rhorer, Astra,<br />
Merck and Johnson & Johnson, among others.<br />
He is also a Director of following Companies/ Body<br />
Corporates:-<br />
S.No. Name of Company Position<br />
1 G M Pharma Ltd. Chairman<br />
2 <strong>Glenmark</strong> Exports limited Director<br />
3 <strong>Glenmark</strong> Pharmaceuticals Inc.USA Chairman<br />
4 <strong>Glenmark</strong> Pharmaceuticals (U.K) Ltd. Chairman<br />
5 <strong>Glenmark</strong> Farmaceutica Ltda. Chairman<br />
6 <strong>Glenmark</strong> Philippines Inc. Chairman<br />
7 <strong>Glenmark</strong> Pharmaceuticals (Nigeria) Ltd Chairman<br />
8 <strong>Glenmark</strong> Chemicals Inc., USA Director<br />
Mr. Glenn Saldanha is also member of Compensation and<br />
Share Transfer & Shareholders/Investors Grievance<br />
Committee of your Company.<br />
ii. Cheryl Pinto: - 36, is a graduate in Pharmacy from<br />
University of Bombay .She has over 10 years experience in<br />
pharmaceutical business.<br />
She is also Director of following Companies/Body<br />
Corporates:-<br />
S.No. Name of Company Position<br />
1 <strong>Glenmark</strong> Pharmaceuticals Inc. USA Director<br />
2 <strong>Glenmark</strong> Pharmaceuticals (U.K) Ltd. Director<br />
iii. J. F. Riberio: 73 is, a retired government official and has<br />
served the country under various assignments .Amongst<br />
the major positions held, he has been Ex-Commissioner of<br />
Police, Mumbai, Former Special Secretary to Government<br />
of India, Ministry of Home Affairs, Former Director General<br />
of Police, Punjab, Ex-adviser to the Governor of Punjab, Ex-<br />
Ambassador of India to Romania. He is also a Director of<br />
following Companies:-<br />
S. No. Name of company Position Held<br />
1 IIT Corporate Services Ltd. Director<br />
2 V. V. F Limited Director<br />
Mr. Ribeiro is Chairman of all three committees in your<br />
Company and also the Chairman of Audit Committee of V.<br />
V. F Ltd.<br />
65
iv. Steven Bates is a CFA from UK with over 20 years experience in the financial industry. He has worked with top firms<br />
viz. James Capel, JP Morgan Fleming Asset Management, as head of Global emergence market. Mr. Bates is at present<br />
Director, Zephyr Management UK Ltd, an FSA regulated investment business investing in emerging markets. He is also an<br />
Investment Advisor to Cardiff & Vale of Glamorgan Pension Fund, Member of the Governing Board of Kosovo Pensions<br />
Savings Trust (UN appointment) and Consultant on pension reform to Russian Foundation for Social Reform (World Bank).<br />
Mr. Bates is also a Director of following Companies/Body Corporates:-<br />
S. No. Name of company Position Held<br />
1 Zephyr Management UK Ltd. Director<br />
2 Baring Emerging Europe plc Non Executive Director<br />
3 Magna Umbrella Fund plc Non Executive Director<br />
4 Novy Neft Non Executive Director<br />
5 Occo Eastern European Fund Non Executive Director<br />
b) Share Transfer Process: The shares are sent<br />
/received for physical transfer at R& T’s office<br />
and all valid transfer requests are processed and<br />
returned within a period of 30 days from the date<br />
of receipt. The Share transfers are approved on<br />
weekly basis by the Share Transfer and<br />
Shareholders’ /Investors’ Grievance Committee.<br />
c) Dematerialisation of shares: As of March 31, 2004, 96.33<br />
% of shares have been dematerialised and held in<br />
electronic form through NSDL and CDSL. The shares of<br />
your company are permitted to be traded only in<br />
dematerialised form.<br />
d) Share Holding Pattern as at March 31, 2004:<br />
Promoter’s<br />
Resident Individuals<br />
Foreign Institutional<br />
Investor<br />
Bodies corporates<br />
Indian financial<br />
institutions<br />
Others<br />
20.41<br />
18.26<br />
2.42<br />
54.57<br />
2.11 2.23<br />
Distribution of Share Holding as on 31/03/04<br />
Pie chart of Share Holding<br />
Sl. No. Description No.of Share holders Shares Per cent<br />
1 Promoter`s 22 32333088 54.57<br />
2 Resident Individuals 22657 12092835 20.41<br />
3 Foreign Institutional Investor 11 10815935 18.26<br />
4 Bodies corporates 928 1430727 2.42<br />
5 Indian financial institutions 4 1250655 2.11<br />
6 Others:<br />
i Non resident Indians/OCBs 220 564628 0.95<br />
ii Trusts 8 4700 0.01<br />
iii Mutual funds 4 309045 0.52<br />
iv Employees 446 181000 0.31<br />
v Clearing members 2 108527 0.18<br />
vi H. U. F. 207 104580 0.18<br />
vii Directors & relatives 8 49850 0.08<br />
895 1322330 2.23<br />
Total 24517 59245570 100.00<br />
e) General Body Meetings<br />
The last three Annual General Meetings of the Company were held at the venue and time as under:-<br />
66
AGM No. Date Time Venue<br />
23 September 28, 2001 11.00 a.m. Sunville Banquet & Conference Hall<br />
3rd floor, Dr. Annie Besant Road,<br />
Worli, Mumbai-400 018.<br />
24 September 27, 2002 11.00 a.m Sunville Banquet & Conference Hall<br />
3rd floor, Dr. Annie Besant Road,<br />
Worli, Mumbai-400 018.<br />
25 September 26,2003 11.00 a.m Sunville Banquet & Conference Hall<br />
3rd floor, Dr. Annie Besant Road,<br />
Worli, Mumbai-400 018.<br />
f) Date Time and Venue of the Ensuing Annual General Meeting:<br />
Annual General Meeting shall be held on September 24, 2004 at 11 a.m. at Sunville Banquet Hall, Annie Besant Road,<br />
Worli, Mumbai 400 018.<br />
g) Record Date/Book Closure:<br />
• April 3, 2004, was fixed as record date for making<br />
payment of Interim dividend on Equity share capital of<br />
the Company.<br />
• September 17, 2004 to September 24, 2004 (both days<br />
inclusive), for the purpose of ensuing Annual General<br />
Meeting.<br />
h) Date of declaration of interim dividend:<br />
Equity Shares - March 29 , 2004<br />
Preference Shares - March 29, 2004<br />
i) Financial Calendar (Tentative and Subject to change)<br />
Financial reporting for the first quarter ending July 2004<br />
June 30, 2004.<br />
Financial reporting for the second quarter ending October 2004<br />
September 30, 2004.<br />
Financial reporting for the third quarter ending January 2005<br />
December 31, 2004.<br />
Financial results for the year ending May 2005<br />
March 31, 2005.<br />
j) Members can avail of nomination facility by filing Form<br />
2B with the Company. Blank forms can be downloaded<br />
from the website of the Company.<br />
k) Members may kindly note that consequent to split in the<br />
face value of equity shares of the Company, the shares in<br />
the face value of Rs.10/- shall cease to be valid for any<br />
purpose whatsoever. Members who are holding shares of<br />
the face value of Rs.10/- each are requested to kindly send<br />
their respective share certificates to the R&T Agents for<br />
receiving five equity shares of face value of Rs. 2/- each in<br />
exchange of one equity share of face value of Rs. 10/- each.<br />
l) Pursuant to the provisions of Section 205A (5) of the<br />
Companies Act, 1956, dividend for the financial year<br />
ended March 31, 2000 and thereafter, which remain<br />
unclaimed for a period of seven years will be transferred by<br />
the Company to the Investor Education and Protection<br />
Fund (IEPF) established by the Central Government<br />
pursuant to Section 205C of the Companies Act, 1956.<br />
Information in respect of such unclaimed dividend when<br />
due for transfer to the said Fund is given below:<br />
Financial Date of declaration Date of transfer to unpaid/ Last date for claiming Due date for<br />
Year Ended of Dividend unclaimed dividend account unpaid Dividend transfer to IEP Fund<br />
31.03.2000 10.03.2000 22.04.2000 21.04.2007 22.04.2007<br />
31.03.2001 28.09.2001 27.10.2001 27.10.2008 28.10.2008<br />
31.03.2002 27.09.2002 27.10.2002 27.10.2009 28.10.2009<br />
31.03.2003 15.05.2003 15.06.2003 15.06.2010 16.06.2010<br />
Shareholders who have not so far encashed their dividend warrant(s) are requested to seek issue of duplicate warrant(s) by<br />
67
writing to the Company’s Registrar and Transfer Agents,<br />
M/s. Karvy Computershare Pvt. Limited immediately.<br />
Shareholders are requested to note that no claims shall lie<br />
against the Company or the said Fund in respect of any<br />
amounts which remain unclaimed and unpaid for a period<br />
of seven years from the dates that they first became due<br />
for payment and no payment shall be made in respect of<br />
any such claims.<br />
m) Means of Communication:<br />
a. Quarterly/Half Yearly and Annual Financial Results of the<br />
Company are published in the Financial Express and<br />
Punyanagri newspapers.<br />
b. Your Company’s results & official news releases are<br />
displayed on the Company’s website. There were no<br />
presentations to any Institutional Investors / Analysts<br />
during the year.<br />
(All figures in Indian Rupees)<br />
Month High Low Close BSE Sensex<br />
Apr 03 230.00 206.00 230.00 2959.79<br />
May 03 257.00 216.50 253.80 3180.75<br />
Jun 03 306.35 230.00 297.85 3607.13<br />
Jul 03 340.00 275.10 310.95 3792.61<br />
Aug 03 371.00 293.00 366.30 4244.73<br />
Sep 03 442.00 348.80 391.50 4453.24<br />
Oct 03 409.00 369.25 433.75 4906.87<br />
Nov 03 435.00 380.50 416.25 5044.82<br />
Dec 03 773.50 420.00 714.75 5838.96<br />
Jan 04 987.00 670.50 753.75 5695.67<br />
Feb 04 838.50 681.25 750.75 5667.51<br />
Mar 04 795.50 635.00 721.00 5590.60<br />
* For maintaining parity in comparison, the stock market prices of<br />
the Company’s shares have been taken for Rs. 10 (face value) per<br />
share.<br />
c. All items required to be covered in the Management<br />
800<br />
6000<br />
Discussion & Analysis are included in the Directors' Report<br />
to Members.<br />
686<br />
5392<br />
d. Company has its own web site and all the vital<br />
information relating to the company and its products is<br />
displayed on its web site: www.glenmarkpharma.com.<br />
Share price<br />
572<br />
458<br />
4784<br />
4176<br />
BSE Sensex<br />
Your Company also regularly provides information to the<br />
344<br />
3568<br />
stock exchanges as per the requirements of the Listing<br />
Agreements. The Company’s website is updated<br />
periodically to include information on new developments<br />
and business opportunities of your Company.<br />
230<br />
Apr 03<br />
May 03<br />
Jun 03<br />
Sep 03<br />
Aug 03<br />
Jul 03<br />
<strong>Glenmark</strong><br />
Oct 03<br />
Jan 04<br />
Dec 03<br />
Nov 03<br />
BSE Sensex<br />
Feb 04<br />
Mar 04<br />
2960<br />
9. Company’s Script Information:<br />
• Listing on stock exchanges: The shares of the Company<br />
are listed on The Stock Exchange, Mumbai & The National<br />
Stock Exchange of India Ltd.<br />
• Listing fees for the year 2004-05 have been paid to the<br />
Stock Exchanges.<br />
• Stock Code: 532296 on the BSE<br />
- Electronic Form No.INE935A01027<br />
- Scrip Name<br />
- GLENMARK PHA- BSE<br />
- GLENMARK - NSE<br />
Market Price Data:<br />
High, low during each month in last financial year.<br />
Performance in comparison to broad based indices namely<br />
BSE Sensex.<br />
10. Plant Locations:<br />
The Company's plants are located at:<br />
i) E-37, MIDC Industrial Area, D Road, Satpur, Nasik-<br />
422007, Maharashtra<br />
ii) 3109-C, GIDC Industrial Estate, Ankleshwar – 393 002<br />
Dist. Bharuch.<br />
iii) Plot no. 7, Colvale Industrial Estate, Bardez, Goa.<br />
iv) Plot no. A- 80, MIDC Area, Kurkumbh, Daund, Pune,<br />
Maharashtra.<br />
v) Plot No. 163- 165 & 170 – 172, Chandramouli Industrial<br />
Estate, Mohol, Sholapur, Maharashtra – 413213.<br />
11. Outstanding GDR's/ADR's/Warrants or any<br />
Convertible instruments exercised, date and likely<br />
impact on equity :<br />
During the year, your Company formulated a new<br />
Employees Stock Option Scheme viz. ESOS’ 2003,<br />
68
pursuant to which 7,00,725 options have been issued.<br />
Pursuant to ESOS’1999, your Company had issued 875,000<br />
Convertible Warrants to <strong>Glenmark</strong> Pharmaceuticals Limited<br />
Employees Welfare Trust. During the Financial Year 2003-<br />
2004, 2,00,000 options were exercised and 35,000 options<br />
were cancelled.<br />
As of March 31, 2004, 7,00,725 and 85,000 options were<br />
outstanding under ESOS’ 2003 and ESOS’ 1999<br />
respectively and are due for exercise on the following<br />
dates:<br />
ESOS’ 1999<br />
Date<br />
Number of Options<br />
01.04.2004 15,000<br />
01.07.2004 5,000<br />
01.01.2005 55,000<br />
01.07.2005 5,000<br />
01.01.2006 5,000<br />
ESOS’ 2003<br />
Date<br />
Number of Options<br />
22.10.2005 70,073<br />
22.10.2006 1,40,145<br />
22.10.2007 2,10,218<br />
22.10.2008 2,80,289<br />
On exercising the convertible options, the paid-up equity<br />
share capital of the Company will increase by a like number<br />
of shares.<br />
12. Electronic Clearing System (ECS):<br />
Shareholders are advised to opt for payment of dividend<br />
through ECS<br />
The salient benefits of receiving dividend payment through<br />
ECS amongst others may be listed as below:<br />
a) There are no clearing charges in the hands of the<br />
investor/ recipient, the same are borne by the Company;<br />
b) Risk as to fraudulent encashment of the dividend<br />
warrants, loss / interception of dividend warrants in transit,<br />
are eliminated;<br />
c) The facility ensures instant credit of the dividend amount<br />
in the desired account which to the recipient, means<br />
effortless and speedier transaction and hassles as to<br />
revalidation etc are done away with;<br />
d) Once the payment is made through ECS, Company<br />
issues intimation letters to the investors as to credit /<br />
payment of dividend, providing therein the details of<br />
the account and amount. Investors may download<br />
the ECS Mandate Form from the Company’s website<br />
and send the same duly filed-in to registrars for updation of<br />
records.<br />
13. Investor Helpdesk: for clarifications / assistance, if<br />
any, please contact:-<br />
Registered Office<br />
Registrars & Transfer<br />
Agents<br />
Persons Abhishek Thareja M. S. Madhusudhan<br />
to contact<br />
Add: B/2, Mahalaxmi Chambers "Karvy House",46,Avenue 4,<br />
22, Bhulabhai Desai Road, Street No.1, Banjara Hills,<br />
Mumbai-400 026<br />
Hyderabad-500034.<br />
Telephone (022) 23514893-6 (040) 23312454/23320751<br />
Fax No. (022) 23512531 (040) 23311968<br />
Email abhishekt@glenmarkpharma.com madhusudhan@karvy.com<br />
Website: www.glenmarkpharma.com www.karvy.com<br />
69
Certificate on<br />
Corporate Governance<br />
To the Members of<br />
<strong>Glenmark</strong> Pharmaceuticals Limited<br />
We have examined the compliance of conditions of Corporate Governance by <strong>Glenmark</strong> Pharmaceuticals Ltd., for the year<br />
ended on March 31, 2004, as stipulated in Clause 49 of the Listing Agreement of the said Company with the stock<br />
exchanges in India.<br />
The compliance of conditions of Corporate Governance is the responsibility of the management. Our examination has been<br />
limited to review of procedures and implementation thereof adopted by the Company for ensuring the compliances of the<br />
conditions of Corporate Governance as stipulated in the said Clause. It is neither an audit nor an expression of opinion on<br />
the financial statements of the Company.<br />
In our opinion and to the best of our information and according to the explanations given to us, the Company has complied<br />
with the conditions of Corporate Governance stipulated in Clause 49 of the above mentioned Listing Agreements.<br />
We state that in respect of investor grievances received during the year ended March 31, 2004, no investor grievances are<br />
pending against the Company as per the records maintained by the Registrars and Share Transfer Agents of the Company<br />
and as presented to the Share Transfer and Investor Grievance Committee.<br />
We further state that such compliance is neither an assurance as to the future viability of the Company nor of the<br />
efficiency or effectiveness with which the management has conducted the affairs of the Company.<br />
For and on behalf of Price Waterhouse<br />
Mumbai, July 5, 2004<br />
Partha Ghosh<br />
Partner<br />
Membership Number – F 055913<br />
Chartered Accountants<br />
70
Auditors’ Report<br />
To the members of<br />
GLENMARK PHARMACEUTICALS LIMITED<br />
1. We have audited the attached Balance Sheet of <strong>Glenmark</strong> Pharmaceuticals Limited as at 31st March, 2004, and the<br />
related Profit and Loss Account and Cash Flow Statement for the year ended on that date annexed thereto, which we<br />
have signed under reference to this report. These financial statements are the responsibility of the Company’s<br />
management. Our responsibility is to express an opinion on these financial statements based on our audit.<br />
2. We conducted our audit in accordance with the auditing standards generally accepted in India. Those Standards require<br />
that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of<br />
material mis-statement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures<br />
in the financial statements. An audit also includes assessing the accounting principles used and significant estimates<br />
made by management, as well as evaluating the overall financial statement presentation. We believe that our audit<br />
provides a reasonable basis for our opinion.<br />
3. As required by the Companies (Auditor’s Report) Order, 2003 issued by the Central Government of India in terms of<br />
sub-section (4A) of Section 227 of ‘The Companies Act, 1956’ of India (the ‘Act’) and on the basis of such checks of the<br />
books and records of the Company as we considered appropriate and according to the information and explanations<br />
given to us, we give in the Annexure a statement on the matters specified in paragraphs 4 and 5 of the said Order.<br />
4. Further to the comments in the Annexure referred to in paragraph 3 above, we report that:<br />
(a) We have obtained all the information and explanations, which to the best of our knowledge and belief were<br />
necessary for the purposes of our audit;<br />
(b) In our opinion, proper books of account as required by law have been kept by the Company so far as appears from<br />
our examination of those books;<br />
(c) The Balance Sheet, Profit and Loss Account and Cash Flow Statement dealt with by this report are in agreement<br />
with the books of account;<br />
(d) In our opinion, the Balance Sheet, Profit and Loss Account and Cash Flow Statement dealt with by this report<br />
comply with the accounting standards referred to in sub-section (3C) of Section 211 of the Act;<br />
(e) On the basis of written representations received from the directors, as on 31st March 2004 and taken on record by<br />
the Board of Directors, none of the directors is disqualified as on 31st March 2004 from being appointed as a<br />
director in terms of clause (g) of sub-section (1) of Section 274 of the Act;<br />
(f) In our opinion and to the best of our information and according to the explanations given to us, the said financial<br />
statements together with the notes thereon and attached thereto give in the prescribed manner the information<br />
required by the Act and give a true and fair view in conformity with the accounting principles generally accepted in<br />
India:<br />
(i) in the case of the Balance Sheet, of the state of affairs of the Company as at 31st March 2004;<br />
(ii) in the case of the Profit and Loss Account, of the profit for the year ended on that date; and<br />
(iii) in the case of the Cash Flow Statement, of the cash flows for the year ended on that date.<br />
For and on behalf of Price Waterhouse<br />
Mumbai, April 22, 2004<br />
Partha Ghosh<br />
Partner<br />
Membership Number – F 055913<br />
Chartered Accountants<br />
71
ANNEXURE TO AUDITORS’ REPORT<br />
Referred to in paragraph 3 of the Auditors’ Report of even date to the members of <strong>Glenmark</strong> Pharmaceuticals Limited on<br />
the financial statements for the year ended 31st March 2004<br />
1. (a) The Company is maintaining proper records showing full particulars including quantitative details and situation of<br />
fixed assets.<br />
(b)<br />
(c)<br />
The fixed assets are physically verified by the management according to a phased programme designed to cover<br />
all the items over a period of three years, which in our opinion, is reasonable having regard to the size of the<br />
Company and the nature of its assets. Pursuant to the programme, a portion of the fixed assets has been<br />
physically verified by the management during the year and no material discrepancies between the book records<br />
and the physical inventory have been noticed.<br />
In our opinion and according to the information and explanations given to us, a substantial part of fixed assets has<br />
been disposed by the Company during the year.<br />
2. (a) The inventory (excluding stocks with third parties) has been physically verified by the management during the year.<br />
In respect of inventory lying with third parties, these have substantially been confirmed by them. In our opinion,<br />
the frequency of verification is reasonable.<br />
(b)<br />
(c)<br />
In our opinion, the procedures of physical verification of inventory followed by the management are reasonable<br />
and adequate in relation to the size of the Company and the nature of its business.<br />
On the basis of our examination of the inventory records, in our opinion, the Company is maintaining proper<br />
records of inventory. The discrepancies noticed on physical verification of inventory as compared to book records<br />
were not material.<br />
3. The Company has neither granted nor taken any loans, secured or unsecured, to/from companies, firms or other<br />
parties covered in the register maintained under Section 301 of the Act. As the Company has not granted/taken any<br />
loans, secured or unsecured, to/from companies, firms etc listed in the register maintained under section 301 of the<br />
Act, paragraph 3(b), 3(c) and 3(d) of the order are not applicable.<br />
4. In our opinion and according to the information and explanations given to us, having regard to the explanation that<br />
certain items are purchased are of special nature for which suitable alternative sources do not exist for obtaining<br />
comparative quotations, there are adequate internal control procedures commensurate with the size of the company<br />
and the nature of its business for the purchase of inventory, fixed assets and for the sale of goods. Further, on the<br />
basis of our examination of the books and records of the Company, and according to the information and explanations<br />
given to us, we have neither come across nor have been informed of any continuing failure to correct major<br />
weaknesses in the aforesaid internal control procedures.<br />
5. We are informed by the Management that the transactions with other companies in which Directors of the Company<br />
who are nominated by other companies and are also holding the position as Directors in the other companies (not<br />
holding shares exceeding 2% of the paid up capital) are not required to be entered in the register maintained under<br />
sub-section (1) of Section 301 of the Act.<br />
In view of the above, there are no entries recorded in the Register maintained under Section 301 of the Act.<br />
6. The Company has not accepted any deposits from the public within the meaning of Sections 58A and 58AA of the Act<br />
and the rules framed there under.<br />
7. In our opinion, the Company has an internal audit system commensurate with its size and nature of its business.<br />
8. We have broadly reviewed the books of account maintained by the Company in respect of products where, pursuant<br />
to the Rules made by the Central Government of India, the maintenance of cost records has been prescribed under<br />
clause (d) of sub-section (1) of Section 209 of the Act. We are of the opinion that prima facie, the prescribed accounts<br />
and records have been made and maintained. We have not, however, made a detailed examination of the records<br />
72
with a view to determine whether they are accurate or complete.<br />
9. (a) According to the information and explanations given to us and the records of the Company examined by us, in our<br />
opinion, the Company is generally regular in depositing the undisputed statutory dues including provident fund,<br />
investor education and protection fund, employees’ state insurance, income-tax, sales-tax, wealth tax, customs<br />
duty, excise duty, cess and other material statutory dues as applicable with the appropriate authorities.<br />
(b)<br />
According to the information and explanations given to us and the records of the Company examined by us, there<br />
are no dues of sales tax, income-tax, customs duty, wealth-tax, excise duty and cess which have not been<br />
deposited on account of any dispute.<br />
10. The Company has no accumulated losses as at March 31, 2004 and it has not incurred any cash losses in the financial<br />
year ended on that date or in the immediately preceding financial year.<br />
11. According to the records of the Company examined by us and the information and explanation given to us, the<br />
Company has not defaulted in repayment of dues to any financial institution or bank or debenture holders as at the<br />
balance sheet date.<br />
12. The Company has not granted any loans and advances on the basis of security by way of pledge of shares, debentures<br />
and other securities.<br />
13. The provisions of any special statute applicable to chit fund / nidhi / mutual benefit fund/societies are not applicable to<br />
the Company.<br />
14. In our opinion, the Company is not a dealer to trader in shares, securities, debentures and other investments.<br />
15. In our opinion, and according to the information and explanations given to us, the Company has not given any<br />
guarantee for loans taken by others from banks or financial institutions during the year.<br />
16. In our opinion, and according to the information and explanations given to us, on an overall basis, the term loans have<br />
been applied for the purposes for which they were obtained.<br />
17. On the basis of an overall examination of the balance sheet of the Company, in our opinion and according to the<br />
information and explanations given to us, there are no funds raised on a short-term basis which have been used for<br />
long-term investment. However, funds raised on a long term basis of Rs. 3530 lakhs have been used for short term<br />
purpose.<br />
18. The Company has not made any preferential allotment of shares to parties and companies covered in the register<br />
maintained under Section 301 of the Act during the year.<br />
19. The Company has created securities in respect of debentures issued and outstanding at the year-end.<br />
20. The Company has not raised any money by public issues during the year.<br />
21. During the course of our examination of the books and records of the Company, carried out in accordance with the<br />
generally accepted auditing practices in India, and according to the information and explanations given to us, we have<br />
neither come across any instance of fraud on or by the Company, noticed or reported during the year, nor have we<br />
been informed of such case by the management.<br />
For and on behalf of Price Waterhouse<br />
Mumbai, April 22, 2004<br />
Partha Ghosh<br />
Partner<br />
Membership Number – F 055913<br />
Chartered Accountants<br />
73
Balance Sheet<br />
Rs. In ('000s)<br />
As at 31st March, Schedules 2004 2003<br />
SOURCES OF FUNDS<br />
Shareholders' Funds<br />
a) Share capital 1 218,546 201,811<br />
b) Reserves and surplus 2 2,132,109 1,323,944<br />
2,350,655 1,525,755<br />
Loan Funds<br />
a) Secured loans 3 1,029,429 1,193,564<br />
b) Unsecured loans 4 119,314 53,864<br />
1,148,743 1,247,428<br />
Deferred Tax Liability 5 293,181 269,856<br />
Total 3,792,579 3,043,039<br />
APPLICATION OF FUNDS<br />
Fixed Assets 6<br />
a) Gross Block 1,723,534 1,246,518<br />
b) Less : Depreciation 346,864 243,340<br />
c) Net Block 1,376,670 1,003,178<br />
d) Capital Work-in-progress 244,966 232,471<br />
1,621,636 1,235,649<br />
Investments 7 299,647 161,126<br />
Deferred Tax Assets 8 23,627 15,383<br />
Current Assets, Loans and Advances<br />
a) Inventories 9 821,601 444,371<br />
b) Sundry debtors 10 1,304,813 1,236,691<br />
c) Cash and bank balances 11 67,694 40,518<br />
d) Loans and advances 12 392,263 502,704<br />
2,586,371 2,224,284<br />
Less : Current Liabilities and Provisions<br />
a) Current liabilities 13 705,349 605,762<br />
b) Provisions 14 39,772 11,717<br />
745,121 617,479<br />
Net Current Assets 1,841,250 1,606,805<br />
Miscellaneous Expenditure 15 6,419 24,076<br />
(to the extent not written off or adjusted)<br />
Total 3,792,579 3,043,039<br />
Notes to the Financial Statements 22<br />
Schedules referred to above and notes attached there to form an<br />
integral part of the Balance Sheet.<br />
This is the Balance Sheet referred to in our report of even date.<br />
For and on behalf of<br />
Price Waterhouse<br />
Chartered Accountants<br />
Membership No. F 055913<br />
For and on behalf ofthe Board of Directors.<br />
Partha Ghosh M. J. Mendonza Glenn Saldanha Rajesh Desai<br />
Partner Vice President - Legal & Managing Director & CEO Director - Finance<br />
Mumbai, April 22, 2004<br />
Company Secretary<br />
74
Profit and Loss Accounts<br />
Rs. In ('000s)<br />
For the year ended 31st March, Schedules 2004 2003<br />
INCOME<br />
Sales & Operating Income 16 3,806,606 3,336,365<br />
Other income 17 34,573 42,823<br />
3,841,179 3,379,188<br />
EXPENDITURE<br />
Cost of sales 18 1,793,191 1,644,080<br />
Selling and operating expenses 19 1,065,026 942,417<br />
Depreciation 6 108,891 103,326<br />
Interest 20 100,578 106,884<br />
Research and development expenses 21 248,073 147,257<br />
3,315,759 2,943,964<br />
Profit Before Tax 525,420 435,224<br />
Provision for taxation<br />
– Current Year [includes wealth tax provision Rs. 300 (Prev. Year–Rs. 300)] 90,298 35,300<br />
– Earlier Years – 18,429<br />
– Deferred tax 15,081 49,600<br />
Net Profit After Tax 420,041 331,895<br />
Balance Profit brought forward 235,883 29,073<br />
Net Profit Available for Appropriation 655,924 360,968<br />
Interim Dividend paid on pref shares 10,500 10,500<br />
Tax on dividend paid on pref shares 1,345 –<br />
Interim Dividend on Equity Shares 77,020 66,114<br />
Tax on interim dividend paid on equity shares 9,868 8,471<br />
Transfer to Debenture Redemption Reserve 253 –<br />
Transfer to General Reserve 60,000 40,000<br />
Balance Carried to Balance Sheet 496,938 235,883<br />
Earnings per share (Rs.) 22(2)<br />
Basic 7.41 6.33<br />
Diluted 7.37 5.67<br />
Face Value of Shares 2.00 2.00<br />
Notes to the Financial Statements 22<br />
Schedules referred to above and notes attached there to form an<br />
integral part of the Profit and Loss Account.<br />
This is the Profit and Loss Account referred to in our report of even date.<br />
For and on behalf of<br />
Price Waterhouse<br />
Chartered Accountants<br />
Membership No. F 055913<br />
For and on behalf ofthe Board of Directors.<br />
Partha Ghosh M. J. Mendonza Glenn Saldanha Rajesh Desai<br />
Partner Vice President - Legal & Managing Director & CEO Director - Finance<br />
Mumbai, April 22, 2004<br />
Company Secretary<br />
75
Cash Flow Statement<br />
Rs. In ('000s)<br />
Year ended 31st March, 2004 2003<br />
A. Cash flow from operating activities:<br />
Net Profit before tax 525,420 435,224<br />
Adjustments for:<br />
Depreciation 108,891 103,326<br />
Interest Expense 98,677 105,313<br />
Interest Expense - Finance lease 1,901 1,571<br />
Interest Income (2,333) (1,433)<br />
Income from Investment - Dividends (1,245) (20)<br />
(Profit)/Loss on Fixed Assets sold 720 (1,494)<br />
Deferred revenue expenditure written off 17,657 34,933<br />
Provision for Doubtful Advances – 4,000<br />
Provision for Bad & Doubtful Debts 11,800 16,000<br />
Provision for Gratuity & Leave Encashment 500 1,500<br />
Unrealised foreign exchange (gain) /loss (9,102) 965<br />
Employee stock option plan (4,593) 1,808<br />
Operating profit before working capital changes 748,293 701,693<br />
Adjustments for changes in working capital :<br />
– (increase)/decrease in Sundry Debtors (71,640) (511,630)<br />
– (increase)/decrease in Other Receivables (125,189) (10,917)<br />
– (increase)/decrease in Inventories (377,230) (122,091)<br />
– increase/(decrease) in Trade and Other Payables 126,466 83,581<br />
Cash generated from operations 300,700 140,636<br />
– Taxes (Paid) / Received (Net of Tax deducted at source) (62,343) (47,380)<br />
Net cash from operating activities 238,357 93,256<br />
B. Cash flow from Investing activities:<br />
Purchase of fixed assets (522,806) (182,528)<br />
Capital Work in Progress (12,495) (170,687)<br />
Proceeds from Sale of fixed assets 250,000 99,336<br />
Proceeds from Sale of Investments – 5,000<br />
Purchase of investments (138,521) (139,968)<br />
Finance Lease Rent payment against principal amount (6,974) (4,724)<br />
Interest Received 3,613 1,765<br />
Dividend Received 1,245 20<br />
Net cash used in investing activities (425,938) (391,786)<br />
76
Cash Flow Statement (Contd.)<br />
Rs. In ('000s)<br />
Year ended 31st March, 2004 2003<br />
C. Cash flow from financing activities:<br />
Proceeds from fresh issue of<br />
Share Capital (including Share Premium ) 508,185 (6,140)<br />
Proceeds / (Repayment) of long term borrowings (340,079) 393,251<br />
Receipt /(Repayment) of short term borrowings 65,450 (19,521)<br />
Proceeds from Cash Credits (NET) 182,918 107,326<br />
Finance Lease Rent (Interest Part only) (1,901) (1,571)<br />
Interest Paid (115,121) (110,536)<br />
Dividend Paid (76,224) (69,103)<br />
Dividend Tax Paid (8,471) –<br />
Net cash used in financing activities 214,757 293,706<br />
Net Increase/(Decrease) in Cash & Cash Equivalents 27,176 (4,824)<br />
Cash and cash equivalents as at 31.03.2003 40,518 45,342<br />
Cash and cash equivalents as at 31.03.2004 67,694 40,518<br />
Cash and cash equivalents comprise<br />
Cash 836 619<br />
Deposits 46,411 21,443<br />
Balance with Scheduled Banks 20,447 18,456<br />
67,694 40,518<br />
Notes :<br />
1 The Cash Flow Statement has been prepared under the "Indirect Method" as<br />
set out in Accounting Standard - 3 on Cash Flow Statements issued by the<br />
Institute of Chartered Accountants of India.<br />
2 Cash and cash equivalents includes Rs. 2,364 which are not available<br />
for use by the Company (Refer schedule 13 to the financial statements).<br />
3 Figures in bracket indicate Cash outgo.<br />
This is the Cash Flow Statement referred to in our report of even date<br />
For and on behalf of<br />
Price Waterhouse<br />
Chartered Accountants<br />
Membership No. F 055913<br />
For and on behalf ofthe Board of Directors.<br />
Partha Ghosh M. J. Mendonza Glenn Saldanha Rajesh Desai<br />
Partner Vice President - Legal & Managing Director & CEO Director - Finance<br />
Mumbai, April 22, 2004<br />
Company Secretary<br />
77
Schedules forming part of the Balance Sheet<br />
Rs. In ('000s)<br />
As at 31st March, 2004 2003<br />
1 Share Capital<br />
Authorised<br />
125,000,000 (2003 – 125,000,000) Equity shares of Rs 2/- each 250,000 250,000<br />
4,000,000 (2003 – 4,000,000) Cumulative redeemable<br />
non convertible preference shares of Rs 100/- each 400,000 400,000<br />
Unclassified capital 100,000 100,000<br />
Issued, Subscribed and Paid-up<br />
59,245,570 (2003 -- 50,860,000) Equity shares of Rs 2/- each 118,491 101,720<br />
Less: Calls in arrears 3 7<br />
118,488 101,713<br />
10.5%, 1,000,000 (2003 – 1,000,000) redeemable cumulative non-convertible<br />
preference shares of Rs. 100/- each (Redeemed on April 1, 2004 at par) 100,000 100,000<br />
Equity Share Warrants<br />
290,000 (2003 – 490,000) Equity share warrants of Rs. 0.20/- each 58 98<br />
Total 218,546 201,811<br />
Notes :<br />
1. At the Annual general meeting held on september 26, 2003, the shareholders approved the sub-division of Equity<br />
share from face value of Rs. 10 each to Rs. 2 each.Accordingly previous year's figures have been recast.<br />
2. Of the above equity shares,19,875,000 shares of Rs 2 each are allotted as fully paid-up Bonus Shares by capitalisation<br />
of reserves.<br />
3. In terms of Employee Stock Option Plan approved by the members,290,000 (2003 – 490,000) convertible warrants have<br />
been allotted to <strong>Glenmark</strong> Pharmaceuticals Limited Employees Welfare Trust. Each warrant carries an option and is<br />
convertible to one equity share of Rs 2. During the year, 200,000 (2003 – 225,000) warrants were converted into Equity<br />
Shares at an exercise price of Rs. 14.34 per share.<br />
4. During the Year Company formulated a new Employees Stock Option Scheme viz. ESOS’ 2003, pursuant to which<br />
7,00,725 options of Rs.2. each were granted at average market price. On exercise of the options so granted the Paid<br />
up equity share capital of the company will increase by a like number of shares.<br />
5. 5% Convertible Debentures aggregating to Rs. 5,000 Lakhs issued to CDC Financial Services (Mauritius) Ltd. and South<br />
Asia Regional Fund were converted into 8,185,570 fully paid up equity shares of Rs. 2 each at a premium of Rs.59.08<br />
per share as per the agreement based on predetermined price earning ratio on the profits for the year ended March<br />
31,2003 at the meeting of board of directors held on September 29,2003.<br />
6. As of March 31, 2004, the Trust has allotted 85,000 (2003 -- 320,000) warrants to the employees of the Company,<br />
which are outstanding. The options have been granted at an exercise price of Rs 14.34 per share in respect of 85,000<br />
shares.<br />
78
Schedules forming part of the Balance Sheet<br />
Rs. In ('000s)<br />
As at 31st March, 2004 2003<br />
2 Reserves and Surplus<br />
Share premium account<br />
Balance at the beginning of the year 500,518 507,064<br />
Addition on issue of shares 491,502 6,257<br />
Less: Calls in arrears 52 124<br />
Less : Issue cost – 12,679<br />
Closing balance 991,968 500,518<br />
General reserve<br />
Balance at the beginning of the year 550,773 510,773<br />
Add : Transferred during the year 60,000 40,000<br />
Closing balance 610,773 550,773<br />
Debenture Redemption Reserve<br />
Balance at the beginning of the year 29,664 29,664<br />
Addition during the year 253 –<br />
Closing balance 29,917 29,664<br />
Capital Reserve<br />
Balance at the beginning of the year 1,000 1,000<br />
Addition during the year – –<br />
Closing balance 1,000 1,000<br />
Employee stock option<br />
Employee stock options outstanding 8,219 8,763<br />
Add : Grant of Options at discount – 370<br />
Less : Conversion of Option 5,283 231<br />
Less : Cancellation of Option 884 683<br />
2,052 8,219<br />
Deferred employee stock compensation 2,113 4,465<br />
Add : Grant of Options at discount – 370<br />
Less : Amortisation of ESOP expense 1,436 2,039<br />
Less : Cancellation of Option 138 683<br />
539 2,113<br />
Net Employee Stock option 1,513 6,106<br />
Profit and loss account<br />
Balance 496,938 235,883<br />
Total 2,132,109 1,323,944<br />
3 Secured Loans<br />
note<br />
Term loan 1 350,000 200,000<br />
Convertible Debentures 2 – 500,000<br />
Non Convertible Debentures 3 140,000 140,000<br />
Working capital facilities 4 528,414 345,496<br />
Other loans 5 11,015 8,068<br />
Total 1,029,429 1,193,564<br />
Notes :<br />
1. Term loan is secured by the equitable mortgage of assets as a security.The creation of Charge is under process.<br />
2 5% Convertible Debentures aggregating to Rs. 5,000 Lakhs issued to CDC Financial Services (Mauritius) Ltd. and South<br />
Asia Regional Fund were converted into 8,185,570 fully paid up equity shares of Rs. 2 each at a premium of Rs.59.08<br />
per share as per the agreement based on predetermined price earning ratio on the profits for the year ended March<br />
31,2003 at the meeting of board of directors held on September 29,2003.<br />
79
Schedules forming part of the Balance Sheet<br />
3 Non convertible debentures 'NCD' includes:<br />
a) 10.05% NCD - A series aggregating Rs. Nil (Previous year Rs 50 million), allotted on September 7, 2001with a put<br />
and call option after twelve months and redeemable after three years.<br />
b) 10.35% NCD - B series aggregating Rs Nil (Previous year Rs. 90 million, allotted on September 7, 2001 with a call<br />
option after twelve months and redeemable after three years.<br />
All the above debentures are secured by way of a mortgage favouring the debenture trustees on the land and<br />
buildings, plant and machinery and other fixed assets and hypothecation of all movable assets (except raw materials<br />
and book debts, both present and future), situated at the research and development center at Mahape, Navi Mumbai.<br />
4 Working capital loan from banks are secured by hypothecation of stocks of raw materials, packing materials, finished<br />
goods, work in progress, receivables etc. They are additionally secured by way of personal guarantees of some of the<br />
directors and equitable mortgage on fixed assets at the manufacturing facility at Nasik and research and development<br />
centre at Sinnar, Nasik.<br />
5 Other loans are secured by way of hypothecation of vehicles.<br />
Rs. In ('000s)<br />
As at 31st March, 2004 2003<br />
4 Unsecured Loans<br />
Short term loan 105,117 –<br />
Security deposit 3,752 43,375<br />
Deferred Sales Tax Loan 10,445 10,445<br />
Other Deposits – 44<br />
Total 119,314 53,864<br />
Notes :<br />
1. The Company has availed of an interest free sales tax deferral loan under Part I of the 1983 and 1988 Package<br />
Schemes of the Government of Maharashtra, repayable after twelve years in six half-yearly installments.<br />
5 Deferred Tax Liability [Refer Note (1) (x) of Schedule 22]<br />
Liabilities<br />
Depreciation 291,291 263,116<br />
Others 1,890 6,740<br />
Total 293,181 269,856<br />
6 Fixed Assets [refer note (1) (ii) and (6) of Schedule 22]<br />
(Rs. In '000s)<br />
GROSS BLOCK DEPRECIATION NET BLOCK<br />
As on Additions Sales/Disposals As on As on For the year Sales/ As on As on As on<br />
1st Apr, during during 31st Mar, 1st Apr, Disposals 31st Mar, 31st Mar, 31st Mar,<br />
2003 the year the year 2004 2003 of Assets 2004 2004 2003<br />
Freehold Land 5,710 – – 5,710 – – – – 5,710 5,710<br />
Leasehold Land 21,125 35,914 – 57,039 775 505 – 1,280 55,759 20,350<br />
Factory Buildings 87,030 72,257 – 159,287 18,936 3,770 – 22,706 136,581 68,094<br />
Other Buildings & Premises 222,411 44,999 – 267,410 12,213 3,832 – 16,045 251,365 210,198<br />
Plant and Machinery 55,627 140,688 9,196 187,119 10,595 11,353 706 21,242 165,877 45,032<br />
Furniture and Fixtures 101,770 27,326 154 128,942 23,269 12,070 7 35,332 93,610 78,501<br />
Equipments 373,233 170,982 8,281 535,934 71,219 37,562 203 108,578 427,356 302,014<br />
Vehicles 29,543 8,502 6,021 32,024 12,981 4,792 4,451 13,322 18,702 16,562<br />
Brands 350,069 – – 350,069 93,352 35,007 – 128,359 221,710 256,717<br />
Total 1,246,518 500,668 23,652 1,723,534 243,340 108,891 5,367 346,864 1,376,670 1,003,178<br />
Previous Year 1,416,755 213,172 383,409 1,246,518 175,581 103,326 35,567 243,340 1,003,178 1,241,174<br />
Capital Work-in-process including Capital advances. 244,966 232,471<br />
80
Schedules forming part of the Balance Sheet<br />
Notes :<br />
1. Equipment and vehicles include assets aggregating Rs.19,308 (2003 -- Rs.19,559) [net book value as at March 31, 2004 -- Rs. 14,338 (2003 – Rs.17,023)] and Rs. 5,536<br />
(2003 -- Rs. 5,536) [net book value as at March 31, 2004 -- Rs. 3,708 (2003 -- Rs. 4,631)], respectively, which have been acquired on finance lease.<br />
2. Additions to assets include Rs. 4,488 being borrowing costs.<br />
3. Estimated amount of contracts remaining to be executed on capital account, net of advances, not provided for as at March 31, 2004 aggregate Rs. 59,039 (2003 --<br />
Rs.14,566)<br />
4. Capital Work in progress includes : 2004 2003<br />
Cost of Ankleshwar Plant plus addition thereto 68,157 160,283<br />
Capitalisation of Interest & Relevant costs of Ankleshwar Plant – 16,653<br />
Cost of Goa Plant plus addition there to 57,145<br />
Capital Advances 109,854 49,667<br />
Other work-in-processes 9,810 5,868<br />
Rs. In ('000s)<br />
As at 31st March, 2004 2003<br />
7 Investments [refer Note (1)(iv) of Schedule 22]<br />
LONG TERM INVESTMENTS<br />
Quoted - traded<br />
Equity shares<br />
9,000 (2003 – 9,000) Bank of India of Rs.10 each [Market Value Rs. 530 (2003 – Rs. 341)] 405 405<br />
1,718 (2003– 1,000) IDBI Bank Limited of Rs. 10 each [Market Value Rs. 85 (2003-- Rs. 23)] 34 18<br />
439 423<br />
Unquoted - non trade<br />
National Savings Certificate -Sixth Issue 48 48<br />
1 (2003 – 1) Time share of Dalmia Resorts Limited 20 20<br />
1 (2003 – 1) equity share of Esquados 340,000 of <strong>Glenmark</strong><br />
Pharmaceutica Limitada, Lisbon (Portugal) 48 48<br />
1,93,665 (2003 – 193665) shares of Bharuch Eco-Aqua Infrastructure Limited of Rs. 10<br />
each, fully paid up (Previous year Rs. 4 per share was paid up). 1,937 775<br />
100,000 12% cumulative preference shares of Rs 100 each fully<br />
paid up of Cheryl Laboratories (P) Limited 10,000 10,000<br />
1,350,000 7% cumulative preference shares of Rs 100 each fully<br />
paid up of <strong>Glenmark</strong> Laboratories Limited 135,000 135,000<br />
Investments in subsidiary companies<br />
a) 100,000 (2003 – 100,000) equity shares of Rs 10 each<br />
fully paid up of <strong>Glenmark</strong> Exports Limited 1,000 1,000<br />
b) 100,000 (2003 – 100,000) equity shares of Rs 10 each<br />
fully paid up of GM Pharma Limited 1,000 1,000<br />
c) Investment in equity and chartered of <strong>Glenmark</strong> Impex LLC, Russia 12,812 12,812<br />
[Roubles 8,235,958 (2003 - 8,235,958)]<br />
d) <strong>Glenmark</strong> Pharmaceuticals Inc.USA 110,492 –<br />
[2400000 shares of USD 1 each]<br />
e) <strong>Glenmark</strong> Farmaceutica Ltda, Brazil [Reals 1118857] 17,526 –<br />
f) <strong>Glenmark</strong> Philippines Inc., Philippines 9,325 –<br />
[Share capital -Pesos 11200000 (INR 9050) and<br />
Share application money Pesos 340260 (INR 275)]<br />
299,208 160,703<br />
Total 299,647 161,126<br />
81
Schedules forming part of the Balance Sheet<br />
Rs. In ('000s)<br />
As at 31st March, 2004 2003<br />
8 Deferred Tax Assets [Refer Note (1) (x) of Schedule 22]<br />
Assets<br />
Provision for Bad Debts and Doubtful Advances 23,102 13,340<br />
Other 525 2,043<br />
Total 23,627 15,383<br />
9 Inventories [Refer Note (1) (v) of Schedule 22]<br />
(As certified by the management)<br />
Raw materials 280,413 120,987<br />
Packing material 29,685 29,793<br />
Work-in-process 223,462 51,850<br />
Stores & Spares 2,574 3,419<br />
Finished goods 285,467 238,322<br />
Total 821,601 444,371<br />
10 Sundry Debtors<br />
Outstanding for more than six months<br />
Secured, considered good – –<br />
Unsecured, considered good 147,936 193,971<br />
Unsecured, considered doubtful 38,998 27,198<br />
186,934 221,169<br />
Less: Provision for doubtful debts 38,998 27,198<br />
147,936 193,971<br />
Other debts-<br />
Secured, considered good – –<br />
Unsecured, considered good 1,156,877 1,042,720<br />
1,156,877 1,042,720<br />
Total 1,304,813 1,236,691<br />
Debts due from <strong>Glenmark</strong> Exports Limited, subsidiary company, aggregate Rs 218,617 (2003 -- Rs 181,309).<br />
11 Cash and Bank Balances<br />
Cash in hand 836 619<br />
Balances with Schedule banks<br />
– Current accounts 18,244 7,653<br />
– Margin Money Account 42,092 17,283<br />
– EEFC Account 2,203 10,803<br />
– Deposit accounts 4,319 4,160<br />
Total 67,694 40,518<br />
The balances in the margin money accounts are given as security against guarantees issued by banks on behalf of the<br />
Company.<br />
82
Schedules forming part of the Balance Sheet<br />
Rs. In ('000s)<br />
As at 31st March, 2004 2003<br />
12 Loans and Advances (unsecured, considered good)<br />
<strong>Glenmark</strong> Laboratories Limited * 17,985 267,988<br />
Advances to subsidiaries<br />
<strong>Glenmark</strong> Pharmaceuticals Inc., U.S.A. [Maximum during the year – 7,944<br />
Rs. 7,944 (Previous Year -- Rs. 7,944)]<br />
<strong>Glenmark</strong> Exports Limited [Maximum during the year Rs 13,673 13,673 4,848<br />
(Previous Year -- Rs 4,848)]<br />
<strong>Glenmark</strong> Farmaceutica Ltda, Brazil [Maximum during the year 1,139 –<br />
Rs. 1,139 (Previous year - Rs.Nil)]<br />
Advance to Vendors 130,275 85,161<br />
Advances recoverable in cash or kind or for value to be received 149,681 82,345<br />
Balance with Excise authorities 19,620 13,530<br />
Deposits 59,890 40,888<br />
Total 392,263 502,704<br />
* Last year being an associate company [maximum amount outstanding during the year Rs. 267,988]<br />
13 Current Liabilities<br />
Acceptances 92,842 62,618<br />
Sundry creditors - Small scale industrial undertakings [Refer note (5) of Schedule 22] 20,867 16,474<br />
- Others 350,891 309,111<br />
Investor Education and Protection Fund shall be credited by<br />
– Unclaimed Dividend 2,364 1,974<br />
[There are no amounts due and outstanding to be credited to<br />
Investor Education and Protection Fund.]<br />
Advances from Customers 3,049 1,206<br />
Payable to Subsidiaries<br />
– <strong>Glenmark</strong> Pharmaceuticals Inc., U.S.A. 199 –<br />
Other Liabilities 138,662 115,108<br />
Interest accrued but not due 8,242 24,686<br />
Interim Dividend on Equity Shares 77,020 66,114<br />
Tax on Interim Dividend 9,868 8,471<br />
Tax on Preference Dividend 1,345 –<br />
Total 705,349 605,762<br />
14 Provisions<br />
Wealth tax 300 300<br />
Income-tax (net of advance tax) [Refer Note (1) (x) of Schedule 22] 37,472 9,917<br />
Provision for Gratuity and leave encashment 2,000 1,500<br />
Total 39,772 11,717<br />
15 Miscellaneous Expenditure [(refer Note (1) (vii) of Schedule 22]<br />
(to the extent not written off or adjusted)<br />
Implementation Expenses of ERP system 1,147 5,737<br />
Product launch, investigation and registration expenses 5,272 18,339<br />
Total 6,419 24,076<br />
83
Schedules forming part of the Profit and Loss Account<br />
Rs. In ('000s)<br />
For the year ended 31st March, 2004 2003<br />
16 Sales [(refer Note (1) (viii) of Schedule 22]<br />
Sale of goods* 3,796,569 3,326,949<br />
Income from services 10,037 9,416<br />
Total 3,806,606 3,336,365<br />
* includes sales tax and excise duty aggregating Rs 255,771 (2003 – Rs 251,600) and Rs 340,561 (2003 – Rs 307,802)<br />
respectively.<br />
17 Other Income<br />
Lease Rent [tax deducted at source Rs.403 (Prev year – Rs. 302)] 1,920 23,940<br />
Interest Income [tax deducted at source Rs. 400 (Prev year – Rs. 427)] 2,333 1,433<br />
Dividend received 1,245 20<br />
Exchange gain 4,642 4,109<br />
Export Incentive 7,613 61<br />
Profit on sale of fixed assets 128 1,631<br />
Miscellaneous income 16,692 11,629<br />
Total 34,573 42,823<br />
18 Cost of Sales<br />
Salary, wages and allowances 38,320 29,565<br />
Labour charges 82,227 61,133<br />
Consumption of raw & packing materials 966,088 712,886<br />
Purchase of Trading goods 286,013 306,895<br />
Excise duty paid 308,398 327,169<br />
Sales tax 255,771 251,600<br />
Power, fuel and water charges 31,365 7,462<br />
Consumable stores 24,583 15,601<br />
Repairs and maintenance - plant and machinery 17,066 5,513<br />
Rent, rates and taxes 1,578 1,560<br />
Other manufacturing expenses 540 2,638<br />
(Increase)/decrease in inventory (218,758) (77,942)<br />
Total 1,793,191 1,644,080<br />
84
Schedules forming part of the Profit and Loss Account<br />
Rs. In ('000s)<br />
For the year ended 31st March, 2004 2003<br />
19 Selling and Operating Expenses<br />
Salary and allowances 256,391 204,693<br />
Staff welfare 7,801 7,455<br />
Directors' salaries and allowances 44,762 25,514<br />
Incentive and commission 40,978 31,923<br />
Sales promotion expenses 184,386 184,292<br />
Export Commission 7,935 11,107<br />
Commission on sales 4,154 34,678<br />
Travelling expenses 151,626 133,243<br />
Freight outward 71,847 92,774<br />
Telephone expenses 19,431 13,948<br />
Rates and taxes 14,040 8,719<br />
Provision for doubtful debts 11,800 16,000<br />
Provision for doubtful advances – 4,000<br />
Insurance premium 9,173 7,437<br />
Electricity charges 7,258 7,805<br />
Rent 12,975 11,440<br />
Repairs & Maintenance 39,135 24,029<br />
Auditors remuneration<br />
Audit fees 1,100 750<br />
Other matters 1,020 620<br />
Out of pocket expenses 30 5<br />
Loss on sale of assets 848 137<br />
Other operating Expenses 178,336 121,848<br />
Total 1,065,026 942,417<br />
20 Interest Expense<br />
On loans from banks 35,777 30,713<br />
Other interest 64,801 76,171<br />
Total 100,578 106,884<br />
21 Research and Development Expenses [refer Note (1) (ix) of Schedule 22]<br />
Salary and other allowances 71,328 41,406<br />
Staff welfare expenses 4,633 2,527<br />
Directors Remuneration 2,517 1,368<br />
Consumable & Chemicals 80,560 52,971<br />
Electricity charges 12,777 9,542<br />
Repairs and maintenance 12,192 7,709<br />
Insurance premium 283 377<br />
Other expenses 63,783 31,357<br />
Total 248,073 147,257<br />
85
Schedules forming part of the Financial Statements<br />
For the year ended 31st March, 2004<br />
22 Notes to the Financial Statement<br />
1) SIGNIFICANT ACCOUNTING POLICIES<br />
i) Basis of Accounting<br />
The financial statements are prepared under the historical cost convention, on the accrual basis of accounting, in<br />
conformity with accounting principles generally accepted in India.<br />
ii) Fixed assets and depreciation<br />
Fixed assets are stated at cost less accumulated depreciation. The Company capitalises all costs relating to the<br />
acquisition and installation of fixed assets. Expenditure of revenue nature, incurred in setting up of new projects, is<br />
capitalised as an indirect cost towards construction of the fixed assets. Exchange differences relating to the<br />
acquisition of fixed assets are adjusted in the cost of the assets.<br />
Depreciation is provided using the straight line method, pro-rata to the period of use of assets, based on the useful<br />
lives of fixed assets as estimated by management, or at the rates specified in Schedule XIV of the Companies Act,<br />
1956, whichever is higher.<br />
Fixed assets having aggregate cost of Rs 5,000 or less are depreciated fully in the year of acquisition.<br />
The company has estimated the useful life of its assets as follows:<br />
Category<br />
Estimated useful life<br />
(in years)<br />
Plant and machinery 8 - 20<br />
Vehicles 5 - 6<br />
Equipments and air conditioners 4 - 20<br />
Furniture and fixtures 10<br />
Brands 10<br />
Leasehold land is amortised over the period of lease.<br />
iii) Foreign currency transactions<br />
Foreign currency transactions during the year are recorded at the rates of exchange prevailing on the date of the<br />
transaction. Foreign currency assets and liabilities are translated into rupees at the exchange rates prevailing on the<br />
date of the balance sheet. All exchange differences are dealt with in the statement of profit and loss, except those<br />
relating to the acquisition of fixed assets, which are adjusted in the cost of the respective fixed assets.<br />
iv) Investments<br />
Long term investments are stated at cost. Provision, where necessary, is made to recognise a decline, other than<br />
temporary, in the value of the investments.<br />
v) Inventories<br />
Inventories of raw materials, packing materials, work-in-process and finished goods are valued at cost or net<br />
realisable value, whichever is lower. Cost of raw materials and packing materials is ascertained on a first-in-first out<br />
basis. Cost of work-in-process and finished goods includes the cost of materials consumed, labour and<br />
manufacturing overheads. Excise and customs duty accrued on production or import of goods, as applicable, is<br />
included in the valuation of inventories.<br />
vi) Employee Benefits<br />
Retirement benefits to employees comprise payments towards gratuity, superannuation and provident fund under<br />
the schemes of the Company and encashment of leave. Annual contributions to the superannuation and provident<br />
funds are charged to the statement of profit and loss.<br />
The employee leave encashment and gratuity schemes are administered through the Life Insurance Corporation of<br />
India. Annual contributions as determined by the LIC are charged to the statement of profit and loss.<br />
86
Schedules forming part of the Financial Statements<br />
For the year ended 31st March, 2004<br />
22 Notes to the Financial Statement (Contd.)<br />
In respect of stock options granted to employees under the Company’s Employee Stock Option Plan (‘ESOP’), the<br />
excess of the market price of the share at the date of grant of the option over its exercise price is treated as a form<br />
of employee compensation in the financial statements of the company. This amount is amortised on a straight-line<br />
basis over the vesting period. The unamortised portion is carried forward as deferred employee compensation.<br />
vii) Miscellaneous expenditure<br />
Product launch expenditure<br />
Earlier years' expenditure on launch of new products and their sales promotion is being amortised over a period of<br />
three years.<br />
Implementation expenses of Enterprise Resource Planning system.<br />
Earlier year’s expenditure incurred on payments for infrastructure facilities and expenditure incurred on user license<br />
fees for an Enterprise Resource Planning system is being amortised over a period of thirty-six months.<br />
viii) Revenue recognition<br />
The Company recognises revenues on despatch of goods to customers. Revenues from services are recognised on<br />
completion of such services. Revenues are recorded at invoice value, inclusive of excise duty and sales-tax, but net<br />
of returns and trade discounts.<br />
ix) Research and development<br />
Capital expenditure on research and development ('R&D') is capitalised as fixed assets. Other expenditure on R&D<br />
is expensed as incurred.<br />
x) Income-tax<br />
Provision for current income-taxes is made on the assessable income at the tax rate applicable to the relevant<br />
assessment year. Deferred income taxes are recognised for the future tax consequences attributable to timing<br />
differences between the financial statement determination of income and their recognition for tax purposes. The<br />
effect on deferred tax assets and liabilities because of a change in tax rates is recognised in statement of profit and<br />
loss using the tax rates and tax laws that have been enacted or substantively enacted by the balance sheet date.<br />
Deferred tax assets are recognised and carried forward only to the extent that there is a reasonable certainty that<br />
sufficient future taxable income will be available against which such deferred tax assets can be realised.<br />
Prior / Earlier year's income tax is charged to the Profit and Loss account on payment and the same is disclosed<br />
separately.<br />
xi) Leases<br />
Finance leases<br />
Assets acquired under finance lease are recognised as assets with corresponding liabilities in the balance sheet at<br />
the inception of the lease at amounts equal to the fair value of the leased asset or, if lower, at the present value of<br />
the minimum lease payments. These leased assets are depreciated in line with the Company’s policy on<br />
depreciation of fixed assets. The interest is allocated to periods during the lease term so as to produce a constant<br />
periodic rate of interest on the remaining balance of the liability for each period.<br />
Operating leases<br />
Lease payments for operating leases are recognised as expense on a straight-line basis over the lease term.<br />
Lease income from operating leases is recognised as income on a straight-line basis over the lease term. Initial<br />
direct costs are recognised immediately as an expense.<br />
2) EARNINGS PER SHARE<br />
Basic earnings per share are calculated by dividing the net profit for the year attributable to equity shareholders (net<br />
profit for the year less dividends on preference shares) by the weighted average number of equity shares<br />
outstanding during the year.<br />
87
Schedules forming part of the Financial Statements<br />
For the year ended 31st March, 2004<br />
22 Notes to the Financial Statement (Contd.)<br />
For the purpose of calculating diluted earnings per share, the net profit attributable to equity shareholders and the<br />
weighted average number of shares outstanding are adjusted for the effects of all dilutive potential equity shares<br />
from the exercise of options on unissued share capital. The number of equity shares is the aggregate of the<br />
weighted average number of equity shares and the weighted average number of equity shares, which would be<br />
issued on the conversion of all the dilutive potential equity shares into equity shares. Options on unissued equity<br />
share capital are deemed to have been converted into equity shares on the date when the options are exercised.<br />
The calculations of earnings per share (basic and diluted) are based on the earnings and number of shares as<br />
computed below.<br />
Rs. In ('000s)<br />
Reconciliation of earnings 31st March, 2004 31st March, 2003<br />
Profit after tax for the financial year 420,041 331,895<br />
Less: Preference dividends 10,500 10,500<br />
Dividend tax on preference shares 1,345 –<br />
Net profit attributable to equity shareholders for calculation of Basic EPS 408,196 321,395<br />
Add : Interest accrued on Convertible Debentures less tax thereon – 15,333<br />
Net profit attributable to equity shareholders for calculation of Diluted EPS 408,196 336,728<br />
Reconciliation of number of shares<br />
Shares<br />
Shares<br />
Weighted average number of shares:<br />
For basic earnings per share 55,111 50,780<br />
Add: Deemed exercise of options on unissued equity share capital 274 215<br />
Conversion of Debentures – 8,515<br />
For diluted earnings per share 55,385 59,510<br />
Earnings per share (nominal value Rs 2 each) Rs Rs<br />
Basic 7.41 6.33<br />
Diluted 7.37 5.67<br />
3) SEGMENT INFORMATION<br />
Business segments<br />
The Company is primarily engaged in a single segment business of manufacturing and marketing of pharmaceuticals<br />
formulations and active ingredients and is managed as one entity, for its various activities and is governed by a similar<br />
set of risks and returns.<br />
Geographical segments<br />
The operations of the Company are largely confined to India, with exports contributing to 17.17 percent of its annual<br />
sales. Hence, in the view of the management, the Indian and export markets represent geographical segments.<br />
Sales by market -- The following is the distribution of the Company's sale by geographical market:<br />
2004 2003<br />
Geographical segment Rs. In '000s Rs. In '000s<br />
India 3,152,961 3,023,242<br />
Other than India* 653,645 313,123<br />
Total 3,806,606 3,336,365<br />
* includes deemed exports aggregating Rs 152,642 (2003 – Rs. 126,723)<br />
88
Schedules forming part of the Financial Statements<br />
For the year ended 31st March, 2004<br />
22 Notes to the Financial Statement (Contd.)<br />
Assets and additions to fixed assets by geographical area – The following table shows the carrying amount of segment<br />
assets and additions to fixed assets by geographical area in which the assets are located:<br />
India Others* India Others*<br />
2004 2004 2003 003<br />
Rs. In '000s Rs. In '000s Rs. In '000s Rs. In '000s<br />
Carrying amount of segment assets 3,773,369 459,143 3,119,819 237,323<br />
Additions to tangible assets 500,669 – 213,172 –<br />
* Others represent receivables from debtors located outside India including those related to deemed exports.<br />
4) RELATED PARTY DISCLOSURES<br />
a) Parties where control exists<br />
Wholly owned subsidiary companies<br />
<strong>Glenmark</strong> Exports Limited<br />
GM Pharma Limited<br />
<strong>Glenmark</strong> Impex LLC , Russia<br />
<strong>Glenmark</strong> Farmaceutica Ltda, Brazil<br />
<strong>Glenmark</strong> Philippines Inc., Philippines<br />
<strong>Glenmark</strong> Pharmaceuticals (U.K.) Ltd , U.K.<br />
Subsidiary companies<br />
<strong>Glenmark</strong> Pharmaceuticals inc, USA<br />
b) Related party relationships where transactions have taken place during the year<br />
Subsidiary Companies<br />
<strong>Glenmark</strong> Exports Limited<br />
<strong>Glenmark</strong> Pharmaceuticals Inc., USA<br />
<strong>Glenmark</strong> Farmaceutica Ltda, Brazil<br />
<strong>Glenmark</strong> Philippines Inc., Philippines<br />
Key management personnel (includes directors of the Company)<br />
Mrs B.E. Saldanha<br />
Mr Glenn Saldanha<br />
Mrs Cheryl Pinto<br />
Mr. R.V. Desai<br />
Mr. A.S. Mohanty<br />
Mr. S.A. Paigankar (Resigned on 29.03.04)<br />
c) Transactions with related parties during the year<br />
Rs. In ('000s)<br />
31st March,<br />
31st March,<br />
2004 2003<br />
Subsidiary company<br />
Sale of finished products 150,750 126,723<br />
<strong>Glenmark</strong> Exports Limited 149,009 126,723<br />
<strong>Glenmark</strong> Pharmaceuticals Inc, USA 1,741 –<br />
Lease rent received from <strong>Glenmark</strong> Laboratories Ltd – 22,500<br />
Advances given 2,223 –<br />
<strong>Glenmark</strong> Pharmaceuticals Inc., USA 1,084 –<br />
<strong>Glenmark</strong> Farmaceutica Ltda, Brazil 1,139 –<br />
89
Schedules forming part of the Financial Statements<br />
For the year ended 31st March, 2004<br />
22 Notes to the Financial Statement (Contd.)<br />
c) Transactions with related parties during the year (Contd.)<br />
Rs. In ('000s)<br />
31st March,<br />
31st March,<br />
2004 2003<br />
Purchase of finished goods from <strong>Glenmark</strong> Pharmaceuticals Inc, USA 3,025<br />
Reimbursement of expenses to <strong>Glenmark</strong> Exports Limited 26,740 15,111<br />
Purchase of fixed assets from G M pharma Limited – 39,671<br />
Share application money <strong>Glenmark</strong> Impex LLC – 4,193<br />
Investment in Share Capital 129,399 –<br />
<strong>Glenmark</strong> Pharmaceuticals Inc, USA 102,548 –<br />
<strong>Glenmark</strong> Farmaceutica Ltda, Brazil 17,526 –<br />
<strong>Glenmark</strong> Philippines Inc Philippines 9,325 –<br />
Associate enterprises<br />
Sale of fixed assets to <strong>Glenmark</strong> Laboratories Limited – 345,000<br />
Investment in Preference Share Capital<br />
of <strong>Glenmark</strong> Laboratories Limited – 135,000<br />
Corporate guarantees given for <strong>Glenmark</strong> Laboratories Limited – 50,000<br />
* glenmark Laboratories Limited [Last year being an associate company]<br />
Key management personnel<br />
Professional fees – 61<br />
Share in profit of Cardiac Centre – 59<br />
Remuneration paid to these personnel have been<br />
disclosed in note 8 of Schedule 22<br />
Personal guarantees given by directors have been disclosed in Schedule 3<br />
d) Related party balances<br />
Receivable from wholly owned subsidiary companies 233,429 189,253<br />
<strong>Glenmark</strong> Exports Limited 232,291 181,309<br />
<strong>Glenmark</strong> Farmaceutica Ltda, Brazil 1,138 –<br />
<strong>Glenmark</strong> Pharmaceuticals Inc, USA – 7,944<br />
Payable to subsidiary company<br />
<strong>Glenmark</strong> Pharmaceuticals Inc, USA 199 –<br />
Receivable from associate enterprises<br />
<strong>Glenmark</strong> Laboratories Limited – 267,988<br />
e) Debts written off / Provisions make against related party balances<br />
Debts written off of wholly owned subsidiary company<br />
(provided for the same in 2001-02) – 10,300<br />
5) OUTSTANDING DUES TO SMALL SCALE INDUSTRIAL UNDERTAKINGS<br />
The small scale industrial undertakings to whom amounts are outstanding for<br />
more than 30 days are:<br />
Alcap Containers Pvt Ltd 456 21<br />
Blown Enterprise 76 –<br />
Corneilo Packaging 59 –<br />
Joy Enterprise 3 3<br />
K K Alu Foil 706 105<br />
K Laminates 249 71<br />
Manju Industrial Ancillaries 126 –<br />
Print Paks (India) 26 26<br />
90
Schedules forming part of the Financial Statements<br />
For the year ended 31st March, 2004<br />
22 Notes to the Financial Statement (Contd.)<br />
Rajlaxmi Plastics 330 –<br />
Super Label Mfg Co. 14 14<br />
Varsha Plast 2 2<br />
Waxoils Pvt Ltd 126 3<br />
Aviditya Chem. Corpn. 36 –<br />
Plascap Industries 162 2<br />
Mahesh Industry 3,306 –<br />
Kraft-Pack Containers 94 –<br />
Autofits 13 –<br />
Crown Closures Pvt Ltd 186 –<br />
D M Printers 32 –<br />
Servewell Printers 1,086 166<br />
Pharma Plastics 254 251<br />
Desicca Chemicals 1 1<br />
Renuka Industries 73 –<br />
Carewell Printers Pvt. Ltd. – 3<br />
Standard Packprints Pvt Ltd 207 86<br />
Eskay Packaging 107 –<br />
Mahalsa Chemicals – 2<br />
Bina Packaging & Printers Pvt Ltd 413 217<br />
Supreme Alutainers p. ltd 91 –<br />
Akshar Ent. 4 –<br />
Dayaram Chemicals 128 –<br />
Plastic pigments p. ltd 8 –<br />
Devi Industrial Corporation – 14<br />
Minicon Enterprises – 131<br />
Synthochem Pvt. Ltd. 59 30<br />
Urmi Chemicals – 156<br />
8433 1,304<br />
Amount outstanding for less than 30 days 12,434 15,170<br />
Total 20,867 16,474<br />
6) LEASES<br />
a) The Company has entered into operating and finance lease agreements for<br />
the rental of property, vehicles,computers, equipment and other assets.<br />
Typically, lease agreements are for a period of three to five years.<br />
At March 31 2004, the Company had commitments under<br />
non-cancellable finance leases as follows:<br />
Rs. In ('000s)<br />
31st March, 31st March, 2003<br />
2004 2003<br />
Minimum lease payments<br />
Due within one year 7,368 8,875<br />
Due later than one year and not later than five years 4,146 11,514<br />
Total 11,514 20,389<br />
Present value of minimum lease payments<br />
Due within one year 5,528 7,454<br />
Due later than one year and not later than five years 2,728 8,256<br />
Total 8,256 15,710<br />
91
Schedules forming part of the Financial Statements<br />
For the year ended 31st March, 2004<br />
22 Notes to the Financial Statement (Contd.)<br />
b) The Company has leased out its manufacturing facility at Panoli,<br />
Gujarat and the same has been capitalised in the books of account in<br />
accordance with Accounting Standard 19 - "Leases" issued by The<br />
Institute of Chartered Accountants of India in this regard.<br />
Rs. In ('000s)<br />
31st March, 2004 31st March, 2003<br />
Depreciation has been provided based on the estimated useful life of the asset.<br />
(i) Details in respect of assets given on operating Lease<br />
Gross Block<br />
Leasehold Land 4,259 4,259<br />
Factory Buildings 22,065 22,065<br />
Plant and Machinery 5,838 5,838<br />
Equipments 3,764 3,764<br />
Furniture and Fixtures 165 165<br />
36,091 36,091<br />
Accumulated depreciation<br />
Leasehold Land 172 129<br />
Factory Buildings 3,302 2,566<br />
Plant and Machinery 1,104 826<br />
Equipments 1,272 971<br />
Furniture and Fixtures 66 49<br />
5,916 4,541<br />
Depreciation 1,375 1,375<br />
(ii) The lease income of Rs.1,920 (Previous Year -- Rs. 1,440) has been<br />
accrued on the basis of the lease agreement executed with the lessees.<br />
This lease is cancellable by notice of 30 days on either side.<br />
c) The Company has taken on operating lease a Bulk Drug Plant at<br />
Kurkumbh and godowns/residential & office premises at various<br />
locations in the country.<br />
i) The Company's significant leasing arrangements are in respect of the<br />
above plant, godowns & premises (Including furniture and fittings<br />
therein, as applicable ). The aggregate lease rentals payable are charged<br />
to Profit and Loss Account as Rent in Schedule 18 & 20.<br />
ii) The Leasing arrangements which are cancellable range between<br />
11 months and 3 years. They are usually renewable by mutual consent<br />
on mutually agreeable terms. Under these arrangements, generally<br />
refundable interest free deposits have been given. An amount of Rs.13,742<br />
towards deposit and unadjusted advance rent is recoverable from the lessor.<br />
92
Schedules forming part of the Financial Statements<br />
For the year ended 31st March, 2004<br />
22 Notes to the Financial Statement (Contd.)<br />
7) CONTINGENT LIABILITIES NOT PROVIDED FOR<br />
Rs. In ('000s)<br />
31st March, 2004 31st March, 2003<br />
Bank guarantees 9,981 6,856<br />
Corporate guarantee 50,000 50,000<br />
Disputed taxes/duties – 3,185<br />
Labour / Industrial disputes 1,120 –<br />
Open letters of credit 83,350 43,853<br />
Call Money payable to Bharuch Eco-aqua Infrastructure (@Rs. 6/- per share) – 1,162<br />
Sundry debtors factored with recourse option 91,677 126,285<br />
Channel financing with recourse option 14,159 –<br />
8) COMPUTATION OF NET PROFITS IN ACCORDANCE WITH<br />
SECTION 349 AND SECTION 309(5) OF THE COMPANIES ACT, 1956<br />
Profit before taxation as per statement of profit and loss 525,420 435,224<br />
Add: Depreciation as per statement of profit and loss 108,891 103,326<br />
Provision for Doubtful Debts & Advances 11,800 20,000<br />
Loss on sale of assets as per statement of profit and loss 848 137<br />
646,959 558,687<br />
Less: Depreciation calculated under section 350 of the<br />
Companies Act, 1956 108,891 103,326<br />
Profit on sale of assets as per statement of profit and loss 128 1,631<br />
Net profit in accordance with Section 349 537,940 453,730<br />
Add: Managerial remuneration paid/payable to directors 48,850 29,268<br />
Net profit in accordance with Section 309(3) of the 586,790 482,998<br />
Companies Act, 1956<br />
Maximum managerial remuneration allowed under Section 198 of the<br />
Companies Act, 1956, 10 per cent of the above 58,679 48,300<br />
Managerial remuneration paid/payable to directors<br />
Salaries 15,742 10,976<br />
Commission 26,806 7,501<br />
Perquisites 1,571 2,386<br />
Sitting Fees 201 37<br />
Contribution to PF & Superannuation Fund 2,234 935<br />
Gratuity – 5,669<br />
Other benefits / reimbursements 2,296 1,764<br />
Total 48,850 29,268<br />
Name of Directors 2004 2003<br />
1 Mr. Gracias Saldanha 10,104 7,660<br />
2. Mrs. B. E. Saldanha 7,177 5,155<br />
3. Mr. Glenn Saldanha 14,560 5,752<br />
4. Mrs. Cheryl Pinto 7,411 3,365<br />
5. Others 9,598 7,335<br />
93
Schedules forming part of the Financial Statements<br />
For the year ended 31st March, 2004<br />
22 Notes to the Financial Statement (Contd.)<br />
9) CAPACITY, PRODUCTION, SALES AND STOCKS<br />
(a) Capacities and actual production (includes samples)<br />
Rs. In ('000s)<br />
Class of goods Installed capacity Actual production<br />
UoM 2004 2003 2004 2003<br />
Injectibles Ltrs – – 75,646 69,958<br />
Liquid orals Ltrs 2,475,000 2,475,000 1,860,730 1,684,833<br />
Lotions and externals Ltrs 450,000 450,000 385,689 393,566<br />
Ointments and creams Kgs 780,000 330,000 408,965 449,075<br />
Solids and powders Kgs 105,000 105,000 198,151 139,305<br />
Tablets and capsules Nos 240,000,000 240,000,000 385,100,000 381,500,000<br />
Bulk drugs Kgs 60,000 – 18,832 6,681<br />
Notes:<br />
(i) The products of the Company are exempt from licencing procedures.<br />
(ii) Installed capacity, being a technical matter, has not been verified by the auditors However, the management has<br />
certified the same.<br />
(iii) Actual production includes goods manufactured at third party manufacturing facilities on loan licence basis and at<br />
leased facilities.<br />
(iv) Installed capacity of Ointment and cream has been increased due to production started at Goa plant from<br />
23.03.2004.<br />
(b) Sales<br />
Rs. In ('000s)<br />
Product 2004 2003<br />
UoM Qty Value Qty Value<br />
Injectibles Ltrs 76,211 139,548 62,481 111,086<br />
Liquid orals Ltrs 2,298,267 676,726 2,007,858 573,725<br />
Lotions and externals Ltrs 492,514 435,245 445,512 433,673<br />
Ointments and creams Kgs 407,882 723,888 429,302 718,635<br />
Solids and powders Kgs 186,404 68,423 128,615 49,669<br />
Tablets and capsules Nos 441,025,700 1,433,031 407,300,000 1,366,315<br />
Bulk drugs 295,681 68,979<br />
Cardiac diagnostic services 10,037 9,416<br />
Others 24,027 4,867<br />
Total 3,806,606 3,336,365<br />
1. Sales are net of sales returns.<br />
2. Sales quantities does not include free issues, samples and breakages.<br />
(c) Finished goods purchased (includes samples)<br />
Rs. In ('000s)<br />
Product 2004 2003<br />
UoM Qty Value Qty Value<br />
Injectibles Ltrs 1,293 1,699 9,734 33,698<br />
Liquid orals Ltrs 445,181 80,331 515,182 73,437<br />
Lotions and externals Ltrs 147,840 41,184 90,916 28,856<br />
Ointments and creams Kgs 13,553 12,328 11,530 11,943<br />
Tablets and capsules Nos 65,887,200 132,944 75,659,900 158,961<br />
Bulk Drugs/ Drug intermediate 2,797 17,527<br />
Total 286,013 306,895<br />
94
Schedules forming part of the Financial Statements<br />
For the year ended 31st March, 2004<br />
22 Notes to the Financial Statement (Contd.)<br />
(d) Raw and packing materials consumed (quantities in kgs)<br />
Rs. In ('000s)<br />
Product 2004 2003<br />
Qty Value Qty Value<br />
Valdecoxib 2,698 60,191 2,113 77,327<br />
Etoricoxib 840 59,742 – –<br />
Rosuvastatin Calcium 92 48,175 – –<br />
Linezolid 702 21,852 655 26,332<br />
Linezolid purified IV grade 242 6,946 118 8,439<br />
Telmisartan 408 27,438 – –<br />
Mupirocin 148 24,853 77 15,159<br />
Cefuroxime sodium sterile BP / USP 678 19,843 – –<br />
Cefdinir 445 18,914 521 26,325<br />
Propylene Glycol I.P./ B.P. 264,335 17,337 181,486 11,523<br />
Tacrolimus 1,587 16,373 – –<br />
Acarbose 355 15,356 258 13,204<br />
Glycerine Refined IP/BP 219,247 11,985 217,515 12,792<br />
Sugar - S/30 888,662 11,806 790,453 10,259<br />
Itraconazole Pellets 1,072 9,514 840 7,463<br />
Pregelatinised starch BP/USP 4,188 9,134 – –<br />
Sorbitol Soln 70% IP 448,457 9,099 408,756 8,042<br />
Beclomethasone Dipropionate IP/BP 47 8,918 67 14,542<br />
Cefuroxime Axetil 100% Amorophous 431 8,652 – –<br />
Esomeprazole Mag Trihydrate 838 8,558 1,175 22,066<br />
Ketoconazole IP/BP 1,214 8,432 966 6,797<br />
Cilastazol 378 8,033 – –<br />
Nateglinide 279 7,774 564 25,980<br />
Others 527,163 – 426,636<br />
966,088 712,886<br />
(e) Break-up of Materials and Consumable stores consumed<br />
Rs. In ('000s)<br />
Product 2004 2003<br />
Value % Value %<br />
Materials<br />
Imported materials 282,466 29.24 144,515 20.27<br />
Indigenously procured 683,622 70.76 568,371 79.73<br />
966,088 100.00 712,886 100.00<br />
Consumable stores<br />
Imported – – – –<br />
Indigenously procured 24,583 100.00 15,601 100.00<br />
24,583 100.00 15,601 100.00<br />
95
Schedules forming part of the Financial Statements<br />
For the year ended 31st March, 2004<br />
22 Notes to the Financial Statement (Contd.)<br />
(f) Inventories of finished goods (includes samples)<br />
Rs. In ('000s)<br />
Opening Stock<br />
Closing Stock<br />
2004 2003 2004 2003<br />
Products UoM Qty Value Qty Value Qty Value Qty Value<br />
Injectibles Ltrs 16,597 29,066 6,185 10,448 20,004 19,330 16,597 29,066<br />
Liquid orals Ltrs 201,403 34,683 258,084 36,098 148,822 29,755 201,403 34,683<br />
Lotions and externals Ltrs 40,772 14,959 39,596 14,048 63,206 21,578 40,772 14,959<br />
Ointments and creams Ltrs 39,019 26,088 47,032 29,471 53,169 41,851 39,019 26,088<br />
Solids and powders Kgs 5,876 1,228 6,739 1,947 15,680 2,874 5,876 1,228<br />
Tablets and capsules Nos 70,909,000 132,247 56,086,700 113,676 67,633,200 157,822 70,909,000 132,247<br />
Pesticides – 1,491 – –<br />
Others 51 – 8,807 51<br />
Bulk Drugs 3,451<br />
238,322 207,179 285,468 238,322<br />
10) VALUE OF IMPORTS ON CIF BASIS<br />
Rs. In ('000s)<br />
31st March, 2004 31st March, 2003<br />
Capital Goods 76,821 30,509<br />
Raw Materials 219,869 90,159<br />
296,690 120,668<br />
11) EARNINGS IN FOREIGN CURRENCY<br />
Export of goods 486,384 179,496<br />
Others 3,155 3,665<br />
489,539 183,161<br />
12) EXPENDITURE IN FOREIGN CURRENCY<br />
Travelling expenses 4,568 8,149<br />
Professional & Consultancy charges 34,847 10,687<br />
Others 116,312 47,133<br />
155,727 65,969<br />
13) DIVIDEND REMITTANCE IN FOREIGN CURRENCY<br />
Number of Non-resident Shareholders 12 13<br />
Number of Equity Shares held by them 290,250 293,250<br />
Amount of dividend paid (Gross), TDS Rs Nil (2003 -- Rs 71,739) 377,325 351,900<br />
Year to which dividend relates 2002-2003 2001-2002<br />
14) PRIOR YEAR COMPARATIVES<br />
Prior year's figures have been regrouped wherever necessary.<br />
96
Balance Sheet Abstract and Company’s General Business Profile<br />
(a) Registration Details<br />
Registration No. 1 9 9 8 2<br />
State Code<br />
Rs. In ('000s)<br />
1 1<br />
Balance Sheet Date<br />
(b) Capital raised during the year<br />
Public Issue<br />
N I L<br />
Bonus Issue<br />
N I L<br />
(c) Position of Mobilisation and Deployment of Funds<br />
Total Liabilities and shareholders funds<br />
4 5 3 7 7 0 0<br />
Sources of Funds<br />
Paid-up Capital<br />
2 1 8 5 4 6<br />
Secured loan<br />
1 0 2 9 4 2 9<br />
Application of Funds<br />
Net fixed assets<br />
1 6 2 1 6 3 6<br />
Net current assets<br />
1 8 4 1 2 5 0<br />
Accumulated losses<br />
N I L<br />
(D) PERFORMANCE OF THE COMPANY<br />
Turnover<br />
3 8 0 6 6 0 6<br />
Profit/(Loss) Before Tax<br />
5 2 5 4 2 0<br />
3 1 0 3 2 0 0 4<br />
Date Month Year<br />
Rights Issue<br />
N I L<br />
Private Placement<br />
1 6 7 7 5<br />
Total Assets<br />
4 5 3 7 7 0 0<br />
Reserves & Surplus<br />
2 1 3 2 1 0 9<br />
Unsecured loans<br />
1 1 9 3 1 4<br />
Investments<br />
2 9 9 6 4 7<br />
Miscellaneous expenditure<br />
6 4 1 9<br />
Total Expenditure<br />
3 3 1 5 7 5 9<br />
Profit/(Loss) After Tax<br />
4 2 0 0 4 1<br />
Basic Earnings per share in Rs.<br />
7 . 4 1<br />
Dividend rate<br />
65%<br />
(E) Generic Names of Three Principal Products of Company<br />
Item Code No (ITC code)<br />
3 0 0 4 9 0 . 9 9<br />
3 0 0 4 9 0 . 9 9<br />
3 0 0 4 8 0 . 0 0<br />
Signatures to Schedules 1 to 22<br />
Diluted Earnings per share in Rs.<br />
7 . 3 7<br />
Product Description<br />
Clotrimazole<br />
Valdecoxib<br />
Terbutaline sulphate + Bromhexine Hydrocloride + Guaifenesin<br />
For and on behalf of<br />
Price Waterhouse<br />
Chartered Accountants<br />
Membership No. F 055913<br />
For and on behalf ofthe Board of Directors.<br />
Partha Ghosh M. J. Mendonza Glenn Saldanha Rajesh Desai<br />
Partner Vice President - Legal & Managing Director & CEO Director - Finance<br />
Mumbai, April 22, 2004<br />
Company Secretary<br />
97
SECTION 212<br />
STATEMENT PURSUANT TO SECTION 212 OF THE COMPANIES ACT, 1956, RELATING TO COMPANY’S INTEREST<br />
IN SUBSIDIARY COMPANIES:<br />
Name of the Company <strong>Glenmark</strong> Exports GM Pharma <strong>Glenmark</strong> <strong>Glenmark</strong> <strong>Glenmark</strong> <strong>Glenmark</strong> <strong>Glenmark</strong><br />
Limited Limited Impex L.L.C Farmaceutica Pharmaceuticals Philippines Pharmaceuticals<br />
Ltda. (U.K.) Ltd. Inc., Inc., USA<br />
1. The financial year of the Subsidiary March 31, March 31, December 31, December 31, March 31, March 31, March 31,<br />
Companies ended 2004 2004 2003 2003 2004 2004 2004<br />
2. Date from which they became subsidiary September 10, October 5, March 7, November 18, February 10, January 28, December 10,<br />
1996 1999 2001 2003 2004 2004 2002<br />
3. a. Number of shares held by <strong>Glenmark</strong> 1,00,020 Equity 1,00,000 Equity 1 share of 111,88,57 Equity 99 shares of 56,000 2400,000 shares<br />
Pharmaceuticals Ltd. in the subsidiary Shares of Rs. Shares of Rs. US $ 263740 £1.00 shares of of 1 $<br />
companies at the end of financial year of 10/- each fully 10/- each fully 200 pesos each<br />
Subsidiary Companies paid up paid up each<br />
b. Extent of interest of holding Company 100% 100% 100% 100% 100% 100% 96%<br />
at the end of the financial year of the<br />
subsidiary companies<br />
4. The net aggregate amount of the<br />
subsidiary companies’ Profit/ (Loss)<br />
so far as it concerns the members<br />
of the holding company:<br />
a. Not dealt within the holding<br />
company’s accounts:<br />
1) For the financial year ended Nil (7420) Nil Nil Nil Nil (63,70,683)<br />
31st March, 2004<br />
2) For the previous financial years of the Rs. 73,78,445 Nil Nil Nil Nil Nil Nil<br />
Subsidiary Companies since they became<br />
the holding company’s subsidiaries<br />
b. Dealt within the holding<br />
company’s accounts:<br />
1) For the financial year ended Nil Nil Nil Nil Nil Nil Nil<br />
31st March, 2004<br />
2) For the previous financial years of Nil Nil Nil Nil Nil Nil Nil<br />
the subsidiary companies since they<br />
became the holding<br />
company’s subsidiaries<br />
98
Consolidated Auditors’ Report<br />
To the Board of Directors of <strong>Glenmark</strong> Pharmaceuticals Limited on the Consolidated Financial Statements of<br />
GLENMARK PHARMACEUTICALS LIMITED AND ITS SUBSIDIARIES<br />
We have audited the attached consolidated Balance Sheet of <strong>Glenmark</strong> Pharmaceuticals Limited and its subsidiaries (the<br />
Group), as at March 31, 2004, and also the consolidated Profit and Loss Account and the consolidated Cash Flow<br />
Statement for the year ended on that date annexed thereto. These consolidated financial statements are the responsibility<br />
of the <strong>Glenmark</strong> Pharmaceuticals Limited’s management and have been prepared by the management on the basis of<br />
separate financial statements and other financial information regarding components. Our responsibility is to express an<br />
opinion on these consolidated financial statements based on our audit.<br />
We conducted our audit in accordance with the auditing standards generally accepted in India. Those Standards require<br />
that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of<br />
material mis-statement. An audit includes examining, on a test basis, evidence supporting the amounts and disclosures in<br />
the financial statements. An audit also includes assessing the accounting principles used and significant estimates made<br />
by management, as well as evaluating the overall financial statement presentation. We believe that our audit provides a<br />
reasonable basis for our opinion.<br />
We did not audit the financial statements of subsidiaries <strong>Glenmark</strong> Exports Limited, G M Pharma Limited, <strong>Glenmark</strong> Impex,<br />
Russia, <strong>Glenmark</strong> Farmaceutica Ltda, Brazil, <strong>Glenmark</strong> Phillipines Inc, Phillipines and <strong>Glenmark</strong> Pharmaceuticals Inc, USA<br />
whose financial statements reflect the Group’s share of total assets of Rs.405,498 (‘000’s) as at March 31, 2004 and the<br />
Group’s share of total revenues of Rs.149,009 (‘000’s) and net cash inflows amounting to Rs.2,451 (‘000’s) for the year<br />
ended on that date as considered in the consolidated financial statements. These financial statements and other<br />
information of the subsidiaries and joint ventures have been audited by other auditors whose reports have been furnished<br />
to us, and our opinion, in so far as it relates to the amounts included in respect of these subsidiaries is based solely on the<br />
reports of other auditors.<br />
We report that the consolidated financial statements have been prepared by the <strong>Glenmark</strong> Pharmaceuticals Limited’s<br />
management in accordance with the requirements of Accounting Standard 21, Consolidated Financial Statements issued<br />
by the Institute of Chartered Accountants of India.<br />
Based on our audit and on consideration of the reports of other auditors on separate financial statements and on the other<br />
financial information of the components, in our opinion and to the best of our information and according to the<br />
explanations given to us, the attached consolidated financial statements give a true and fair view in conformity with the<br />
accounting principles generally accepted in India:<br />
(a) in the case of the consolidated Balance Sheet, of the state of affairs of <strong>Glenmark</strong> Pharmaceuticals Limited Group as at<br />
March 31, 2004;<br />
(b) in the case of the consolidated Profit and Loss Account, of the profit for the year ended on that date; and<br />
(c) in the case of the consolidated Cash Flow Statement, of the cash flows for the year ended on that date.<br />
For and on behalf of Price Waterhouse<br />
Mumbai, July 5, 2004<br />
Partha Ghosh<br />
Partner<br />
Membership Number – F 055913<br />
Chartered Accountants<br />
99
Consolidated Balance Sheet<br />
Rs. In ('000s)<br />
As at 31st March, Schedules 2004 2003<br />
SOURCES OF FUNDS<br />
Shareholders' Funds<br />
a) Share capital 1 218,546 201,811<br />
b) Reserves and surplus 2 2,125,091 1,331,038<br />
2,343,637 1,532,849<br />
Minority Interest - Share Capital 4,365 –<br />
Loan Funds<br />
a) Secured loans 3 1,029,429 1,193,564<br />
b) Unsecured loans 4 119,314 53,864<br />
1,148,743 1,247,428<br />
Deferred Tax Liability 5 293,181 269,856<br />
Total 3,789,926 3,050,133<br />
APPLICATION OF FUNDS<br />
Fixed Assets 6<br />
a) Gross Block 1,838,830 1,247,428<br />
b) Less : Depreciation 349,176 243,613<br />
c) Net Block 1,489,654 1,003,815<br />
d) Capital Work-in-progress 251,853 232,471<br />
1,741,507 1,236,286<br />
Investments 7 147,492 146,314<br />
Deferred Tax Asset 8 28,153 15,383<br />
Current Assets, Loans and Advances<br />
a) Inventories 9 821,601 444,371<br />
b) Sundry debtors 10 1,304,813 1,232,627<br />
c) Cash and bank balances 11 81,255 45,699<br />
d) Loans and advances 12 402,470 512,501<br />
2,610,139 2,235,198<br />
Less : Current Liabilities and Provisions<br />
a) Current liabilities 13 727,726 607,749<br />
b) Provisions 14 39,702 11,647<br />
767428 619,396<br />
Net Current Assets 1,842,711 1,615,802<br />
Miscellaneous Expenditure 15 30,063 36,348<br />
(to the extent not written off or adjusted)<br />
Total 3,789,926 3,050,133<br />
Notes to the Financial Statements 22<br />
Schedules referred to above and notes attached there to form an integral part of the Balance Sheet.<br />
This is the Balance Sheet referred to in our report of even date.<br />
For and on behalf of<br />
Price Waterhouse<br />
Chartered Accountants<br />
Membership No. F 055913<br />
For and on behalf ofthe Board of Directors.<br />
Partha Ghosh M. J. Mendonza Glenn Saldanha Rajesh Desai<br />
Partner Vice President - Legal & Managing Director & CEO Director - Finance<br />
Mumbai, July 5, 2004<br />
Company Secretary<br />
100
Consolidated Profit and Loss Account<br />
Rs. In ('000s)<br />
For the year ended 31st March, Schedules 2004 2003<br />
INCOME<br />
Sales & Operating Income 16 3,806,606 3,703,350<br />
Other income 17 34,662 30,572<br />
3,841,268 3,733,922<br />
EXPENDITURE<br />
Cost of sales 18 1,793,191 1,917,292<br />
Selling and operating expenses 19 1,074,266 1,010,036<br />
Depreciation 6 110,930 106,567<br />
Interest 20 100,578 123,430<br />
Research and development expenses 21 248,073 147,257<br />
3,327,038 3,304,582<br />
Profit Before Tax 514,230 429,340<br />
Provision for taxation<br />
– Current Year [includes wealth tax provision Rs. 300 (Prev. Year–Rs. 300)] 90,334 35,800<br />
– Earlier Years – 18,429<br />
– Deferred tax 10,555 49,600<br />
Net Profit After Tax before minority interest 413,341 325,511<br />
Share of loss transfer to Minority 265 –<br />
Net Profit After Tax & Minority Interest 413,606 325,511<br />
Balance Profit brought forward 243,262 42,836<br />
Net Profit Available for Appropriation 656,868 368,347<br />
Interim Dividend paid on pref shares 10,500 10,500<br />
Tax on dividend paid on pref shares 1,345 –<br />
Interim Dividend on Equity Shares 77,020 66,114<br />
Tax on interim dividend paid on equity shares 9,868 8,471<br />
Transfer to Debenture Redemption Reserve 253 –<br />
Transfer to General Reserve 60,000 40,000<br />
Balance Carried to Balance Sheet 497,882 243,262<br />
Earnings per share (Rs.) (Refer Note 3 of Schedule 22)<br />
Basic 7.29 6.20<br />
Diluted 7.25 5.56<br />
Face Value of Shares 2.00 2.00<br />
Notes to the Financial Statements 22<br />
Schedules referred to above and notes attached there to form an integral part of the Profit and Loss Account.<br />
This is the Profit and Loss Account referred to in our report of even date.<br />
For and on behalf of<br />
Price Waterhouse<br />
Chartered Accountants<br />
Membership No. F 055913<br />
For and on behalf ofthe Board of Directors.<br />
Partha Ghosh M. J. Mendonza Glenn Saldanha Rajesh Desai<br />
Partner Vice President - Legal & Managing Director & CEO Director - Finance<br />
Mumbai, July 5, 2004<br />
Company Secretary<br />
101
Consolidated Cash Flow Statement<br />
Rs. In ('000s)<br />
Year ended 31st March, 2004 2003<br />
A. Cash flow from operating activities:<br />
Net Profit before tax 514,230 429,340<br />
Adjustments for:<br />
Depreciation 110,930 106,567<br />
Interest Expense 98,677 121,859<br />
Interest Expense - Finance lease 1,901 1,571<br />
Interest Income (2,422) (1,433)<br />
Income from Investment - Dividends (1,245) (20)<br />
(Profit)/Loss on Fixed Assets sold 720 (1,494)<br />
Deferred revenue expenditure written off 6,285 30,288<br />
Provision for Doubtful Advances – 4,000<br />
Provision for Bad & Doubtful Debts 11,800 16,000<br />
Provision for Gratuity & Leave Encashment 500 1,500<br />
Unrealised foreign exchange (gain) /loss (9,102) 965<br />
Employee stock option plan (4,593) 1,808<br />
Operating profit before working capital changes 727,681 710,951<br />
Adjustments for changes in working capital :<br />
– (increase)/decrease in Sundry Debtors (75,705) (424,482)<br />
– (increase)/decrease in Other Receivables (125,596) (104,144)<br />
– (increase)/decrease in Inventories (377,230) 38,719<br />
– increase/(decrease) in Trade and Other Payables 146,856 26,151<br />
Cash generated from operations 296,006 247,195<br />
– Taxes (Paid) / Received (Net of Tax deducted at source) (62,379) (49,075)<br />
Net cash from operating activities 233,627 198,120<br />
B. Cash flow from Investing activities:<br />
Purchase of fixed assets (637,192) (201,456)<br />
Capital Work in Progress (19,382) (166,412)<br />
Proceeds from Sale of fixed assets 250,000 125,293<br />
Proceeds from Sale of Investments – (135,775)<br />
Purchase of investments (1,178) –<br />
Finance Lease Rent payment against principal amount (6,974) (4,724)<br />
Interest Received 3,701 1,765<br />
Dividend Received 1,245 20<br />
Net cash used in investing activities (409,780) (381,289)<br />
102
Consolidated Cash Flow Statement (Contd.)<br />
Rs. In ('000s)<br />
Year ended 31st March, 2004 2003<br />
C. Cash flow from financing activities:<br />
Proceeds from fresh issue of<br />
Share Capital (including Share Premium ) 508,185 (6,139)<br />
Minority interest 4,365 –<br />
Proceeds from long term borrowings (340,079) 359,191<br />
Receipt /(Repayment) of short term borrowings 65,450 (44,058)<br />
Proceeds from Cash Credits (NET) 182,918 16,671<br />
Finance Lease Rent (Interest Part only) (1,901) (1,571)<br />
Interest Paid (115,121) (127,651)<br />
Dividend Paid (76,224) (69,103)<br />
Dividend Tax Paid (8,471) –<br />
Net cash used in financing activities 219,122 127,340<br />
Net Increase/(Decrease) in Cash & Cash Equivalents 42,969 (55,829)<br />
Cash and cash equivalents as at 31.03.2003 45,699 101,812<br />
Consolidation Adjustment (7,413) (284)<br />
Cash and cash equivalents as at 31.03.2004 81,255 45,699<br />
Cash and cash equivalents comprise<br />
Cash 897 619<br />
Deposit 4,319 4,160<br />
Balance with Scheduled Banks 76,039 40,920<br />
81,255 45,699<br />
Notes :<br />
1 The Cash Flow Statement has been prepared under the "Indirect Method" as<br />
set out in Accounting Standard - 3 on Cash Flow Statements issued by the<br />
Institute of Chartered Accountants of India.<br />
2 Cash and cash equivalents includes Rs. 2,364 which are not available<br />
for use by the Company (Refer schedule 13 to the financial statements).<br />
3 Figures in bracket indicate Cash outgo.<br />
This is the Cash Flow Statement referred to in our report of even date<br />
For and on behalf of<br />
Price Waterhouse<br />
Chartered Accountants<br />
Membership No. F 055913<br />
For and on behalf ofthe Board of Directors.<br />
Partha Ghosh M. J. Mendonza Glenn Saldanha Rajesh Desai<br />
Partner Vice President - Legal & Managing Director & CEO Director - Finance<br />
Mumbai, July 5, 2004<br />
Company Secretary<br />
103
Schedules forming part of the Consolidated Balance Sheet<br />
Rs. In ('000s)<br />
As at 31st March, 2004 2003<br />
1 Share Capital<br />
Authorised<br />
125,000,000 (2003 – 125,000,000) Equity shares of Rs 2/- each 250,000 250,000<br />
4,000,000 (2003 – 4,000,000) Cumulative redeemable<br />
non convertible preference shares of Rs 100/- each 400,000 400,000<br />
Unclassified capital 100,000 100,000<br />
Issued, Subscribed and Paid-up<br />
59,245,570 (2003 – 50,860,000) Equity shares of Rs 2/- each 118,491 101,720<br />
Less: Calls in arrears 3 7<br />
118,488 101,713<br />
10.5%, 1,000,000 (2003 – 1,000,000) redeemable cumulative non-convertible<br />
preference shares of Rs. 100/- each (Redeemed on April 1, 2004 at par) 100,000 100,000<br />
Equity Share Warrants<br />
290,000 (2003 -- 490,000) Equity share warrants of Rs. 0.20/- each 58 98<br />
Total 218,546 201,811<br />
Notes :<br />
1. At the Annual general meeting held on September 26, 2003, the shareholders approved the sub-division of Equity share<br />
from face value of Rs. 10 each to Rs. 2 each. Accordingly previous year’s figures have been recast.<br />
2. Of the above equity shares, 19875,000 shares of Rs 2 each are allotted as fully paid-up Bonus Shares by capitalisation<br />
of reserves.<br />
3. In terms of Employee Stock Option Plan approved by the members, 290,000 (2003 – 490,000) convertible warrants<br />
have been allotted to <strong>Glenmark</strong> Pharmaceuticals Limited Employees Welfare Trust. Each warrant carries an option and<br />
is convertible into one equity share of Rs 2 each. During the year, 200,000 (2003 – 225,000) warrants were converted<br />
into Equity Shares at an exercise price of Rs. 14.34 per share.<br />
4. During the Year Company formulated a new Employees Stock Option Scheme viz. ESOS’ 2003 pursuant to which<br />
7,00,725 options of Rs. 2. each were granted at average market price. On exercise of the options so granted the Paidup<br />
equity share capital of the company will increase by a like number of shares.<br />
5. 5% Convertible Debentures aggregating to Rs. 5,000 Lakhs issued to CDC Financial Services (Mauritius) Ltd. and South<br />
Asia Regional Fund were converted into 8,185,570 fully paid up equity shares of Rs. 2 each at a premium of Rs. 59.08<br />
per share as per the agreement based on predetermined price earning ratio on the profits for the year ended March 31,<br />
2003 at the meeting of board of directors held on September 29,2003.<br />
6. As of March 31, 2004, the Trust has allotted 85,000 (2003–320,000) warrants to the employees of the Company, which<br />
are outstanding. The options have been granted at an exercise price of Rs. 14.34 per share in respect of 85,000 shares.<br />
104
Schedules forming part of the Consolidated Balance Sheet<br />
Rs. In ('000s)<br />
As at 31st March, note 2004 2003<br />
2 Reserves and Surplus<br />
Share premium account<br />
Balance at the beginning of the year 500,518 507,064<br />
Addition on issue of shares 491,502 6,257<br />
Less: Calls in arrears 52 124<br />
Less : Issue cost – 12,680<br />
Closing balance 991,968 500,517<br />
General reserve<br />
Balance at the beginning of the year 550,773 510,773<br />
Less : Loss pertains to Minority Interest 265 –<br />
Add : Transferred during the year 60,000 40,000<br />
Closing balance 610,508 550,773<br />
Debenture Redemption Reserve<br />
Balance at the beginning of the year 29,664 29,664<br />
Addition during the year 253 –<br />
Closing balance 29,917 29,664<br />
Capital Reserve<br />
Balance at the beginning of the year 1,000 1,000<br />
Addition during the year – –<br />
Closing balance 1,000 1,000<br />
Currency fluctuation reserve on Consolidation (7,697) (284)<br />
Employee stock option<br />
Employee stock options outstanding 8,219 8,763<br />
Add: Grant of Options at discount – 370<br />
Less: Conversion of Option 5,283 231<br />
Less: Cancellation of Option 884 683<br />
A 2,052 8,219<br />
Deferred employee stock compensation 2,113 4,465<br />
Add: Grant of Options at discount – 370<br />
Less: Amortisation of ESOP expense. 1,436 2,039<br />
Less: Cancellation of Option 138 683<br />
B 539 2,113<br />
Net Employee Stock option A – B 1,513 6,106<br />
Profit and loss account<br />
Balance 497,882 243,262<br />
Total 2,125,091 1,331,038<br />
3 Secured Loans<br />
Term loan 1 350,000 200,000<br />
Convertible Debentures 2 – 500,000<br />
Non Convertible Debentures 3 140,000 140,000<br />
Working capital facilities 4 528,414 345,496<br />
Other loans 5 11,015 8,068<br />
Total 1,029,429 1,193,564<br />
Notes :<br />
1. Term loan is secured by the equitable mortgage of assets as a security. Creation of Charge is under process.<br />
2. 5% Convertible Debentures aggregating to Rs. 5,000 Lakhs issued to CDC Financial Services (Mauritius) Ltd. and<br />
105
Schedules forming part of the Consolidated Balance Sheet<br />
3 Secured Loans (contd.)<br />
South Asia Regional Fund were converted into 8,185,570 fully paid up equity shares of Rs. 2 each at a premium of<br />
Rs. 59.08 per share as per the agreement based on predetermined price earning ratio on the profits for the year ended<br />
March 31, 2003 at the meeting of board of directors held on September 29, 2003.<br />
3. Non convertible debenture ‘NCD’ includes:<br />
a) 10.05% NCD – A series aggregating Rs. Nil (Previous year Rs 50 million), allotted on September 7, 2001 with a put<br />
and call option after twelve months and redeemable after three years.<br />
b) 10.35% NCD – B series aggregating Rs Nil (Previous year Rs. 90 million, allotted on September 7, 2001 with a call<br />
option after twelve months and redeemable after three years.<br />
All the above debentures are secured by way of a mortgage favouring the debenture trustees on the land and buildings,<br />
plant and machinery and other fixed assets and hypothecation of all movable assets (except raw materials and book<br />
debts, both present and future), situated at the research and development center at Mahape, Navi Mumbai.<br />
4. Working capital loan from banks are secured by hypothecation of stocks of raw materials, packing materials, finished<br />
goods, work in progress, receivables etc. They are additionally secured by way of personal guarantees of some of the<br />
directors and equitable mortgage on fixed assets at the manufacturing facility at Nasik and research and development<br />
centre at Sinnar, Nasik.<br />
5. Other loans are secured by way of hypothecation of vehicles.<br />
4 Unsecured Loans<br />
Short term loan 105,117 –<br />
Security deposit 3,752 43,375<br />
Deferred Sales Tax Loan 10,445 10,445<br />
Other Deposits – 44<br />
Total 119,314 53,864<br />
Notes :<br />
1. The Company has availed of an interest free sales tax deferral loan under Part I of the 1983 and 1988 Package<br />
Schemes of the Government of Maharashtra, repayable after twelve years in six half-yearly installments.<br />
5 Deferred Tax Liability [refer note (2) (X) of Schedule 22]<br />
Liabilities<br />
Depreciation 291,291 263,116<br />
Others 1,890 6,740<br />
Total 293,181 269,856<br />
106
Schedules forming part of the Balance Sheet<br />
as at March 31, 2004 & Profit & Loss account for the year ended on that date.<br />
6 Fixed Assets [refer note (2) (ii) and (6) of Schedule 22]<br />
(Rs. In '000s)<br />
GROSS BLOCK DEPRECIATION NET BLOCK<br />
As on Additions Sales/Disposals As on As on For the year Sales/ As on As on As on<br />
1st Apr, during during 31st Mar, 1st Apr, Disposals 31st Mar, 31st Mar, 31st Mar,<br />
2003 the year the year 2004 2003 of Assets 2004 2004 2003<br />
Freehold Land 5,710 – – 5,710 – – – – 5,710 5,710<br />
Leasehold Land 21,125 35,914 – 57,039 775 505 – 1,280 55,759 20,350<br />
Factory Buildings 87,030 72,257 – 159,287 18,935 3,770 – 22,705 136,582 68,095<br />
Other Buildings & Premises 222,411 44,999 – 267,410 12,213 3,832 – 16,045 251,365 210,198<br />
Plant and Machinery 55,627 140,688 9,196 187,119 10,595 11,353 706 21,242 165,877 45,032<br />
Furniture and Fixtures 101,801 28,965 154 130,612 23,269 12,398 7 35,660 94,952 78,532<br />
Equipments 373,647 179,240 8,281 544,606 71,271 39,229 203 110,297 434,309 302,376<br />
Vehicles 30,008 8,503 6,021 32,490 13,203 4,836 4,451 13,588 18,902 16,805<br />
Brands (Note-5) 350,069 104,488 – 454,557 93,352 35,007 – 128,359 326,198 256,717<br />
Total 1,247,428 615,054 23,652 1,838,830 243,613 110,930 5,367 349,176 1,489,654 1,003,815<br />
Previous Year 1,429,572 232,100 414,244 1,247,428 177,491 106,567 40,445 243,613 1,003,815 1,252,081<br />
Capital Work-in-process including Capital advances. 251,853 232,471<br />
Notes :<br />
1. Equipment and vehicles include assets aggregating Rs. 19,308 (2003 – Rs. 19,559) {net book value as at March 31, 2004 – Rs. 14,338 (2003 – Rs. 17,023)] and Rs. 5,536<br />
(2003 – Rs. 5,536) [net book value as at March 31, 2004 – Rs. 3,708 (2003 – Rs. 4,631)], respectively, which have been acquired on finance lease.<br />
2. Additions to assets include Rs. 4,488 being borrowing costs.<br />
3. Estimated amount of contracts remaining to be executed on capital account, net of advances, not provided for as at March 31, 2004 aggregate Rs. 59,039 (2003 –<br />
Rs. 14,566)<br />
4. Capital Work in progress includes : 2004 2003<br />
Cost of Ankleshwar Plant plus addition thereto 68,157 160,283<br />
Capitalisation of Interest & Relevant costs of Ankleshwar Plant – 16,653<br />
Cost of Goa Plant plus addition there to 57,145 –<br />
Capital Advances 109,854 49,667<br />
Other work-in-processes 9,811 5,868<br />
Brazil CWIP 6,282 –<br />
Philippines CWIP 605 –<br />
5. Additions for <strong>Glenmark</strong> Pharmaceuticals Inc, USA includes Product development process, patents and Trademarks. However pending completion of the development<br />
process and registration of Products and Trademarks, no amortisation of Intangible assets has been provided for during the year.<br />
7 Investments {refer Note (2)(iv) of Schedule 22)<br />
Long Term Investments<br />
Quoted - traded<br />
Equity shares<br />
9,000 (2003 – 9,000) Bank of India of Rs. 10 each [Market Value Rs. 530 (2003 – Rs. 341)] 405 405<br />
1,718 (2003 – 1,000) IDBI Bank Limited of Rs. 10 each [Market Value Rs. 85 (2003 – Rs. 23)] 34 18<br />
439 423<br />
Unquoted – non trade<br />
National Savings Certificate – Sixth Issue 48 48<br />
1 (2003) – 1) Time share of Dalmia Resorts Limited 20 20<br />
1 (2003 – 1) equity share of Esquados 340,000 of <strong>Glenmark</strong><br />
Pharmaceutical Limitada, Lisbon (Portugal) 48 48<br />
1,93,665 shares of Bharuch Eco-Aqua Infrastructure Limited of Rs. 10<br />
each, fully paid up (Previous year Rs. 4 per share was paid up). 1,937 775<br />
100,000 12% cumulative preference shares of Rs 100 each fully<br />
paid up of Chery Laboratories (P) Limited 10,000 10,000<br />
1350,000 7% cumulative preference shares of Rs 100 each fully paid up<br />
of <strong>Glenmark</strong> Laboratories Limited 135,000 135,000<br />
147,053 145,891<br />
Total 147,492 146,314<br />
107
Schedules forming part of the Consolidated Balance Sheet<br />
Rs. In ('000s)<br />
As at 31st March, 2004 2003<br />
8 Deferred Tax Asset [refer Note (2)(X) of Schedule 22)]<br />
Assets<br />
Provision for Bad Debts and Doubtful Advances 23,102 13,340<br />
Others 525 2,043<br />
Unabsorbed losses and Depreciation 4,526 –<br />
Total 28,153 15,383<br />
9 Inventories [refer Note (2)(v) of Schedule 22)]<br />
(As certified by the management)<br />
Raw & Packing materials 310,098 150,780<br />
Work-in-process 223,462 51,850<br />
Stores and Spares 2,574 3,419<br />
Finished goods 285,467 238,322<br />
Total 821,601 444,371<br />
10 Sundry Debtors<br />
Outstanding for more than six months<br />
Secured, considered good – –<br />
Unsecured, considered good 147,936 277,909<br />
Unsecured, considered doubtful 43,061 31,260<br />
190,997 309,169<br />
Less: Provision for doubtful debts 43,061 31,260<br />
147,936 277,909<br />
Other debts-<br />
Secured, considered good – –<br />
Unsecured, considered good 1,156,877 954,718<br />
1,156,877 954,718<br />
Total 1,304,813 1,232,627<br />
11 Cash and Bank Balances<br />
Cash in hand 897 619<br />
Balances with banks<br />
– Current accounts 31,744 12,834<br />
– Margin Money Account 42,092 17,283<br />
– EEFC Account 2,203 10,803<br />
– Deposit accounts 4,319 4,160<br />
Total 81,255 45,699<br />
The balances in the margin money accounts are given as security against guarantees issued by banks on behalf of the<br />
Company.<br />
108
Schedules forming part of the Consolidated Balance Sheet<br />
Rs. In ('000s)<br />
As at 31st March, 2004 2003<br />
12 Loans and Advances (unsecured, considered good)<br />
<strong>Glenmark</strong> Laboratories Limited * 17,985 267,988<br />
Advances to subsidiaries<br />
<strong>Glenmark</strong> Pharmaceuticals Inc., U.S.A. [Maximum during the year – 7,944<br />
Rs. 7,944 (Previous Year – Rs. 7,944)]<br />
Advance to Vendors 130,275 85,161<br />
Advances recoverable in cash or kind or for value to be received 174,700 96,985<br />
Balance with Excise & Sales tax Authorities 19,620 13,535<br />
Deposits 59,890 40,888<br />
Total 402,470 512,501<br />
* Last year being an associate company<br />
[maximum amount outstanding during the year Rs. 267,988]<br />
13 Current Liabilities<br />
Acceptances 92,842 62,618<br />
Sundry creditors - Small scale industrial undertakings 20,867 16,474<br />
- Others 372,691 310,969<br />
Investor Education and Protection Fund shall be credited by<br />
– Unclaimed Dividend 2,364 1,974<br />
[There are no amounts due and outstanding to be credited to<br />
Investor Education and Protection Fund.]<br />
Advances from Customers 3,049 1,206<br />
Other Liabilities 139,438 115,237<br />
Interest accrued but not due 8,242 24,686<br />
Interim Dividend on Equity Shares 77,020 66,114<br />
Tax on Interim Dividend 9,868 8,471<br />
Tax on Preference Dividend 1,345 –<br />
Total 727,726 607,749<br />
14 Provisions<br />
Wealth tax 300 300<br />
Income-tax (net of advance tax) [Refer Note (2) (X) of Schedule 22] 37,402 9,847<br />
Provision for Gratuity and leave encashment 2,000 1,500<br />
Total 39,702 11,647<br />
15 Miscellaneous Expenditure [(refer note (2) (vii) of Schedule 22]<br />
(to the extent not written off or adjusted)<br />
Implementation Expenses of ERP system 1,147 5,737<br />
Product launch, investigation and registration expenses 5,272 18,339<br />
Pre-operative / Preliminary expenses 23,644 12,272<br />
Total 30,063 36,348<br />
109
Schedules to the Consolidated Profit & Loss Account<br />
Rs. In ('000s)<br />
For the year ended 31st March, 2004 2003<br />
16 Sales [(refer note 2 (viii) of Schedule 22]<br />
Sale of goods* 3,796,569 3,693,934<br />
Income from services 10,037 9,416<br />
Total 3,806,606 3,703,350<br />
* includes sales tax and excise duty aggregating Rs 255,771 (2003-Rs 251,600) and Rs 340,561 (2003-Rs 307,802)<br />
respectively.<br />
17 Other Income<br />
Lease Rent [tax deducted at source Rs.403 (Prev year -- Rs. 302)] 1,920 1,440<br />
Interest Income [tax deducted at source Rs. 400 (Prev year -- Rs. 427)] 2,422 2,019<br />
Dividend received 1,245 20<br />
Exchange gain 4,642 4,109<br />
Export Incentive 7,613 8,661<br />
Profit on sale of fixed assets 128 1,631<br />
Miscellaneous income 16,692 12,116<br />
Profit on sale of Subsidiary – 576<br />
Total 34,662 30,572<br />
18 Cost of Sales<br />
Salary, wages and allowances 38,320 38,033<br />
Labour charges 82,227 63,620<br />
Consumption of raw & packing materials 966,088 927,049<br />
Purchase of Trading goods 286,013 306,895<br />
Excise duty paid 308,398 327,169<br />
Sales tax 255,771 251,600<br />
Power, fuel and water charges 31,365 21,878<br />
Consumable stores 24,583 39,224<br />
Repairs and maintenance - plant and machinery 17,066 13,183<br />
Rent, rates and taxes 1,578 1,560<br />
Other manufacturing expenses 540 6,123<br />
(Increase)/decrease in inventory (218,758) (79,042)<br />
Total 1,793,191 1,917,292<br />
110
Schedules to the Consolidated Profit & Loss Account<br />
Rs. In ('000s)<br />
For the year ended 31st March, 2004 2003<br />
19 Selling and Operating Expenses<br />
Salary and allowances 256,391 214,908<br />
Staff welfare 7,801 7,780<br />
Directors' salaries and allowances 44,762 27,207<br />
Incentive and commission 40,978 31,923<br />
Sales promotion expenses 184,386 209,137<br />
Export Commission 7,935 11,107<br />
Commission on sales 4,154 34,678<br />
Travelling expenses 151,626 136,969<br />
Freight outward 71,847 85,147<br />
Telephone expenses 19,431 17,888<br />
Rates and taxes 14,040 8,719<br />
Provision for doubtful debts 11,800 16,000<br />
Provision for doubtful advances – 4,000<br />
Insurance 9,173 7,437<br />
Electricity charges 7,258 7,805<br />
Rent 15,885 16,052<br />
Repairs & Maintenance 39,135 26,180<br />
Auditors remuneration<br />
Audit fees 1,322 750<br />
Other matters 1,020 620<br />
Out of pocket expenses 30 5<br />
Loss on sale of assets 848 137<br />
Other operating expenses 184,444 145,587<br />
Total 1,074,266 1,010,036<br />
20 Interest Expense<br />
On loans from banks 35,777 41,798<br />
Other interest 64,801 81,632<br />
Total 100,578 123,430<br />
21 Research and Development Expenses [refer Note (2) (ix) of Schedule 22]<br />
Salary and other allowances 71,328 41,406<br />
Staff welfare expenses 4,633 2,527<br />
Directors Remuneration 2,517 1,368<br />
Consumable & Chemicals 80,560 52,971<br />
Electricity charges 12,777 9,542<br />
Repairs and maintenance 12,192 7,709<br />
Insurance premium 283 377<br />
Other expenses 63,783 31,357<br />
Total 248,073 147,257<br />
111
Notes to the Consolidated Accounts<br />
For the year ended 31st March, 2004<br />
22 Notes to the Financial Statement<br />
1) Principles of Consolidation<br />
(a) The consolidated financial statements of the Group have been prepared in accordance with Accounting Standard<br />
(AS-21) - "Consolidated Financial Statements" issued by the Institute of Chartered Accountants of India.<br />
(b)<br />
<strong>Glenmark</strong> Pharmaceuticals, the holding company, had controlling interest in the following entities as at 31st March,<br />
2004:<br />
Name of the Subsidiary Country of Incorporation Percentage of ownership<br />
<strong>Glenmark</strong> Exports Limited India 100%<br />
GM Pharma Limited India 100%<br />
<strong>Glenmark</strong> Impex, Russia Russia 100%<br />
<strong>Glenmark</strong> Farmaceutica Ltda, Brazil Brazil 100%<br />
<strong>Glenmark</strong> Philippines Inc., Philippines Philippines 100%<br />
<strong>Glenmark</strong> Pharmaceuticals Inc., USA USA 96%<br />
(c)<br />
Assets and liabilities of foreign subsidiaries are translated into Indian rupees at the rate of exchange prevailing as<br />
at the Balance Sheet date. Revenues and expenses are translated into India rupees at yearly average exchange<br />
rates prevailing during the year and the resulting net translation adjustment has been adjusted to Reserves.<br />
2) Significant Accounting Policies<br />
i) Basis of Accounting<br />
The consolidated financial statements are prepared under the historical cost convention, on the accrual basis of<br />
accounting, in conformity with accounting principles generally accepted in India. These consolidated financial<br />
statements have been prepared to meet the requirements of clause 32 of the listing agreement with the Stock<br />
Exchanges.<br />
ii)<br />
Fixed assets and depreciation<br />
Fixed assets are stated at cost less accumulated depreciation. The Company capitalises all costs relating to the<br />
acquisition and installation of fixed assets. Expenditure of revenue nature, incurred in setting up of new projects, is<br />
capitalised as an indirect cost towards construction of the fixed assets. Exchange differences relating to the<br />
acquisition of fixed assets are adjusted in the cost of the assets.<br />
Depreciation is provided using the straight line method, pro-rata to the period of use of assets, based on the useful<br />
lives of fixed assets as estimated by management, or at the rates specified in Schedule XIV of the Companies Act,<br />
1956, whichever is higher.<br />
Fixed assets having aggregate cost of Rs 5,000 or less are depreciated fully in the year of acquisition.<br />
The group has estimated the useful life of its assets as follows:<br />
Category<br />
Estimated useful life<br />
(in years)<br />
Plant and machinery 8 - 20<br />
Vehicles 5 - 6<br />
Equipments and air conditioners 4 - 20<br />
Furniture and fixtures 5 - 10<br />
Brands 5 - 10<br />
Leasehold land is amortised over the period of lease.<br />
iii)<br />
Foreign currency transactions<br />
Foreign currency transactions during the year are recorded at the rates of exchange prevailing on the date of the<br />
transaction. Foreign currency assets and liabilities are translated into rupees at the exchange rates prevailing on<br />
the date of the balance sheet.<br />
All exchange differences are dealt with in the statement of profit and loss, except those relating to the acquisition<br />
of fixed assets, which are adjusted in the cost of the respective fixed assets.<br />
112
Notes to the Consolidated Accounts<br />
For the year ended 31st March, 2004<br />
22 Notes to the Financial Statement (Contd.)<br />
iv) Investments<br />
Long term investments are stated at cost. Provision, where necessary, is made to recognise a decline, other than<br />
temporary, in the value of the investments.<br />
v) Inventories<br />
Inventories of raw materials, packing materials, work-in-process and finished goods are valued at cost or net<br />
realisable value, whichever is lower. Cost of raw materials and packing materials is ascertained on a first-in-first<br />
out basis. Cost of work-in-process and finished goods includes the cost of materials consumed, labour and<br />
manufacturing overheads. Excise and customs duty accrued on production or import of goods, as applicable, is<br />
included in the valuation of inventories.<br />
vi)<br />
Employee Benefits<br />
Retirement benefits to employees comprise payments towards gratuity, superannuation and provident fund under<br />
the schemes of the Company and encashment of leave. Annual contributions to the superannuation and provident<br />
funds are charged to the statement of profit and loss.<br />
The employee leave encashment and gratuity schemes are administered through the Life Insurance Corporation of<br />
India. Annual contributions as determined by the LIC are charged to the statement of profit and loss.<br />
In respect of stock options granted to employees under the Company’s Employee Stock Option Plan (‘ESOP’), the<br />
excess of the market price of the share at the date of grant of the option over its exercise price is treated as a<br />
form of employee compensation in the financial statements of the company. This amount is amortised on a<br />
straight-line basis over the vesting period. The unamortised portion is carried forward as deferred employee<br />
compensation.<br />
vii)<br />
Miscellaneous expenditure<br />
Product launch expenditure<br />
Earlier years' expenditure on launch of new products and their sales promotion is being amortised over a period of<br />
three years.<br />
Implementation expenses of Enterprise Resource Planning system.<br />
Earlier year's expenditure incurred on payments for infrastructure facilities and expenditure incurred on user<br />
license fees for an Enterprise Resource Planning system is being amortised over a period of thirty-six months.<br />
Preliminary expenses<br />
Preliminary expenses of <strong>Glenmark</strong> Pharmaceuticals Inc, USA being expenses incurred prior to the incorporation of<br />
the company have been amortised over a period of 5 years.<br />
viii) Revenue recognition<br />
The group recognises revenues on despatch of goods to customers. Revenues from services are recognised on<br />
completion of such services. Revenues are recorded at invoice value, inclusive of excise duty and sales-tax, but<br />
net of returns and trade discounts.<br />
ix)<br />
Research and development<br />
Capital expenditure on research and development ('R&D') is capitalised as fixed assets. Other expenditure on<br />
R&D is expensed as incurred.<br />
x) Income-tax<br />
Provision for current income-taxes is made on the assessable income at the tax rate applicable to the relevant<br />
assessment year. Deferred income taxes are recognised for the future tax consequences attributable to timing<br />
differences between the financial statement determination of income and their recognition for tax purposes. The<br />
effect on deferred tax assets and liabilities because of a change in tax rates is recognised in statement of profit<br />
and loss using the tax rates and tax laws that have been enacted or substantively enacted by the balance sheet<br />
date.<br />
Deferred tax assets are recognised and carried forward only to the extent that there is a reasonable certainty that<br />
sufficient future taxable income will be available against which such deferred tax assets can be realised.<br />
113
Notes to the Consolidated Accounts<br />
For the year ended 31st March, 2004<br />
Prior / Earlier year's income tax is charged to the Profit and Loss account on payment and the same is disclosed<br />
separately.<br />
xi)<br />
Leases<br />
Finance leases<br />
Assets acquired under finance lease are recognised as assets with corresponding liabilities in the balance sheet at<br />
the inception of the lease at amounts equal to the fair value of the leased asset or, if lower, at the present value of<br />
the minimum lease payments. These leased assets are depreciated in line with the Company’s policy on<br />
depreciation of fixed assets. The interest is allocated to periods during the lease term so as to produce a constant<br />
periodic rate of interest on the remaining balance of the liability for each period.<br />
Operating leases<br />
Lease payments for operating leases are recognised as expense on a straight-line basis over the lease term.<br />
Lease income from operating leases is recognised as income on a straight-line basis over the lease term. Initial<br />
direct costs are recognised immediately as an expense.<br />
3) Earnings per Share<br />
Basic earnings per share are calculated by dividing the net profit for the year attributable to equity shareholders (net<br />
profit for the year less dividends on preference shares) by the weighted average number of equity shares outstanding<br />
during the year.<br />
For the purpose of calculating diluted earnings per share, the net profit attributable to equity shareholders and the<br />
weighted average number of shares outstanding are adjusted for the effects of all dilutive potential equity shares from<br />
the exercise of options on unissued share capital. The number of equity shares is the aggregate of the weighted<br />
average number of equity shares and the weighted average number of equity shares, which would be issued on the<br />
conversion of all the dilutive potential equity shares into equity shares. Options on unissued equity share capital are<br />
deemed to have been converted into equity shares on the date when the options are exercised.<br />
The calculations of earnings per share (basic and diluted) are based on the earnings and number of shares as computed<br />
below.<br />
Rs. In ('000s)<br />
Reconciliation of earnings 31st March, 2004 31st March, 2003<br />
Profit after tax for the financial year 413,606 325,511<br />
Less:<br />
Preference dividends 10,500 10,500<br />
Dividend tax on preference shares 1,345 –<br />
Net profit attributable to equity shareholders for calculation of Basic EPS 401,761 315,011<br />
Add :<br />
Interest accrued on Convertible Debentures less tax theron – 15,333<br />
Net profit attributable to equity shareholders for calculation of Diluted EPS 401,761 330,344<br />
Reconciliation of number of shares Shares Shares<br />
(in ‘000) (in ‘000)<br />
Weighted average number of shares:<br />
For basic earnings per share 55,111 50,780<br />
Add:<br />
Deemed exercise of options on unissued equity share capital 274 215<br />
Conversion of Debentures – 8,515<br />
For diluted earnings per share 55,385 59,510<br />
Earnings per share (nominal value Rs 2 each) Rs Rs<br />
Basic 7.29 6.20<br />
Diluted 7.25 5.56<br />
114
Notes to the Consolidated Accounts<br />
For the year ended 31st March, 2004<br />
22 Notes to the Financial Statement (Contd.)<br />
4) Segment Information<br />
Business segments<br />
The group is primarily engaged in a single segment business of manufacturing and marketing of pharmaceuticals<br />
formulations and active ingredients and is managed as one entity, for its various activities and is governed by a similar<br />
set of risks and returns.<br />
Geographical segments<br />
The operations of the group are largely confined to India, with exports contributing to 17.12 percent of its annual sales.<br />
Hence, in the view of the management, the Indian and export markets represent geographical segments.<br />
Sales by market -- The following is the distribution of the Company's sale by geographical market<br />
Rs. In ('000s)<br />
Geographical segment 31st March, 2004 31st March, 2003<br />
India 3,154,903 3,023,242<br />
Other than India 651,703 313,123<br />
Total 3,806,606 3,336,365<br />
Assets and additions to fixed assets by geographical area – The following table shows the carrying amount of segment<br />
assets and additions to fixed assets by geographical area in which the assets are located:<br />
Rs. In ('000s)<br />
India Others* India Others*<br />
2004 2004 2003 2003<br />
Carrying amount of segment assets 3,633,914 583,192 3,123,522 229,048<br />
Additions to tangible assets 615,053 – 232,100 –<br />
* Others represent receivables from debtors located outside India including those related to deemed exports.<br />
5) Related Party Disclosures<br />
a) Related party relationships where transactions have taken place during the year<br />
Key management personnel (includes directors of the Company and its subsidiaries)<br />
Mrs BE Saldanha<br />
Mr Glenn Saldanha<br />
Mr Robin Pinto<br />
Mr. RV Desai<br />
Mr. AS Mohanty<br />
Mr. SA Paigankar (Resigned on 29.03.04)<br />
Mr. Jeffery Weiss (appointed director effective 01.03.03)<br />
Mr. Sangeet Mehrotra (appointed director effective 28.01.04)<br />
Mr. Sudip Majumdar (resigned on 31.07.03)<br />
Mr. Anil Muddanna (appointed director effective 01.08.03)<br />
115
Notes to the Consolidated Accounts<br />
For the year ended 31st March, 2004<br />
22 Notes to the Financial Statement (Contd.)<br />
b) Transactions with related parties during the year Rs. In ('000s)<br />
Associate enterprises<br />
31st March, 2004 31st March, 2003<br />
Sale of fixed assets to <strong>Glenmark</strong> Laboratories Limited – 345,000<br />
Corporate guarantee given for <strong>Glenmark</strong> Laboratories Limited – 50,000<br />
* <strong>Glenmark</strong> Laboratories Limited [Last year being an associate company]<br />
Key management personnel<br />
Professional fees – 61<br />
Share in profit of Cardiac Centre – 59<br />
Personal guarantees given by directors have been disclosed in Schedule 3.<br />
Managerial Remuneration<br />
Name of Directors<br />
1. Mr. Gracias Saldanha 10,104 7,660<br />
2. Mrs. B. E. Saldanha 7,177 5,155<br />
3. Mr. Glenn Saldanha 14,560 5,752<br />
4. Mrs. Cheryl Pinto 7,411 3,365<br />
c) Related party balances<br />
Receivable from associate enterprises – 267,988<br />
6) Leases<br />
a) The Company has entered into operating and finance lease agreements for the rental of property, vehicles,<br />
computers, equipment and other assets. Typically, lease agreements are for a period of three to five years. At<br />
March 31 2004, the Company had commitments under non-cancellable finance leases as follows:<br />
Rs. In ('000s)<br />
31st March, 2004 31st March, 2003<br />
Minimum lease payments<br />
Due within one year 7,368 8,875<br />
Due later than one year and not later than five years 4,146 11,514<br />
Total 11,514 20,389<br />
Present value of minimum lease payments<br />
Due within one year 5,528 7,454<br />
Due later than one year and not later than five years 2,728 8,256<br />
Total 8,256 15,710<br />
b) The Company has leased out certain equipments and these equipments have been capitalised in the books of<br />
account in accordance with Accounting Standard 19 - "Leases" issued by The Institute of Chartered Accountants<br />
of India in this regard. Depreciation has been provided based on the estimated useful life of the asset.<br />
116
Notes to the Consolidated Accounts<br />
For the year ended 31st March, 2004<br />
22 Notes to the Financial Statement (Contd.)<br />
6) Leases (Contd.)<br />
(i) Details in respect of assets given on operating Lease<br />
Rs. In ('000s)<br />
31st March, 2004 31st March, 2003<br />
Gross Block<br />
Leasehold Land 4,259 4,259<br />
Factory Buildings 22,065 22,065<br />
Plant and Machinery 5,838 5,838<br />
Equipments 3,764 3,764<br />
Furniture and Fixtures 165 165<br />
36,091 36,091<br />
Accumulated depreciation<br />
Leasehold Land 172 129<br />
Factory Buildings 3,302 2,566<br />
Plant and Machinery 1,104 826<br />
Equipments 1,272 971<br />
Furniture and Fixtures 66 49<br />
5,916 4,541<br />
Depreciation 1,375 1,375<br />
(ii) The lease income of Rs.1,920 (Previous Year -- Rs. 1,440) has been accrued on the basis of the lease agreement<br />
executed with the lessees. This lease is cancellable by notice of 30 days on either side.<br />
c) The Company has taken on operating lease a Bulk Drug Plant at Kurkumbh and godowns/residential & office<br />
premises at various locations in the country.<br />
i) The Company's significant leasing arrangements are in respect of the above plant, godowns & premises (Including<br />
furniture and fittings therein, as applicable ). The aggregate lease rentals payable are charged to Profit and Loss<br />
Account as Rent in Schedule 18 & 19.<br />
ii)<br />
The Leasing arrangements which are cancellable range between 11 months and 3 years. They are usually<br />
renewable by mutual consent on mutually agreeable terms. Under these arrangements, generally refundable<br />
interest free deposits have been given. An amount of Rs.13,742 towards deposit and unadjusted advance rent is<br />
recoverable from the lessor.<br />
117
Consolidated Schedules forming part of the Financial Statements<br />
For the year ended 31st March, 2004<br />
2 Notes to the Financial Statement (Contd.)<br />
7) Contingent liabilities not provided for<br />
Rs. In ('000s)<br />
31st March, 2004 31st March, 2003<br />
Guarantees issued by banks 9,981 6,856<br />
Disputed taxes/duties – 3,185<br />
Labour / Industrial disputes 1,120 –<br />
Open letters of credit 83,350 43,853<br />
Call Money payable to Bharuch Eco-acqua Infrastructure (@Rs. 6 per share) – 1,162<br />
Sundry debtors factored with recourse 91,677 126,285<br />
Corporate Guarantee 50,000 50,000<br />
Channel financing with recourse option 14,159 –<br />
8) Prior year comparatives<br />
Prior year's figures have been regrouped wherever necessary.<br />
Signatures to the Schedules 1 to 22 which form an integral part of the Consolidated Financial Statements.<br />
For and on behalf of<br />
Price Waterhouse<br />
Chartered Accountants<br />
Membership No. F 055913<br />
For and on behalf ofthe Board of Directors.<br />
Partha Ghosh M. J. Mendonza Glenn Saldanha Rajesh Desai<br />
Partner Vice President - Legal & Managing Director & CEO Director - Finance<br />
Mumbai, July 5, 2004<br />
Company Secretary<br />
118
GLENMARK EXPORTS LIMITED<br />
GLENMARK EXPORTS LIMITED<br />
B. E. Saldanha<br />
Gracias Saldanha<br />
Glenn Saldanha<br />
Mark Saldanha<br />
Auditors' Report<br />
Directors’ Report<br />
To<br />
The Member of<br />
GLENMARK EXPORTS LIMITED<br />
We have audited the attached Balance Sheet of<br />
GLENMARK EXPORTS LIMITED as at 31st March, 2004<br />
and the related Profit & Loss Account and Cash Flow<br />
Statement for the year ended on that date annexed<br />
thereto. These financial statements are the responsibility of<br />
the Company’s management. Our responsibility is to<br />
express an opinion on these financial statement based on<br />
our audit.<br />
We conducted our audit in accordance with the auditing<br />
standards generally accepted in India. These Standards<br />
require that we plan and perform the audit to obtain<br />
reasonable assurance about whether the financial<br />
statements are free from any material mis statement. An<br />
audit also includes, examining on a test basis, evidence<br />
supporting the amounts and disclosures in the financial<br />
statements. An audit also includes, assessing the<br />
accounting principles used and significant estimates made<br />
by management, as well as evaluating the overall financial<br />
statements presentation. We believe that our audit<br />
provides a reasonable basis for our opinion.<br />
1) As required by the Companies (auditors’ Report) order,<br />
2003, issued by the Central Government of India in<br />
terms of Sections 227 (4A) of the Companies Act,<br />
1956, and on the basis of such checks of books and<br />
records of the company as we considered appropriate<br />
and according to the information given to us, we give in<br />
the Annexure A statement on the matters specified in<br />
paragraphs 4 & 5 of the said order, to the extent<br />
applicable to the company.<br />
Further to our comments in the Annexure referred to in<br />
paragraph (1) above, we report that,<br />
a) We have obtained all the information and<br />
explanations, which to the best of our knowledge<br />
and belief were necessary for the purpose of our<br />
audit;<br />
b) In our opinion, proper book of accounts as required<br />
by law have been kept by the company, so far as it<br />
appears form our examination of those books.<br />
c) The Balance Sheet, Profit & Loss Account and Cash<br />
Flow Statement dealt with by this report are in<br />
agreement with the books of account.<br />
d) In our opinion, the Balance Sheet, Profit & Loss<br />
Account and Cash Flow Statement dealt with by<br />
this report comply with the accounting standard<br />
referred to in sub-sections (3c) of section 211 of the<br />
companies Act, 1956.<br />
e) Based on the written representations received from<br />
the Directors and taken on record by the Board of<br />
Directors and the information and explanations<br />
given to us, we report that none of the Directors<br />
are, as at 31st March, 2004, prima facie disqualified<br />
from being appointed as a Director in terms of<br />
clause (g) of sub-section (1) of section 274 of the<br />
Companies Act, 1956.<br />
f) In our opinion and to the best of our information and<br />
according to the explanation given to us, the said<br />
account give the information required by the<br />
Companies Act, 1956, in the manner so required<br />
and give a true and fair view in conformity with the<br />
accounting principles generally accepted in India;<br />
i) In the case of the Balance Sheet, of the State of<br />
affairs of the Company as at 31st March’ 2004<br />
ii) In the case of Profit & Loss Account, of the loss<br />
for the year ended on that date, and<br />
iii) In the case of Cash Flow Statement, of the cash<br />
flows for the year ended on that date.<br />
For N. K. Mittal & Associates<br />
Chartered Accountants<br />
To the Members,<br />
<strong>Glenmark</strong> Exports Limited<br />
Your Directors have pleasure in presenting their eight<br />
Annual Report together with the Audited Accounts of the<br />
Company for the year ended March 31, 2004.<br />
FINANCIAL RESULTS<br />
During the year the Company recorded a turnover of<br />
Rs.149.00 million as against Rs.125.55 million in the<br />
previous year.<br />
DIVIDEND<br />
Since there are inadequate profits your Directors regret<br />
their inability to recommend any dividend<br />
DIRECTORS<br />
Mrs. B. E. Saldanha retires by rotation at the ensuing<br />
Annual General Meeting and being eligible, offer herself for<br />
re-appointment.<br />
AUDITORS<br />
M/s. N. K. Mittal & Associates, Chartered Accountants,<br />
Auditors of the Company hold office until the conclusion of<br />
the ensuing Annual General Meeting and being eligible<br />
offer themselves for re-appointment.<br />
PERSONNEL<br />
As the Company has no employee, information as per<br />
Section 217(2A) of the Companies Act, 1956 read with the<br />
Companies (Particulars of Employees) Rules, 1975 are not<br />
applicable.<br />
COMPANIES (DISCLOSURE OF PARTICULARS IN THE<br />
REPORT OF BOARD OF DIRECTORS) RULES, 1988<br />
As the Company is not engaged in any manufacturing<br />
activity or Operations, the Companies (Disclosure of<br />
Particulars in the Report of Directors) Rules, 1988 so far as<br />
conservation of energy and technology absorption are not<br />
applicable.<br />
The Company earned foreign exchange amounting to<br />
Rs. 137.13 million on exports of products and the outflow<br />
was Rs. 15.90 million.<br />
DIRECTORS’ RESPONSIBILITY STATEMENT<br />
Pursuant to section 217 (2AA) of the Companies Act, 1956,<br />
the Directors confirm:<br />
(i) that in the preparation of the annual accounts, the<br />
applicable accounting standards had been followed.<br />
(ii) that appropriate accounting policies have been<br />
selected and applied consistently and have made<br />
judgments and estimates that are reasonable and<br />
prudent so as to give a true and fair view of the<br />
state of affairs of the Company as at March 31, 2004 and<br />
of the Profit or Loss of the Company for the year ended<br />
March 31, 2004.<br />
(iii) that proper and sufficient care has been taken for the<br />
maintenance of adequate accounting records in accordance<br />
with the provisions of the Companies Act, 1956 for<br />
safeguarding the assets of the Company and for preventing<br />
and detecting fraud and other irregularities.<br />
(iv) that the annual accounts have been prepared on a<br />
going concern basis.<br />
Place: Mumbai<br />
Date: July 5, 2004<br />
For and on behalf of the Board of Directors<br />
B. E. Saldanha<br />
Chairperson<br />
Place: Mumbai<br />
Date: 20/04/2004<br />
N. K. Mittal<br />
Proprietor<br />
Membership No. 46785<br />
GLENMARK EXPORTS LIMITED<br />
GLENMARK EXPORTS LIMITED<br />
Annexure (‘A’) to Auditor's Report<br />
Referred to in paragraph 4 of the Auditor’s Report of even<br />
date to the members of <strong>Glenmark</strong> Exports Limited on the<br />
financial statements for the year ended March 31, 2004.<br />
1.a)The Company is maintaining proper records showing<br />
full particulars including quantitative details and<br />
situation of fixed assets.<br />
b) The fixed assets are physically verified by the<br />
management according to a phased programmed<br />
designed to cover all the items over a period of three<br />
years, which in our opinion, is reasonable having<br />
regard to the size of the Company and the nature of its<br />
assets. Pursuant to the programme, a portion of the<br />
fixed assets has been physically verified by the<br />
management during the year and no material<br />
discrepancies between the book records and the<br />
physical inventory have been noticed.<br />
c) In our opinion and according to the information<br />
explanations given to us, no fixed assets has been<br />
disposed off by the Company during the year.<br />
2.(a)The inventory has been physically verified by the<br />
managament during the year. In our opinion, the<br />
frequency of verification is reasonable.<br />
b) In our opinion, the procedures of physical verification<br />
of inventory followed by the management are<br />
reasonable and adequate in relation to the size of the<br />
Company and the nature of its business.<br />
c) On the basis of our examination of the inventory<br />
records, in our opinion, the company is maintaining<br />
proper records of inventory. The discrepancies noticed<br />
on physical verification of inventory as compared to<br />
book records which have been properly dealt with in<br />
the books of account were not material.<br />
3. The Company has not granted unsecured loans, to<br />
Companies covered in the register maintained under<br />
Section 301 of the Act. The maximum amount involved<br />
during the year and the year end balance of such loans<br />
aggregates to Rs. Nil. The Company has not taken any<br />
loans, secured or unsecured, from Companies covered<br />
in the register maintained under Section 301 of the<br />
Act.<br />
4. In our opinion and according to the information and<br />
explanations given to us, having regard to the<br />
explanation that certain items purchased/ sold are of<br />
special nature for which suitable alternative sources do<br />
not exist for obtaining comparative quotations there<br />
are adequate internal control procedures<br />
commensurate with the size of the Company and the<br />
nature of its business for the purchase of inventory,<br />
fixed assets and for the sale of goods. Further, on the<br />
basis of our examination of the books and records of<br />
the Company, and according to the information and<br />
explanations given to us, we have neither come across<br />
nor have been informed of any continuing failure to<br />
correct major weaknesses in the aforesaid internal<br />
control procedures.<br />
5.a) In our opinion and according to the information and<br />
explanations given to us, the transactions that need to<br />
be entered into the register in pursuance of Section<br />
301 of Act, have been so entered.<br />
b) In our opinion and according to the information and<br />
explanations given to us having regard to the fact that<br />
the items purchased/sold/services rendered/received<br />
are of special nature and suitable alternate sources do<br />
not exist for obtaining comparative quotations, the<br />
transactions made in pursuance of contracts or<br />
arrangements entered into the register in pursuance of<br />
Section 301 of the Act and exceeding the value of<br />
Rupees Five Lakhs in respect of any party during the<br />
year, have been made at prices which are reasonable<br />
having regard to the prevailing market prices at the<br />
relevant time or the prices at which the transactions<br />
for similar goods have been made with other parties.<br />
6. The company has not accepted any deposits from the<br />
public within the meaning of Sections 58A and 58AA<br />
of the Act and the rules framed there under.<br />
7. In our opinion, the Company has an internal audit<br />
system commensurate with its size and nature of its<br />
business.<br />
8. The Central Government has not prescribed<br />
maintenance of cost records under Section 209 (1) (d)<br />
of the Companies Act 1956 for any of the products of<br />
the Company<br />
9.a) According to the information and explanations given to<br />
us and the records of the Company examined by us, in<br />
our opinion, the Company is generally regular in<br />
depositing the undisputed statutory dues including<br />
provident fund, investor education and protection fund,<br />
employees’ state insurance, income tax, sales tax,<br />
wealth tax, customs duty, excise duty, cess and other<br />
material statutory dues as applicable with the<br />
appropriate authorities.<br />
10.The company has no accumulated losses as at March<br />
31, 2004 and it has not incurred any cash losses in the<br />
financial year ended on that date or in the immediately<br />
preceding financial year.<br />
11. According to the records of the Company examined by<br />
us and the information and explanations given to us,<br />
the Company has not defaulted in repayment of dues<br />
to any financial institution or bank or debenture holders<br />
as at the balance sheet date.<br />
12. The Company has not granted any loans and advances<br />
on the basis of security by way of pledge of shares,<br />
debentures and other securities.<br />
13. The provisions of any special statute applicable to chit<br />
fund/ nidhi/ mutual beneit fund/ societies are not<br />
applicable to the Company.<br />
14.In our opinion, the Company is not a dealer or trader in<br />
shares, securities debentures and other investments.<br />
15. In our opinion, and according to the information and<br />
explanations given to us, the Company has not given<br />
any guarantee for loans taken by others from banks or<br />
financial institutions during year.<br />
16. in our opinion, and according to the information and<br />
explanations given to us, on an overall basis, the term<br />
loans have been applied for the purposes for which<br />
they obtained.<br />
17. On the basis of an overall examination of the balance<br />
sheet of the Company, in our opinion and according to<br />
the information and explanations given to us, there are<br />
no funds raised on a short term basis which have been<br />
used for long term investment.<br />
18. The Company has not made any preferential allotment<br />
of shares to parties and companies covered in register<br />
maintained under Section 301 of the act during year.<br />
19. The Company has not issued debentures, hence<br />
creation of securities in respect of debentures does<br />
not arise.<br />
20.The Company has not raised any money by public<br />
issues during year.<br />
21. During the course of our examination of the books and<br />
records of the Company, carried out in accordance<br />
with the generally accepted auditing practice in India,<br />
and according to the information and explanations<br />
given to us, we have neither come across any instance<br />
of material fraud on or by the Company, noticed or<br />
reported during the year, nor have we been informed<br />
of such case by the management.<br />
Place: Mumbai<br />
Date: 20/04/2004<br />
For N. K. Mittal & Associates<br />
Chartered Accountants<br />
N. K. Mittal<br />
Proprietor<br />
Membership No. 46785<br />
119
GLENMARK EXPORTS LIMITED<br />
GLENMARK EXPORTS LIMITED<br />
Balance Sheet<br />
Amount in Rs.<br />
As at 31st March, Schedules 2004 2003<br />
SOURCES OF FUNDS<br />
Shareholders' Funds<br />
Share Capital 1 10 00 200 10 00 200<br />
Reserves & Surplus 2 73 78 445 73 78 445<br />
Total 83 78 645 83 78 645<br />
APPLICATION OF FUNDS<br />
Fixed Assets 3<br />
Gross Block 6 55 573 6 55 573<br />
Less: Depreciation 3 29 472 2 73 228<br />
Net Block 3 26 101 3 82 345<br />
Current Assets, Loans & Advances<br />
Sundry Debtors 4 21 86 17 589 17 23 97 359<br />
Cash & Bank balances 5 21 48 119 51 04 115<br />
Loans & Advances 6 2 21 03 082 1 36 27 270<br />
24 28 68 790 19 11 28 744<br />
Current Liabilities & Provisions 7 23 48 16 246 18 31 32 444<br />
Net Current Assets 80 52 544 79 96 300<br />
Total 83 78 645 83 78 645<br />
Notes to accounts & Significant Accounting Policies 8<br />
As per our report of even date annexed<br />
For N. K. Mittal & Associates<br />
Chartered Accountants<br />
For & on behalf of the Board of Directors<br />
N. K. Mittal B. E. Saldanha G. Saldanha<br />
(Proprietor) Chairperson Director<br />
Membership No. 46785<br />
Place: Mumbai.<br />
Date: 20 April, 2004<br />
Profit & Loss Account<br />
Amount in Rs.<br />
For the year ended 31st March, Schedules 2003-2004 2002-2003<br />
INCOME<br />
Sales 14 90 08 543 12 55 53 369<br />
Total 14 90 08 543 12 55 53 369<br />
EXPENDITURE<br />
Cost of Goods Traded 14 90 08 543 12 55 53 369<br />
14 90 08 543 12 55 53 369<br />
Profit Before Taxation – –<br />
Provision for Taxation – –<br />
Profit After Taxation – –<br />
Balance Brought Forward from Previous Year 73 78 445 73 78 445<br />
Balance Carried to Balance Sheet 73 78 445 73 78 445<br />
Notes to accounts & Significant Accounting Policies 8<br />
As per our report of even date annexed<br />
For N. K. Mittal & Associates<br />
Chartered Accountants<br />
For & on behalf of the Board of Directors<br />
N. K. Mittal B. E. Saldanha G. Saldanha<br />
(Proprietor) Chairperson Director<br />
Membership No. 46785<br />
Place: Mumbai.<br />
Date: 20 April, 2004<br />
Schedules forming part of Accounts<br />
Amount in Rs.<br />
2003-2004 2002-2003<br />
1 Share Capital<br />
Authorised Capital<br />
250000 Equity Shares of Rs.10/- each 25 00 000 25 00 000<br />
Issued, Subscribed & Paid up Capital<br />
100020 Equity Shares of Rs.10/- each fully paid up 10 00 200 10 00 200<br />
(Fully held by <strong>Glenmark</strong> Pharmaceuticals Limited, the Holding Company)<br />
2 Reserves & Surplus<br />
Profit & Loss Account 73 78 445 73 78 445<br />
GLENMARK EXPORTS LIMITED<br />
GLENMARK EXPORTS LIMITED<br />
Schedules forming part of Accounts<br />
3 Fixed Assets<br />
Amount in Rs.<br />
Particulars Gross Block Depreciation Net Block<br />
Additions/ Additions/ Depreciation the<br />
Bal as the Bal as the Bal as the Bal as<br />
beginning (Deletions) end of the beginning (Deletions) for the end of the<br />
of the year year of the year year year<br />
Office Equipments 1 90 370 – 1 90 370 52 258 – 12 050 64 308 1 26 062<br />
Vehicles 4 65 203 – 4 65 203 2 20 970 – 44 194 2 65 164 2 00 039<br />
Total 6 55 573 – 6 55 573 2 73 228 – 56 244 3 29 472 3 26 101<br />
Previous year 6 55 573 – 6 55 573 2 16 984 – 56 244 2 73 228 3 82 345<br />
Amount in Rs.<br />
2003-2004 2002-2003<br />
4 Sundry Debtors<br />
Unsecured<br />
More than 6 months 13 05 56 384 8 80 00 448<br />
Less: Provision for doubtful debts 40 62 469 40 62 469<br />
12 64 93 915 8 39 37 979<br />
Others considered good 9 21 23 674 8 84 59 380<br />
21 86 17 589 17 23 97 359<br />
5 Cash & Bank Balances<br />
Balance with Banks 21 48 119 51 03 915<br />
Cash in hand – 200<br />
21 48 119 51 04 115<br />
6 Loans & Advances<br />
TDS receivabe 70 226 70 226<br />
Export Incentive Receivable 2 19 62 406 1 25 65 594<br />
Deposits with Sales Tax Authority 5 000 5 000<br />
Advance receivable in cash or in kind or for value to be received 65 450 9 86 450<br />
2 21 03 082 1 36 27 270<br />
NOTES TO THE FINANCIAL STATEMENTS FOR THE YEAR ENDED 31st MARCH, 2004<br />
1. Background<br />
<strong>Glenmark</strong> Exports Limited (the Company) was incorporated on 26 Aug 1996. The Company is principally engaged in<br />
exporting pharmaceutical products.<br />
2. Summary of Significant Accounting Policies<br />
a) The Financial statement are prepared under the historical cost convention, on the accrual basis of accounting and in<br />
accordance with the accounting standards specified in section 211 (3) (c) of the Companies Act, 1956.<br />
b) Fixed Assets and Depreciation :<br />
Fixed assets are stated at cost less depreciation. The Company capitalizes all costs relating to the acquisition and<br />
installation of Fixed Assets. Depreciation is provided using the straight line method, pro-rata to the period of use of<br />
assets, at the rates specified in Schedule XIV of the Companies Act, 1956.<br />
c) Foreign currency transaction :<br />
Foreign currency transactions, during the year are recorded at the rates of exchange prevailing on the date of the<br />
transaction. Foreign currency assets and liabilities are translated into rupees at the rates of exchange prevailing on<br />
the date of the Balance Sheet. All exchange difference are dealt with in the statement of profit and loss, except<br />
those relating to the acquisition of fixed assets which are adjusted in the cost of the respective fixed assets.<br />
3. Earning in Foreign Currency<br />
Amount in Rs.<br />
Current Year Previous Year<br />
a) Earning in Foreign exchange viz., F.O.B. value of exports 13,71,33,516 11,69,42,842<br />
b) Expenditure in Foreign Currency 1,59,19,936 12,88,662<br />
Information required by paras 3 & 4 of Part II of Schedule VI of the Companies Act, 1956.<br />
4. Details of Purchases of Finished Goods Marketed by the Company :<br />
Group Current Year Previous Year<br />
Quantity Amount Quantity Amount<br />
Units (In Nos.) Rs. Units (In Nos.) Rs.<br />
Orals 3658800 12,29,06,188 1888780 6,82,59,139<br />
Lotions 30000 8,25,210 59740 12,65,084<br />
Tablets 100000 40,83,022 60000 14,75,125<br />
Ointments 798400 1,84,32,083 2023347 5,28,91,521<br />
Others 23985 27,62,040 70000 16,62,500<br />
Total 14,90,08,543 12,55,53,369<br />
5) Previous year’s figures have been regrouped and rearranged wherever necessary.<br />
120<br />
7 Current Liabilities & Provisions<br />
Sundry Creditors<br />
For Goods 22 26 80 058 17 64 59 828<br />
For expenses 1 21 32 673 65 75 674<br />
Duty Drawback Payable – 69 292<br />
TDS Payable 3 515 1 313<br />
Sales Tax Payable – 26 337<br />
23 48 16 246 18 31 32 444<br />
For N. K. Mittal & Associates<br />
Chartered Accountants<br />
For & on behalf of the Board of Directors<br />
N. K. Mittal B. E. Saldanha G. Saldanha<br />
(Proprietor) Chairperson Director<br />
Membership No. 46785<br />
Place: Mumbai.<br />
Date: 20 April, 2004
GLENMARK EXPORTS LIMITED<br />
GLENMARK EXPORTS LIMITED<br />
Balance Sheet Abstract and Company’s General Business Profile<br />
(a) Registration Details<br />
Registration No.<br />
Balance Sheet Date<br />
(b) Capital raised during the year<br />
Public Issue<br />
Rights Issue<br />
N I L<br />
N I L<br />
Bonus Issue<br />
Private Placement<br />
N I L N I L<br />
(c) Position of Mobilisation and Deployment of Funds<br />
Total Liabilities and shareholders funds<br />
Total Assets<br />
8 3 7 8 6 4 5<br />
8 3 7 8 6 4 5<br />
Sources of Funds<br />
Paid-up Capital<br />
Reserves & Surplus<br />
1 0 0 0 2 0 0<br />
7 3 7 8 4 4 5<br />
Secured loan<br />
Unsecured loan<br />
N I L N I L<br />
Application of Funds<br />
Net Fixed Assets<br />
3 2 6 1 0 1<br />
Investments<br />
N I L<br />
Miscellaneous expenditure<br />
N I L<br />
1 0 2 1 2 5<br />
(D) PERFORMANCE OF THE COMPANY<br />
Turnover<br />
1 4 9 0 0 8 5 4 3<br />
Profit/(Loss) Before Tax<br />
N I L<br />
Earnings per share in Rs<br />
N I L<br />
3 1 0 3 2 0 0 4<br />
Date Month Year<br />
The accompanying notes and Schedules 1 to 8 are an integral part of this balance sheet.<br />
State Code<br />
1 1<br />
Capital Work in Progress<br />
N I L<br />
Net Current Assets<br />
8 0 5 2 5 4 4<br />
Accumulated losses<br />
N I L<br />
Total Expenditure<br />
1 4 9 0 0 8 5 4 3<br />
Profit/(Loss) After Tax<br />
N I L<br />
Dividend rate<br />
N I L<br />
As per our report of even date.<br />
For N. K. Mittal & Associates<br />
For & on behalf of the Board of Directors<br />
Chartered Accountants<br />
N. K. Mittal B. E. Saldanha G. Saldanha<br />
(Proprietor) Chairperson Director<br />
Membership No. 46785<br />
Place: Mumbai.<br />
Date: 20 April, 2004<br />
COMPLIANCE CERTIFICATE<br />
The Members<br />
<strong>Glenmark</strong> Exports Limited<br />
Mumbai<br />
Company Registration No. Authorised Capital Paid Up Capital<br />
11 - 102125 Rs. 25,00,000/- Rs. 10,00,200/-<br />
I have examined the registers, records, books and papers of GLENMARK EXPORTS LIMITED (the Company) as required to<br />
be maintained under the Companies Act, 1956, (the Act) and the rules made thereunder and also the provisions contained<br />
in the Memorandum of Association and Articles of Association of the Company for the financial year ended on March 31,<br />
2004 (financial year). In my opinion and to the best of my information and according to the examinations carried out by me<br />
and explanations furnished to me by the Company, its officers and agents, I certify that in respect of the aforesaid financial<br />
year:<br />
1. The Company has kept and maintained all registers as stated in Annexure `A' to this certificate, as per the provisions of<br />
the Act and the rules made thereunder and all entries therein have been duly recorded.<br />
2. The Company has duly filed the forms and returns as stated in Annexure `B' to this certificate, with the Registrar of<br />
Companies, Regional Director, Central Government, Company Law Board or other authorities within the time prescribed<br />
under the Act and the rules made thereunder.<br />
3. The Company being a Public Limited Company has the minimum prescribed paid up capital and its maximum number<br />
of members during the said financial year was 7 (seven) excluding its present and past employees<br />
4. The Board of Directors duly met 5 times respectively on May 5, 2003, May 9, 2003, June 16, 2003, December 1, 2003<br />
and March 16, 2004 in respect of which meetings proper notices were given and the proceedings were properly<br />
recorded and signed.<br />
5. The company has not closed its Register of Members under Section 154 of the Act during the financial year under<br />
review.<br />
6. The Annual General Meeting for the financial year ended on March 31, 2003 was held on September 25, 2003 after<br />
giving due notice to the members of the Company and the resolutions passed thereat were duly recorded in Minutes<br />
Book maintained for the purpose.<br />
7. No Extra ordinary General Meeting was held during the financial year.<br />
8. The Company has not advanced any loan to its Directors, or persons, or firms and Companies referred to under section<br />
295 of the Act.<br />
9. The Company has not entered into any contract falling within the purview of Section 297 of the Act.<br />
10. The Company has not required to make any entries in the register maintained under Section 301 of the Act.<br />
11. As there were no instances falling within the purview of section 314 of the Act the Company was not required to<br />
obtain any approval from the Board of Directors, Members and Central Government.<br />
12. The Board of Directors has not issued duplicate share certificates during the financial year under review.<br />
13. The Company has during the financial year:<br />
(i) not made any allotment, transfer/transmission of any shares/debentures and other securities.<br />
(ii) not declared any dividends and hence the Company was not required to deposit any amount as unpaid dividend /<br />
unclaimed dividend in a separate Bank Account.<br />
(iii) not paid / posted warrants for dividends to any members, since no dividend was declared.<br />
(iv) not transferred any amounts to the Investors Education and Protection Fund since there were no unpaid dividends,<br />
application money due for refund, matured deposits, matured debentures and the interest accrued thereon,<br />
outstanding for a period of seven years.<br />
(v) has duly complied with the requirements of Section 217 of the Act.<br />
GLENMARK EXPORTS LIMITED<br />
GLENMARK EXPORTS LIMITED<br />
14. The Board of Directors of the Company is duly constituted and the appointment of directors has been duly made.<br />
15. The Company has not appointed a Managing Director/ Whole time Director/ Manager during the financial year under<br />
review.<br />
16. The Company has not appointed any sole-selling agents during the financial year under review.<br />
17. As explained to me, the Company was not required to obtain any approvals from the Central Government, Company<br />
Law Board, Regional Director, Registrar of Companies and/or such other authorities as may be prescribed under the<br />
various provisions of the Act.<br />
18. The Directors have disclosed their interest in other firms/companies to the Board of Directors pursuant to the<br />
provisions of the Act and the rules made thereunder.<br />
19. The Company has not issued any equity shares/debentures/other securities during the financial year.<br />
20. The Company has not bought back any shares during the financial year ending March 31, 2004.<br />
21. Since the Company has no preference shares/debentures, the Company was therefore not required to redeem any<br />
preference share/debentures during the year under review.<br />
22. There were no transactions necessitating the Company to keep in abeyance the rights to dividend, rights shares and<br />
bonus shares pending registration of transfer of shares.<br />
23. The Company has not invited/accepted any deposits including any unsecured loans falling within the purview of the<br />
provisions of Section 58A and 58AA read with Companies (Acceptance of Deposit) Rules, 1975 during the year under<br />
review.<br />
24. The Company has not made any borrowing during the financial year ended March 31, 2004.<br />
25. The loans given and investments made, or guarantees given or securities provided to other bodies corporate by the<br />
Company during the year does not attract the provision of Section 372A of the Companies Act, 1956<br />
26. The Company has not altered the provisions of the Memorandum of Association with respect to situation of the<br />
Company's registered office from one state to another during the year under scrutiny.<br />
27. The Company has not altered the provisions of the Memorandum of Association with respect to the objects of the<br />
Company during the year under scrutiny.<br />
28. The company has not altered the provisions of the Memorandum of Association with respect to name of the Company<br />
during the year under scrutiny.<br />
29. The Company has not altered the provisions of the Memorandum of Association with respect to share capital of the<br />
Company during the year under scrutiny.<br />
30. The Company has not altered its Articles of Association during the year under scrutiny.<br />
31. There were no prosecution initiated against or show cause notices received by the Company for alleged offences under<br />
the Act and also the fines and penalties or any other punishment imposed on the Company during the financial year<br />
under review.<br />
32. The Company has not received any money as security from its employees during the year under review.<br />
33. The Company has not deposited both employee’s and employer’s contribution to Provident Fund with the prescribed<br />
authorities pursuant to Section 418 of the Act.<br />
For Martinho Ferrao & Associates<br />
Company Secretaries<br />
ANNEXURE ' A '<br />
No. Registers as maintained by the Company Under Section<br />
1 Register of Members 150<br />
2 Minutes Books of proceedings of<br />
i) General Meeting 193<br />
ii) Board of Directors 193<br />
3 Register of Contracts, Companies & Firms in which Directors are interested 301<br />
4 Register of Directors 303<br />
5 Register of Director Shareholding 307<br />
6 Register of Loans & Investments 372A<br />
7 Register of Transfer of Shares Voluntary<br />
8 Register of Charges & Instruments creating charges 136<br />
For Martinho Ferrao & Associates<br />
Company Secretaries<br />
Martinho Ferrao<br />
Place: Mumbai<br />
Proprietor<br />
Dated: July 5, 2004 C P. No. 5676<br />
Returns filed by the Company with the Registrar of Companies, Maharashtra, Mumbai, during the Financial Year<br />
on March 31, 2004<br />
ANNEXURE ' B '<br />
No Form No. Under Purpose When Filed<br />
Section<br />
with ROC<br />
1 Annual Return (as on September 25, 2003) 159 As prescribed in the section November 13, 2003<br />
2 Balance Sheet (as on March 31, 2003) 220 As prescribed in the section October 7, 2003<br />
3 Compliance Certificate 383 (A) As required under the issuance October 7, 2003<br />
of Compliance<br />
Certificate Rules, 2001<br />
For Martinho Ferrao & Associates<br />
Company Secretaries<br />
Martinho Ferrao<br />
Place: Mumbai<br />
Proprietor<br />
Dated: July 5, 2004 C P. No. 5676<br />
Martinho Ferrao<br />
Place: Mumbai<br />
Proprietor<br />
Dated: July 5, 2004 C P. No. 5676<br />
121
GM PHARMA LIMITED<br />
Glenn Saldanha<br />
Gracias Saldanha<br />
B. E. Saldanha<br />
Auditors' Report<br />
GM PHARMA LIMITED<br />
Directors’ Report<br />
To<br />
The Member of GM PHARMA LIMITED.<br />
We have audited the attached Balance Sheet of GM<br />
PHARMA LIMITED as at 31st March, 2004 and the related<br />
Profit & Loss Account and Cash Flow Statement for the<br />
year ended on that date annexed thereto. These financial<br />
statements are the responsibility of the Company’s<br />
management. Our responsibility is to express an opinion on<br />
these financial statement based on our audit.<br />
We conducted our audit in accordance with the auditing<br />
standards generally accepted in India. These Standards<br />
require that we plan and perform the audit to obtain<br />
reasonable assurance about whether the financial<br />
statements are free from any material misstatement. An<br />
audit also includes, examining on a test basis, evidence<br />
supporting the amounts and disclosures in the financial<br />
statements. An audit also includes, assessing the<br />
accounting principles used and significant estimates made<br />
by management, as well as evaluating the overall financial<br />
statements presentation. We believe that our audit<br />
provides a reasonable basis for our opinion.<br />
1) As required by the Companies (auditors’ Report) order,<br />
2003, issued by the Central Government of India in<br />
terms of Sections 227 ( 4A) of the Companies Act,<br />
1956, and on the basis of such checks of books and<br />
records of the company as we considered appropriate<br />
and according to the information given to us, we give<br />
in the Annexure A statement on the matters specified<br />
in paragraphs 4 & 5 of the said order, to the extent<br />
applicable to the company.<br />
Further to our comments in the Annexure referred to in<br />
paragraph (1) above, we report that;<br />
(a) We have obtained all the information and explanations,<br />
which to the best of our knowledge and belief were<br />
necessary for the purpose of our audit;<br />
(b) In our opinion, proper book of accounts as required by<br />
law have been kept by the company, so far as it<br />
appears from our examination of those books.<br />
(c) The Balance Sheet, Profit & Loss Account and Cash<br />
Flow Statement dealt with by this report are in<br />
agreement with the books of account.<br />
To the Members,<br />
GM Pharma Limited<br />
Your Directors have pleasure in presenting their Fifth<br />
Annual Report together with the Audited Accounts of the<br />
Company for the year ended March 31, 2004.<br />
FINANCIAL RESULTS<br />
There were no operations during the financial year.<br />
DIVIDEND<br />
Since there is no operating income, your Directors regret<br />
their inability to recommend any dividend.<br />
DIRECTORS<br />
Mr. Glenn Saldanha retires at the ensuing Annual General<br />
Meeting and being eligible, offer himself for reappointment.<br />
AUDITORS<br />
M/s. N. K. Mittal & Associates, Chartered Accountants,<br />
Auditors of the Company hold office until the conclusion of<br />
the ensuing Annual General Meeting and being eligible<br />
offer themselves for re-appointment.<br />
PERSONNEL<br />
As the Company has no employee, information as per<br />
Section 217(2A) of the Companies Act, 1956 read with the<br />
Companies (Particulars of Employees) Rules, 1975, are not<br />
applicable.<br />
COMPANIES (DISCLOSURE OF PARTICULARS IN THE<br />
REPORT OF BOARD OF DIRECTORS) RULES, 1988<br />
The Directors have nothing to report on conservation of<br />
energy, technology absorption and foreign exchange<br />
earnings and outgo as the Company is not engaged in any<br />
manufacturing activity and has no foreign collaboration.<br />
DIRECTORS’ RESPONSIBILITY STATEMENT<br />
Pursuant to section 217 (2AA) of the Companies Act, 1956,<br />
the Directors confirm:<br />
((i) that in the preparation of the annual accounts, the<br />
applicable accounting standards had been followed.<br />
(ii) that appropriate accounting policies have been selected<br />
and applied consistently and have made judgments and<br />
estimates that are reasonable and prudent so as to give<br />
a true and fair view of the state of affairs of the<br />
Company as at March 31, 2004 and of the Profit or<br />
Loss of the Company for the year ended March 31,<br />
2004.<br />
(iii) that proper and sufficient care has been taken for the<br />
maintenance of adequate accounting records in<br />
accordance with the provisions of the Companies Act,<br />
1956 for safeguarding the assets of the Company and<br />
for preventing and detecting fraud and other<br />
irregularities.<br />
(iv) that the annual accounts have been prepared on a<br />
going concern basis.<br />
Place: Mumbai<br />
Date: July 5, 2004<br />
For and on behalf of the Board of Directors<br />
Glenn Saldanha<br />
Chairman<br />
(d) In our opinion, the Balance Sheet, Profit & Loss<br />
Account and Cash Flow Statement dealt with by this<br />
report comply with the accounting standard referred to<br />
in sub- sections (3c) of section 211 of the companies<br />
Act, 1956.<br />
e) Based on the written representations received from the<br />
Directors as on 31st March, 2004 taken on record by<br />
the Board of Directors and the information &<br />
explanation given to us, we report that non of the<br />
directors are as at 31st March, 2004, prima-facie<br />
disqualified from being appointed as a Director in terms<br />
of clause (g) of sub- section (1) of section 274 of the<br />
Companies Act, 1956.<br />
(f) In our opinion and to the best of our information and<br />
according to the explanation given to us, the said<br />
account give the information required by the<br />
Companies Act, 1956, in the manner so required and<br />
give a true and fair view in conformity with the<br />
accounting principles generally accepted in India;<br />
(i) In the case of the Balance Sheet, of the State of<br />
affairs of the Company as at 31st March’2004<br />
(ii) In the case of Profit & Loss Account, of the loss for<br />
the year ended on that date; and<br />
(iii) In the case of Cash Flow Statement, of the cash<br />
flows for the year ended on that date.<br />
For N. K. Mittal & Associates<br />
Chartered Accountants<br />
Place: Mumbai<br />
Date: 20/04/2004<br />
N. K. Mittal<br />
Proprietor<br />
Membership No. 46785<br />
GM PHARMA LIMITED<br />
GM PHARMA LIMITED<br />
122<br />
Annexure to the Auditors' Report<br />
Referred to in paragraph 1 of the Auditor’s Report of even<br />
date to the members of G M Pharma Limited on the<br />
financial statements for the year ended March 31, 2004.<br />
1. The Company does not have any fixed assets, hence<br />
para 4 A (1) (a),(b) and (c) relating to the maintenance<br />
of records for fixed assets is not applicable to the<br />
company.<br />
2. The Company is not engaged in any manufacturing<br />
activity hence para 4 A (2) (a), (b) and (c), (4), (5) (a) &<br />
(b), are not applicable to the company.<br />
3. The Company has not taken any loans, secured or<br />
unsecured, from Companies covered in the register<br />
maintained under Section 301 of the Act. Hence para<br />
4A, 3 (b),(c) and (d) are not applicable to the Company.<br />
4. The company has not accepted any deposits from the<br />
public within the meaning of Sections 58A and 58AA<br />
of the Act and the rules framed there under.<br />
5. In our opinion, the Company has an internal audit<br />
system commensurate with its size and nature of its<br />
business.<br />
6. The Central Government has not prescribed<br />
maintenance of cost records under Section 209 (1)(d)<br />
of the Companies Act 1956 for any of the products of<br />
the Company<br />
7. According to the information and explanations given to<br />
us and the records of the Company examined by us, in<br />
our opinion, the Company is generally regular in<br />
depositing the undisputed statutory dues including<br />
provident fund, investor education and protection fund,<br />
employees’ state insurance, income-tax, sales-tax,<br />
wealth tax, customs duty, excise duty, cess and other<br />
material statutory dues as applicable with the<br />
appropriate authorities.<br />
8. The Company no accumulated losses as at March<br />
31,2004 and it has incurred cash losses of Rs.7420/- in<br />
the financial year ended on that date.<br />
9. According to the records of the Company examined by<br />
us and the information and explanations given to us,<br />
the Company has not defaulted in repayment of dues<br />
to any financial institution or bank or debenture holders<br />
as at the balance sheet date.<br />
10. The Company has not granted any loans and advances<br />
on the basis of security by way of pledge of shares,<br />
debentures and other securities.<br />
11. The provisions of any special statute applicable to chit<br />
fund/ nidhi/ mutual benefit fund/ societies are not<br />
applicable to the Company.<br />
12. In our opinion, the Company is not a dealer or trader in<br />
shares, securities, debentures and other investments.<br />
13. In our opinion, and according to the information and<br />
explanations given to us, the Company has not given<br />
any guarantee for loans taken by others from banks or<br />
financial institutions during the year.<br />
14 In our opinion, and according to the information and<br />
explanations given to us, on an overall basis, the<br />
Company has not availed any term loans.<br />
15 On the basis of an overall examination of the balance<br />
sheet of the Company, in our opinion and according to<br />
the information and explanations given to us, there are<br />
no funds raised on a short –term basis which have<br />
been used for long- term investment.<br />
16. The Company has not made any preferential allotment of<br />
shares to parties and companies covered in register<br />
maintained under Section 301 of the act during the year.<br />
17. The Company has not issued any debenture, hence<br />
securities created in respect of debenture is not<br />
applicable to the company.<br />
18.The Company has not raised any money by public<br />
issues during the year.<br />
19. During the course of our examination of the books and<br />
records of the Company, carried out in accordance<br />
with the generally accepted auditing practices in India,<br />
and according to the information and explanations<br />
given to us, we have neither come across any instance<br />
of material fraud on or by the Company, noticed or<br />
reported during the year, nor have we been informed<br />
of such case by the management.<br />
For N. K. Mittal & Associates<br />
Chartered Accountants<br />
Place: Mumbai<br />
Date: 20/04/2004<br />
N. K. Mittal<br />
Proprietor<br />
Membership No. 46785<br />
Balance Sheet<br />
Amount in Rs.<br />
As at 31st March, Schedules 2004 2003<br />
SOURCES OF FUNDS<br />
Shareholders' Funds<br />
Share Capital 1 10 00 000 10 00 000<br />
Total 10 00 000 10 00 000<br />
APPLICATION OF FUNDS<br />
Current Assets, Loans & Advances 2<br />
Cash & Bank balances 3 46 107 76 535<br />
Loans & Advances 7 88 054 10 88 055<br />
11 34 161 11 64 590<br />
Current Liabilities & Provisions 3<br />
Sundry Creditors 1 37 820 1 32 420<br />
Other Liabilities 3 762 32 170<br />
1 41 581 1 64 590<br />
Net current Assets 9 92 580 10 00 000<br />
Profit & Loss Account 7 420 –<br />
Total 10 00 000 10 00 000<br />
Notes to accounts & Significant Accounting Policies 4<br />
As per our report of even date annexed<br />
For N. K. Mittal & Associates<br />
Chartered Accountants<br />
For & on behalf of the Board of Directors<br />
N. K. Mittal Glenn Saldanha G. Saldanha<br />
(Proprietor) Chairman Director<br />
Membership No. 46785<br />
Place: Mumbai.<br />
Date: 20 April, 2004
GM PHARMA LIMITED<br />
GM PHARMA LIMITED<br />
Profit & Loss Account<br />
Amount in Rs.<br />
For the year ended 31st March, Schedules 2004 2003<br />
INCOME<br />
Sales – –<br />
Total – –<br />
EXPENDITURE<br />
Bank Charges 20 –<br />
ROC Fees 2 000 –<br />
Auditor's Remuneration 5 400 –<br />
7 420 –<br />
Profit / (Loss) for the year (7 420) –<br />
Profit After Taxation (7 420) –<br />
Profit / (Loss) b/f from the previous year<br />
Balance Carried to Balance Sheet (7 420) –<br />
Schedules forming part of the Balance Sheet<br />
Amount in Rs.<br />
As at 31st March, 2004 2003<br />
1. SHARE CAPITAL<br />
Authorised Share Capital 1,000,000 1,000,000<br />
1,00,000 Equity shares of Rs. 10/- each.<br />
Issued , Subscribed & Paid-up Share capital 1,000,000 1,000,000<br />
1,00,000 Equity shares of Rs. 10/- each.<br />
(Fully held by <strong>Glenmark</strong> Pharmaceuticals Limited, the Holding Company)<br />
2. Current assets , Loans & Advances<br />
Cash & Bank balance 346,107 76,535<br />
Advance recoverable in cash or kind 788,054 1,088,055<br />
1,134,161 1,164,590<br />
3. Current Liabilities & Provisions<br />
Sundry creditors 137,820 132,420<br />
Other liabilities 3,761 32,170<br />
141,581 164,590<br />
As per our report of even date annexed<br />
For N. K. Mittal & Associates<br />
Chartered Accountants<br />
For & on behalf of the Board of Directors<br />
N. K. Mittal Glenn Saldanha G. Saldanha<br />
(Proprietor) Chairman Director<br />
Membership No. 46785<br />
Place: Mumbai.<br />
Date: 20 April, 2004<br />
4. Notes to the Financial Statements for the year ended March 31, 2004<br />
SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES<br />
1. The financial statements are prepared under the historical cost convention, on the accrual basis of accounting and in<br />
accordance with the accounting standards specified in section 211 (3C) of the Companies Act, 1956.<br />
2. In the opinion of the Board, Current Assets, Loans and Advances are approximately of the value stated, if realized in<br />
ordinary course of the business.<br />
3. Previous year’s figures have been regrouped and rearranged wherever necessary.<br />
As per our report of even date attached.<br />
For N. K. Mittal & Associates<br />
Chartered Accountants<br />
For & on behalf of the Board of Directors<br />
N. K. Mittal Glenn Saldanha G. Saldanha<br />
(Proprietor) Chairman Director<br />
Membership No. 46785<br />
Place: Mumbai.<br />
Date: 20 April, 2004<br />
GM PHARMA LIMITED<br />
Compliance Certificate<br />
To,<br />
The Members,<br />
GM PHARMA LIMITED,<br />
Mumbai.<br />
Company Registration No. Authorised Capital Paid Up Capital<br />
11 – 121475 Rs. 10,00,000/- Rs. 10,00,000/-<br />
We have examined the registers, records, books and<br />
papers of GM PHARMA LIMITED (the Company) as<br />
required to be maintained under the Companies Act, 1956,<br />
(the Act) and the rules made thereunder and also the<br />
provisions contained in the Memorandum of Association<br />
and Articles of Association of the Company for the financial<br />
year ended 31st March, 2004 (financial year). In our<br />
opinion, and to the best of our information and according to<br />
the examinations carried out by us and explanations<br />
furnished to us by the Company, its officers and agents,<br />
we certify that in respect of the aforesaid financial year:<br />
1. The Company has kept and maintained all registers as<br />
stated in Annexure `A’ to this certificate, as per the<br />
provisions of the Act and the rules made thereunder<br />
and all entries therein have been duly recorded.<br />
2. The Company has duly filed the forms and returns as<br />
stated in Annexure `B’ to this certificate, with the<br />
Registrar of Companies, Regional Director, Central<br />
Government, Company Law Board or other authorities<br />
within the time prescribed under the Act and the rules<br />
made thereunder.<br />
3. The Company, being a Subsidiary of Public Limited<br />
Company became a Public Limited Company in terms<br />
of section 3(1)(iv)(c) of the Companies Act, 1956 and its<br />
maximum number of members during the financial year<br />
was Seven and has the minimum prescribed paid-up<br />
capital.<br />
4. The Board of Directors duly met four times respectively<br />
on 16th May, 2003, 16th July, 2003, 1st December,<br />
2003 and 16th March, 2004 in respect of which<br />
meetings proper notices were given and the<br />
proceedings were properly recorded and signed.<br />
5. The Company was not required to close its Register of<br />
Members or Debenture holders during the financial<br />
year.<br />
6. The Annual General Meeting for the financial year ended<br />
on 31st March 2003 was held on 25th September, 2003<br />
after giving due notice to the members of the Company<br />
and the resolutions passed thereat were duly recorded<br />
in Minutes Book maintained for the purpose.<br />
7. No Extra Ordinary General Meeting was held during the<br />
financial year.<br />
8. The Company has not advanced any loan to its Directors<br />
or persons or firms or companies referred to under<br />
Section 295 of the Act.<br />
9. The Company has not entered into any contracts falling<br />
within the purview of Section 297 of the Act.<br />
10. The Company was not required to make any entries in<br />
the register maintained under Section 301 of the Act.<br />
11. As there were no instances falling within the purview<br />
of Section 314 of the Act, the Company has not<br />
obtained any approvals from the Board of Directors,<br />
members or Central Government, as the case may be.<br />
12. The Company has not issued any duplicate certificates<br />
during the financial year.<br />
13. During the financial year;<br />
a. There was no allotment transfer/transmission of<br />
securities during the year.<br />
b. The Company has not deposited any amount in a<br />
separate bank account as no dividend was declared.<br />
c. The Company has not posted warrants to any<br />
member of the Company as no dividend was<br />
declared.<br />
d. There was no amounts in unpaid dividend account,<br />
application money due for refund, matured deposits,<br />
matured debentures and the interest accrued which<br />
have remained unclaimed or unpaid for a period of<br />
seven years be transferred to Investor Education<br />
and Protection Fund.<br />
e. The Company has duly complied with the<br />
requirements of Section 217 of the Act.<br />
14. The Board of Directors of the Company is duly<br />
constituted. There was no appointment of additional<br />
directors, alternate directors and directors to fill casual<br />
vacancy during the financial year.<br />
15. The Company has not appointed any Managing<br />
Director/ Whole Time Director/ Manager during the<br />
financial year.<br />
16. The Company has not appointed any sole selling agents<br />
during the financial year.<br />
17. The Company was not required to obtain any approvals<br />
of the Central Government, Company Law Board,<br />
Regional Director, Registrar of Companies and/or such<br />
authorities prescribed under the various provisions of<br />
the Act.<br />
18. The Directors have disclosed their interest in other<br />
firms/ companies to the Board of Directors pursuant to<br />
the provisions of the Act and the rules made<br />
thereunder.<br />
19. The Company has not issued any shares, debentures or<br />
other securities during the financial year.<br />
20. The Company has not bought back any shares during<br />
the financial year.<br />
21. There was no redemption of preference shares or<br />
debentures during the financial year.<br />
22. There were no transactions necessitating the Company<br />
to keep in abeyance the rights to dividend, rights<br />
shares and bonus shares pending registration of<br />
GM PHARMA LIMITED<br />
transfer of shares.<br />
23. The Company has not invited/ accepted any deposits<br />
including any unsecured loans falling within the purview<br />
of Section 58A during the financial year.<br />
24. The Company has not made any borrowings during the<br />
financial year ended 31st March, 2004.<br />
25. The loans given and investments, or given guarantees<br />
or provided securities to other bodies corporate during<br />
the year are within the preview of the provision of<br />
Section 372A of the Companies Act, 1956.<br />
26. The Company has not altered the provisions of the<br />
Memorandum of Association of with respect to<br />
situation of the Company’s registered office from one<br />
state to another during the year under scrutiny.<br />
27. The Company has not altered the provisions of the<br />
Memorandum of Association with respect to the<br />
objects of the Company during the year under scrutiny.<br />
28. The Company has not altered the provisions of the<br />
Memorandum of Association with respect to name of<br />
the Company during the year under scrutiny.<br />
29. The Company has not altered the provisions of the<br />
Memorandum of Association with respect to share<br />
capital of the Company during the year under scrutiny.<br />
30. The Company has not altered its Articles of Association<br />
during the financial year.<br />
31. There was no prosecution initiated against or show<br />
cause notices received by the Company, during the<br />
financial year, for offences under the Act.<br />
32. The Company has not received any money as security<br />
from its employees during the financial year.<br />
33. The Company has not deducted any contribution<br />
towards Provident Fund during the financial year in<br />
terms of Section 418 of the Companies Act, 1956.<br />
For S. S. RAUTHAN & ASSOCIATES,<br />
Company Secretaries<br />
Surjan Singh Rauthan<br />
Place : Mumbai<br />
Proprietor<br />
Date : July 5, 2004 C.P. No. 3233<br />
123
GM PHARMA LIMITED<br />
GLENMARK IMPEX LLC<br />
Muddanna Anil<br />
Annexure – A<br />
Registers as maintained by the Company<br />
1. Register of members under Section 150 and Index of Members under Section 151.<br />
2. Register of Transfers<br />
3. Register of particulars of contracts in which Directors are interested under Section 301.<br />
4. Register of Directors, Managing Director, Manager and Secretary under Section 303.<br />
5. Register of Director’s shareholding under Section 307.<br />
6. Register of investments or loans made, guarantee given or security provided under Section 372A (w.e.f. 31.10.1998).<br />
Directors’ Report<br />
Your Director is pleased to present the Company’s Third<br />
Annual Report together with the Audited Annual Accounts<br />
of the Company for the period ended 31st March, 2004.<br />
FINANCIAL RESULTS<br />
During the aforesaid period, the Company had no trading<br />
activity and hence there is no operational income.<br />
DIVIDEND<br />
Since there is no operational income, no dividend is<br />
proposed to be declared by the Company.<br />
been appointed General Director in place of Mr. Sudip<br />
Majumdar.<br />
AUDITORS<br />
Auditors of the Company M/s. Allent, an accounting and<br />
law firm, hold office until the conclusion of ensuing Annual<br />
General Meeting and being eligible offer themselves for<br />
re-appointment.<br />
For and on behalf of the Board of Directors<br />
For S. S. RAUTHAN & ASSOCIATES,<br />
Company Secretaries<br />
DIRECTORS<br />
Mr. Sudip Majumdar ceased to be the General Director<br />
with effect from August 1, 2003. Mr. Muddanna Anil has<br />
Date: April 12, 2004<br />
Place: Moscow<br />
Muddanna Anil<br />
General Director<br />
Surjan Singh Rauthan<br />
Place : Mumbai<br />
Proprietor<br />
Date : July 5, 2004 C.P. No. 3233<br />
Annexure – B<br />
Forms and Returns as filed by the Company with Registrar of Companies, Regional Director, Central Government or other<br />
authorities during the financial year ended 31st March 2004.<br />
1. Annual Return – AGM held on 25.09.2003 – Filed u/s 159 on 20/11/2003<br />
2. Balance Sheet & Profit & Loss A/c. – Year Ended 31.03.2003 – Filed u/s 220 on 07/10/2003<br />
3. Compliance Certificate - Year Ended 31.03.2003 - Filed u/s 383A(1) on 07/10/2003<br />
For S. S. RAUTHAN & ASSOCIATES,<br />
Company Secretaries<br />
Surjan Singh Rauthan<br />
Place : Mumbai<br />
Proprietor<br />
Date : July 5, 2004 C.P. No. 3233<br />
Auditor’s Report<br />
Introduction<br />
We have audited the financial statements of <strong>Glenmark</strong><br />
Impex LLC, a limited liability Company for the fiscal periods<br />
from Jan 1, 2004 to March 31, 2004 inclusive. The Head<br />
of <strong>Glenmark</strong> Impex LLC, a limited liability Company is<br />
responsible for the preparation of the financial statements,<br />
while our responsibility is to form an independent opinion<br />
based on the audit of those financial statements as well as<br />
oral explanations of the authorized persons of <strong>Glenmark</strong><br />
Impex LLC, Moscow office.<br />
Basis of opinion<br />
We conducted our audit in accordance with Auditing<br />
Standards issued by Auditing Practices Committee. The<br />
audit includes examination of evidence relevant to the<br />
amounts and disclosures in the financial statements.<br />
We planned and performed our audit so as to obtain all the<br />
information and oral explanations which we considered<br />
necessary in order to provide us with sufficient evidence to<br />
give reasonable assurance that the financial statements are<br />
free from material misstatement, whether caused by fraud<br />
or other irregularity or error. In forming our opinion, we<br />
also evaluated the overall adequacy of the presentation of<br />
information in the financial statements.<br />
Opinion<br />
In our opinion, the financial statements give a true and fair<br />
view of the state of the Company’s affairs as at 31st March<br />
2004 and have been properly prepared.<br />
Place: Moscow<br />
Date: 12th April, 2004<br />
For Allent Accounting & Law firm<br />
Public Accountant<br />
Elena V. Ermakova<br />
General Director<br />
GLENMARK IMPEX LLC<br />
Balance Sheet<br />
Amt in USD<br />
As at 31st March, Schedules 2004 2003<br />
SOURCES OF FUNDS<br />
Share Capital<br />
(100% held by <strong>Glenmark</strong> Pharmaceuticals Ltd, Mumbai. India)<br />
Chartered Capital 5 000.00 5 000.00<br />
Equity Capital 2 58 740.00 2 58 740.00<br />
Total 2 63 740.00 2 63 740.00<br />
APPLICATION OF FUNDS<br />
Fixed Assets 5 371.41 5 371.41<br />
Current Assets, Loans & Advances<br />
Balance with bank – –<br />
Miscellaneous expenditure to the extent not written off<br />
Preoperative expenses 2 58 368.59 2 58 368.59<br />
Total 2 63 740.00 2 63 740.00<br />
As per our report of even date annexed<br />
For Allent Accounting & Law Firm<br />
Public Accountants<br />
For <strong>Glenmark</strong> Impex LLC<br />
Glenn Saldanha<br />
R.V. Desai<br />
Marshall J. Mendonza<br />
Directors’ Report<br />
Your Directors are pleased to present the Company’s First<br />
Annual Report together with the Audited Annual Accounts<br />
of the Company for the period ended 31st March, 2004.<br />
FINANCIAL RESULTS<br />
During the aforesaid period, the Company had not<br />
commenced any activity and hence there is no operational<br />
income.<br />
Your Company hs acquired a local manufacturing Company<br />
having an ANVISA approved plant in Brazil for US$ 5.25<br />
million. The facilities shall be used for manufacturing<br />
formulations. The acquisition will facilitate enhancing<br />
business interests in the domestic markets and entry in the<br />
other Latin American markets.<br />
DIVIDEND<br />
Since there is no operational income, no dividend is<br />
GLENMARK FARMACEUTICA LTDA<br />
proposed to be declared by the Company.<br />
DIRECTORS<br />
The Directors, Mr. Glenn Saldanha, Mr. Rajesh. V. Desai<br />
and Mr. Marshall Mendonza retire at the ensuing<br />
Quotaholders Meeting being eligible offer themselves for<br />
re-appointment.<br />
AUDITORS<br />
Auditors of the Company M/s Repus Auditoria &<br />
Constabilidade Ltda hold office until the conclusion of<br />
ensuing Quotaholders Meeting and being eligible offer<br />
themselves for re-appointment.<br />
For and on behalf of the Board of Directors<br />
Date: April 12, 2004<br />
Place: Sao Paulo<br />
Glenn Saldanha<br />
Chairman<br />
Elena V Ermakova<br />
General Director<br />
Date : 12th April 2004<br />
Muddanna Anil<br />
General Director<br />
Auditor’s Report<br />
124<br />
The Board of Directors<br />
<strong>Glenmark</strong> Farmaceutica Ltda<br />
We have audited the accompanying Balance Sheet of<br />
<strong>Glenmark</strong> Farmaceutica Ltda as of 31st March, 2004. The<br />
financial statements are the responsibility of the company’s<br />
management. Our responsibility is to express an opinion<br />
on the Balance sheet based on the audit.<br />
The audit has been conducted in accordance with generally<br />
accepted auditing standards. Those standards require that<br />
we plan and perform the audit to obtain reasonable<br />
assurance about whether the balance sheet is free of<br />
material misstatements. An audit includes examining on, a<br />
test basis, evidence supporting the amounts and<br />
disclosures in the balance sheet. An audit also includes<br />
assessing the accounting principles used and the<br />
significant estimates made by the management as well as<br />
evaluating the overall financial statement presentation. We<br />
believe our audit provides a reasonable basis for opinion.<br />
In our opinion, the Balance sheet referred to above,<br />
present fairly in all material aspects the financial position of<br />
<strong>Glenmark</strong> Farmaceutica Ltda as of 31st March 2004 in<br />
conformity with generally accepted accounting principles.<br />
As per our report of even date annexed<br />
for REPUS Auditoria & Contabilidade Ltda<br />
CRC 2SP 007.220/0-7<br />
Sergio Henrique da Silva Macedo<br />
Accountant, CRC 1SP 185.764/0-4<br />
Rubens Vilibor,<br />
Accountant, CRC 1SP 048.688/0-7
GLENMARK FARMACEUTICA LTDA<br />
GLENMARK FARMACEUTICA LTDA<br />
Balance Sheet<br />
Reals in {000’s)<br />
As at 31st March, Schedules 2004<br />
SOURCES OF FUNDS<br />
Shareholders' Funds<br />
Share capital 1 1,118.86<br />
Reserves and surplus –<br />
1,118.86<br />
Loan Funds –<br />
Total 1,118.86<br />
APPLICATION OF FUNDS<br />
Fixed Assets 2<br />
Gross Block 418.67<br />
Less : Depreciation –<br />
Net Block 418.67<br />
Investments –<br />
Current Assets, Loans and Advances<br />
Inventories –<br />
Sundry debtors –<br />
Cash and bank balances 3 73.43<br />
Loans and advances 4 59.61<br />
133.04<br />
Less : Current Liabilities and Provisions<br />
Current liabilities 127.12<br />
127.12<br />
Net Current Assets 5.91<br />
Miscellaneous Expenditure 5 694.28<br />
(to the extent not written off or adjusted)<br />
Total 1,118.86<br />
Notes to accounts & significant Accounting Policies 6<br />
Schedules referred to above form an integral part of the Balance Sheet.<br />
As per our report of even date annexed<br />
for REPUS Auditoria & Contabilidade Ltda<br />
CRC 2SP 007.220/0-7<br />
Sergio Henrique da Silva Macedo<br />
Accountant<br />
CRC 1SP 185.764/0-4<br />
Rubens Vilibor<br />
CRC 1SP 048.688/0-7<br />
Schedules forming part of the Balance Sheet<br />
For <strong>Glenmark</strong> Farmaceutical Ltd.<br />
Rajesh Desai<br />
Director<br />
Date: 15 April, 2004<br />
Reals in {000’s)<br />
As at 31st March, 2004<br />
1. SHARE CAPITAL<br />
Authorised<br />
1118857 Equity shares of 1 Reals/- each 1,118.86<br />
Issued, Subscribed and Paid-up<br />
1118857 Equity shares of 1 Reals/- each 1,118.86<br />
Total 1,118.86<br />
2. Fixed Assets<br />
Capital WIP 418.67<br />
Total 418.67<br />
Schedules forming part of the Balance Sheet<br />
Reals in {000’s)<br />
As at 31st March, 2004<br />
3. Cash and Bank Balances<br />
Balances with banks 73.43<br />
Total 73.43<br />
4. Loans and Advances (unsecured, considered good)<br />
Advances recoverable in cash or kind or for value to be received 54.29<br />
Prepaid expenses 5.32<br />
Total 59.61<br />
5. Miscellaneous Expenditure<br />
(to the extent not written off or adjusted)<br />
Preoperative expenses 694.28<br />
Total 694.28<br />
As per our report of even date annexed<br />
For <strong>Glenmark</strong> Farmaceutical Ltd.<br />
for REPUS Auditoria & Contabilidade Ltda<br />
CRC 2SP 007.220/0-7<br />
Sergio Henrique da Silva Macedo<br />
Rajesh Desai<br />
Accountant, CRC 1SP 185.764/0-4<br />
Director<br />
Rubens Vilibor,<br />
Accountant,CRC 1SP 048.688/0-7 Date: 15 April, 2004<br />
Schedules annexed to and forming part of Annual Accounts as at 31st March 2004<br />
6. Notes to Financial Statements<br />
1. Significant Accounting Policies<br />
i) Basis of Accounting<br />
The Financial Statement are prepared under the historical cost convention on the accrual basis of accounting, in<br />
confirmity with the generally accepted accounting principles.<br />
ii) Fixed Assets and Depreciation<br />
No depreciation has been provided as no fixed assets have been capitalised during the period.<br />
iii) Foreign Currency Transaction<br />
Foreign currency transaction will be accounted at the exchange rates prevalent on the dates of the transactions.<br />
Assets and Liabilities will be recorded at the year end rates except for non monerary assets which are accounted<br />
for at the exchange rate prevalent on the date of acquision. Exchange gains and losses on translation will be<br />
charged to the Currency fluctuation Reserve Account.<br />
iv) Segment<br />
In the absence of sales, no segment is determined.<br />
2. Contingent Liabilities<br />
The company is not contingently liable for any amount<br />
3. Related Party Transactions<br />
Transactions with Holding Company include: Reals’ 000<br />
Advances payable to defray operational expenses 72,7250<br />
As per our report of even date annexed<br />
For <strong>Glenmark</strong> Farmaceutical Ltd.<br />
for REPUS Auditoria & Contabilidade Ltda<br />
CRC 2SP 007.220/0-7<br />
Sergio Henrique da Silva Macedo<br />
Rajesh Desai<br />
Accountant, CRC 1SP 185.764/0-4<br />
Director<br />
Rubens Vilibor,<br />
Accountant,CRC 1SP 048.688/0-7 Date: 15 April, 2004<br />
GLENMARK PHARMACEUTICALS INC., USA<br />
GLENMARK PHARMACEUTICALS INC., USA<br />
Glenn Saldanha<br />
Jeffrey Weiss<br />
Cheryl Pinto<br />
Balance Sheet<br />
Amount in USD<br />
As at 31st March, Schedules 2004<br />
ASSETS<br />
Current Assets<br />
Cash 51,275<br />
Employee Loans Receivable 17,187<br />
Due From Affiliated Company 4,559<br />
Total Current Assets 73,021<br />
Property and Equipment, net of accumulated depreciation 181,752<br />
Other Assets<br />
Intangible assets, net of amortization 32,992<br />
Product Development 1,936,521<br />
Intellectual Property 457,252<br />
Security Deposit 8,790<br />
Deferred Tax Assets 103,679<br />
Total Other Assets 2,539,234<br />
Total Assets 2,794,007<br />
LIABILITIES & SHAREHOLDERS' EQUITY<br />
Current Liabilities<br />
Trade Accounts Payable 437,183<br />
Total Liabilities 437,183<br />
Equity<br />
Retained Earnings (143,176)<br />
Shareholders' Equity<br />
Common Stock, ($1 par value, 2,500,000<br />
shares issued and outstanding) 2,500,000<br />
Total Shareholders' Equity 2,356,824<br />
Total Liabilities & Shareholders' Equity $2,794,007<br />
Directors’ Report<br />
Your Directors are pleased to present their First Annual<br />
Report together with the Audited Annual Accounts of your<br />
Company for the period ended 31st March, 2004.<br />
FINANCIAL RESULTS<br />
During the financial period your Company set-up operations<br />
at New Jersey, USA to essentially register and market the<br />
products of its parent Company viz. <strong>Glenmark</strong><br />
Pharmaceuticals Limited.<br />
Your Company has entered into a product development and<br />
marketing license agreement with K. V. Pharmaceuticals<br />
Company (KV). Your Company under the terms of the<br />
agreement will initially develop and license to KV generic<br />
products for regulation approval and marketing in the North<br />
American Market. Your Company expects to launch the<br />
first product under the agreement during the last half of<br />
2005.<br />
DIVIDEND<br />
Since there is no operational income, no dividend is<br />
Auditor’s Report<br />
To the Board of Directors<br />
<strong>Glenmark</strong> Pharmaceuticals Inc., USA<br />
Princeton, New Jersey 08540<br />
I have audited the accompanying balance sheet of<br />
<strong>Glenmark</strong> Pharmaceuticals Inc., USA as of March 31, 2004,<br />
and the related statements of income, retained earnings,<br />
and cash flows for the year then ended. These financial<br />
statements are the responsibility of the Company’s<br />
management. My responsibility is to express an opinion<br />
on these financial statements based on my audit.<br />
I conducted my audit in accordance with auditing standards<br />
generally accepted in the United States of America. Those<br />
standards require that I plan and perform the audit to<br />
obtain reasonable assurance about whether the financial<br />
statements are free of material misstatement. An audit<br />
includes examining, on a test basis, evidence supporting<br />
the amounts and disclosures in the financial statements.<br />
proposed by the Directors.<br />
DIRECTORS<br />
Mr. Sameer Paigankar ceased to be a Director with effect<br />
from March 29, 2004. Ms. Cheryl Pinto was appointed as<br />
Director on April 1, 2004. The Directors, Mr. Glenn<br />
Saldanha, Mr. Jeffrey Weiss and Ms. Cheryl Pinto being<br />
eligible have offered themselves for re-appointed.<br />
AUDITORS<br />
M/s Arthur DeDominicis & Company, were appointed as<br />
the First Auditors of the Company by the Board to hold<br />
office upto the date of the ensuing Annual General<br />
Meeting, being eligible, have offered themselves for<br />
re-appointment.<br />
For and on behalf of the Board of<br />
Date: May 7, 2004<br />
Place: Princeton, New Jersey<br />
Glenn Saldanha<br />
Chairman<br />
An audit also includes assessing the accounting principles<br />
used and significant estimates made by management, as<br />
well as evaluating the overall financial statement<br />
presentation. I believe that my audit provides a reasonable<br />
basis for my opinion.<br />
In my opinion, the financial statements referred to above<br />
present fairly, in all material respects, the financial position<br />
of <strong>Glenmark</strong> Pharmaceuticals Inc., USA as of March 31,<br />
2004, and the results of its operations and its cash flows<br />
for the year then ended in conformity with accounting<br />
principles generally accepted in the United States of<br />
America.<br />
Arthur DeDominicis, CPA<br />
May 7, 2004<br />
Beacon, New York<br />
Arthur De Dominicis CPA<br />
May 7, 2004<br />
Beacon, New York<br />
125
GLENMARK PHARMACEUTICALS INC., USA<br />
GLENMARK PHARMACEUTICALS INC., USA<br />
Statement of Income and Retained Deficit<br />
Amount in USD<br />
As at 31st March, 2004<br />
COST AND EXPENSES<br />
General and Administrative 78,962<br />
Occupancy 72,587<br />
Depreciation & Amortization 53,686<br />
Utilities 42,797<br />
Total Expenses 248,032<br />
Loss From Operations (248,032)<br />
Other Income<br />
Interest Income 1,958<br />
Loss Before IncomeTaxes (246,074)<br />
Provision for Income Taxes (Note E) (102,898)<br />
Net Loss (143,176)<br />
Retained Deficit - Begining –<br />
Retained Deficit - Ending $ (143,176)<br />
Cash Flow Statements<br />
CASH FLOWS FROM OPERATING ACTIVITIES:<br />
Net loss $ (143,176)<br />
Adjustments to reconcile net loss to net cash provided by operating activities:<br />
Depreciation and amortization 53,686<br />
Changes in assets and liabilities:<br />
(Increase)/Decrease in security deposits (8,790)<br />
(Increase)/Decrease due from affiliate (4,559)<br />
(Increase)/Decrease in employee loan receivable (17,187)<br />
(Increase)/Decrease in intangible assets (41,240)<br />
(Increase)/Decrease in product development (1,936,521)<br />
(Increase)/Decrease in intellectual property (457,252)<br />
(Increase)/Decrease in deferred tax assets (103,679)<br />
Increase/(Decrease) in accounts payable 437,183<br />
Total Adjustments (2,078,359)<br />
Net Cash Used in Operating Activities (2,221,535)<br />
Cash Flows From Investing Activities:<br />
Increase in building improvements, equipment, furniture and fixtures (227,190)<br />
Net Cash Used in Investing Activities (227,190)<br />
Cash Flows From Financing Activities:<br />
Investment by parent company 2,500,000<br />
Net Cash Provided by Financing Activities 2,500,000<br />
Net Increase in Cash 51,275<br />
Cash - Beginning of Year –<br />
Cash - End of Year $ 51,275<br />
Supplemental Information:<br />
Income taxes paid $ 781<br />
Interest paid –<br />
Note A - Summary of Significant Accounting Policies<br />
Nature of Operations<br />
The Company is a subsidiary of <strong>Glenmark</strong> Pharmaceuticals Ltd. (GPL), an Indian company. GPL holds 96% of the<br />
outstanding shares. The Company will manufacture and distribute both generic and brand drugs to Borth America through<br />
alliances and agreements with other pharmaceutical companies. With strong ties to the Parent company in India, the<br />
Company will have unlimited resources in both research and development. The Company expects to launch several generic<br />
drugs over the next 12 to 16 months.<br />
Property and Equipment<br />
Property, plant and equipment are recorded at cost and depreciation is provided over their estimated useful lives on a<br />
straight-line basis:<br />
The useful lives of property and equipment for purposes of computing depreciation are:<br />
Year<br />
Equipment 5<br />
Furniture & Fixtures 5<br />
Expenditures for major renewals and betterments that extend the useful lives of property and equipment are capitalized.<br />
Expenditures for maintenance and repairs are charged to expense as incurred.<br />
Intangibles<br />
Organizational costs incurred during the year is amortized over 5 years.<br />
Use of Estimates<br />
The preparation of financial statements in conformity with generally accepted accounting principals in the United States of<br />
America requires management to make estimates and assumptions that affect certain reported amounts and disclosures.<br />
Accordingly, actual results could differ from those estimates.<br />
Note B – Related Party Transactions<br />
The Company has a receivable due from <strong>Glenmark</strong> Pharmaceutical Ltd, the Parent company. Advances are non-interest<br />
bearing and due on demand.<br />
Note C – Property and Equipment<br />
Property and Equipment are as follows at March 31, 2004:<br />
Furniture and Fixtures $ 37,590<br />
Equipment 189,600<br />
Total 227,190<br />
Less accumulated depreciation 45,438<br />
Net property and equipment $ 181,752<br />
Depreciation expense for the year ended March 31, 2004 was $ 45,438.<br />
Note D – Intangible Assets<br />
Intangibles are as follows at March, 31 2004:<br />
Organizational cost $ 41,240<br />
Less accumulated amortization 8,248<br />
Net intangibles $ 32,992<br />
Estimated annual amortization expense at March, 31 2004:<br />
Fiscal Year Ended<br />
03/31/05 $ 8,248<br />
03/31/06 8,248<br />
03/31/07 8,248<br />
03/31/08 8,248<br />
03/31/09 –<br />
GLENMARK PHARMACEUTICALS INC., USA<br />
Note E – Provision for Income Taxes<br />
The Company has available at march 31, 2004, unused operating loss carryforwards of $246,855, which may be applied<br />
against future taxable income.<br />
The provision for Income Taxes for the year ended March 31, 2004 consists of the following:<br />
Federal $ –<br />
State 781<br />
Total Current Portion 781<br />
Deferred income Taxes (benefits)<br />
Federal (83,931)<br />
state (19,748)<br />
Total Deferred Taxes (103,679)<br />
Total Tax Provision $ (102,898)<br />
Note F – Capital Stock<br />
The Company has 3,700,000 shares of Class A Common Voting Stock at a par value of $1; 2,500,000 shares are issued and<br />
outstanding at March 31, 2004.<br />
The Company has 300,000 shares of Class B Common Non-Voting Stock at a par value of $1; no shares are issued and<br />
outsnding at March 31, 2004.<br />
Note G – Preferred Stock<br />
The Company has 1,000,000 shares of Preferred Stock at a par value of $1, no shares are issued and outstanding at March<br />
31, 2004.<br />
126
Glenn Saldanha<br />
Marshall J. Mendonza<br />
Sangeet Meherotra<br />
Venay Panemanglor<br />
Celia S. Panemanglor<br />
Directors’ Report<br />
Your Director are pleased to present the Company’s First<br />
Annual Report together with the Audited Annual Accounts<br />
of the Company for the period ended 31st March, 2004.<br />
FINANCIAL RESULTS<br />
The Company is in the early stages of setting-up business<br />
operation and hence there us no operating income, during<br />
the aforesaid period.<br />
DIVIDEND<br />
Since there is no operational income, no dividend is<br />
proposed to be declared by the Directors.<br />
DIRECTORS<br />
The Directors, Mr. Glenn Saldanha, Mr. Marshall<br />
Mendonza, Mr. Sangeet Mehrotra, Mr. Vinay Panemanglor,<br />
Auditor’s Report<br />
THE BOARD OF DIRECTORS<br />
GLENMARK PHILIPPINES INC. 2609<br />
Prestige Tower<br />
Condominium Emerald Avenue,<br />
Ortigas Center Pasig City, MM,<br />
Philippines<br />
I have audited the accompanying Balance Sheet of<br />
<strong>Glenmark</strong> Philippines, Inc as of March 31, 2004. The<br />
financial statements are the responsibility of the company's<br />
management. My responsibility is to express an opinion on<br />
the Balance Sheet based on my audit.<br />
I conducted my audit in accordance with generally<br />
accepted auditing standards. Those standards require that I<br />
plan and perform the audit to obtain reasonable assurance<br />
about whether the financial statements are free of material<br />
misstatement. An audit includes examining, on a test<br />
basis, evidence supporting the amounts and disclosures in<br />
the financial statements. An audit also includes assessing<br />
the accounting principles used and significant estimates<br />
made by management, as welt as evaluating the overall<br />
GLENMARK PHILIPPINES INC.<br />
and Ms. Celia S. Panemanglor as per the bylaws of the<br />
Company, retire at the ensuing Annual General Meeting<br />
and being eligible, offer themselves for re-appointment.<br />
AUDITORS<br />
Audditors of the Company, M/s Straightline Accounting &<br />
Consulting hold office until the conclusion of the ensuing<br />
Annual General Meeting and being eligible have offered<br />
themselves for re-appointment.<br />
For and on behalf of the Board of Directors<br />
Date: April 12, 2004<br />
Place: Manila<br />
Glenn Saldanha<br />
Chairman<br />
financial statement presentation. I believe my audit<br />
provides a reasonable basis for my opinion.<br />
In my opinion, the Balance Sheet referred to above present<br />
fairly, in all material respects, the financial position of<br />
<strong>Glenmark</strong> Philippines, Inc. as of March 31, 2004 in<br />
conformity with generally accepted accounting principles.<br />
Edgar C. Sanchez<br />
CPA Cert. No. 78923, PTR No. 5369596<br />
Quezon City, Philippines<br />
Makati City, Philippines<br />
April 12, 2004<br />
Home Office: 5 Harvard St.,<br />
St. Ignatius Village, Quezon City, Philippines.<br />
Tel. No. 911-1761<br />
GLENMARK PHILIPPINES INC.<br />
Balance Sheet<br />
Pesos in ,000<br />
As at 31st March, Schedules 2004<br />
SOURCES OF FUNDS<br />
Shareholders' Funds<br />
Share capital 1 11,540.26<br />
Reserves and surplus –<br />
11,540.26<br />
Loan Funds –<br />
Total 11,540.26<br />
APPLICATION OF FUNDS<br />
Fixed Assets 2<br />
Gross Block 780.55<br />
Less : Depreciation –<br />
Net Block 780.55<br />
Investments<br />
Current Assets, Loans and Advances<br />
Inventories –<br />
Sundry debtors –<br />
Cash and bank balances 3 9,966.41<br />
Loans and advances 4 218.71<br />
10,185.12<br />
Less : Current Liabilities and Provisions<br />
Current liabilities –<br />
Net Current Assets 10,185.12<br />
Miscellaneous Expenditure 5 574.59<br />
(to the extent not written off or adjusted)<br />
Total 11,540.26<br />
Notes to Accounts and Significant Accounting Policies 6<br />
Schedules referred to above form an integral part of the Balance Sheet.<br />
As per our report of even date annexed<br />
For Straightline Accounting & Consulting<br />
Edgar C. Sanchez<br />
CPA Cert. No. 78923<br />
PTR 8369596<br />
Issued in Quezon City, Philippines<br />
Date: 12 April, 2004<br />
For <strong>Glenmark</strong> Philippines Inc.<br />
Sangeet Mehrotra<br />
Director<br />
GLENMARK PHILIPPINES INC.<br />
Schedules forming part of the Balance Sheet<br />
Pesos in ,000<br />
As at 31st March, 2004<br />
1. Share Capital<br />
Authorised<br />
56000 Equity shares of 200 Pesos/- each 11,200.00<br />
Issued, Subscribed and Paid-up<br />
56000 Equity shares of 200 Pesos/- each 11,200.00<br />
Share Application money 340.26<br />
Total 11,540.26<br />
2. Fixed Assets<br />
Capital WIP 780.55<br />
Total 780.55<br />
3. Cash and Bank Balances<br />
Cash in hand 49.25<br />
Balances with banks –<br />
USD account (Bal. USD 154978) 8,725.26<br />
Peso account 1,191.90<br />
Total 9,966.41<br />
4. Loans and Advances (unsecured, considered good)<br />
Advances recoverable in cash or kind or for value to be received 218.71<br />
Total 218.71<br />
5. Miscellaneous Expenditure<br />
(to the extent not written off or adjusted)<br />
Preoperative expenses 574.59<br />
Total 574.59<br />
6. Notes to Financial Statements<br />
1. Significant Accounting Policies<br />
i) Basis of Accounting<br />
The Financial Statements are prepared under the historical cost convention, on the accrual basis of accounting, in<br />
confirmity with the generally accepted accounting principles.<br />
ii) Fixed Assets and Depreciation<br />
No depreciation has been provided as no fixed assets have been capitalised during the period.<br />
iii) Foreign Currency Transaction<br />
Foreign currency transactions will be accounted at the exchange rates prevalent on the dates of the transactions.<br />
Assets and Liabilities will be recorded at the year end rates except for non monetary assets which are accounted<br />
for at the exchange rate prevalent on the date of acquisition. Exchange gains and losses on translation will be<br />
charged to the Currency fluctuation Reserve Account.<br />
iv) Segment<br />
In the absence of sales, no segment is determined.<br />
2. Contingent Liabilities<br />
The company is not contingently liable for any amount.<br />
127
Management is about<br />
doing things right,<br />
leadership is doing<br />
the right things.<br />
– Peter F. Drucker<br />
128
<strong>Glenmark</strong> Pharmaceuticals Limited<br />
Introduction<br />
<strong>Glenmark</strong> Pharmaceuticals Limited<br />
(<strong>Glenmark</strong>) is an India-based growing<br />
pharmaceuticals company<br />
headquartered at Mumbai.<br />
Incorporated in 1977, the company is<br />
focused on the manufacture and<br />
marketing of branded formulation<br />
products and active pharmaceutical<br />
ingredients. In addition to a strong<br />
Indian franchise, <strong>Glenmark</strong> enjoys a<br />
diversified and growing presence in<br />
regulated and developing international<br />
markets. Over the years, the<br />
company has also catalysed its<br />
growth through investment in<br />
dedicated research and development<br />
teams. Separate teams focus on<br />
process reverse - engineering drugs<br />
for launch across markets and<br />
research in novel drug delivery<br />
systems. In addition, a future-focused<br />
team is presently engaged in the<br />
discovery of new chemical entities<br />
that will translate into wholly new<br />
drugs for a global launch.<br />
Brands<br />
In India, <strong>Glenmark</strong> enjoys a visible and<br />
growing branded formulations<br />
presence. A number of its products<br />
have emerged as brand leaders:<br />
Ascoril, Candid-B and Altacef, three of<br />
<strong>Glenmark</strong>’s top-selling brands, are<br />
ranked high in the community of the<br />
most successful pharmaceutical<br />
brands of India, and are also leaders<br />
in their individual therapeutic baskets.<br />
The success is not only limited to<br />
older brands - several of <strong>Glenmark</strong>’s<br />
recent launches such as Valus and<br />
Vorth have already achieved<br />
leadership position in their respective<br />
segments. <strong>Glenmark</strong>’s brands are<br />
actively promoted by its large field<br />
force, reinforced by focussed support<br />
that is provided through the<br />
company’s three marketing divisions.<br />
Manufacturing<br />
Over the years, <strong>Glenmark</strong><br />
strengthened its integration across<br />
the pharmaceutical value-chain<br />
through operations across four<br />
manufacturing plants:<br />
Formulations: Nasik and Goa,<br />
APIs: Kurkumbh and Ankleshwar.<br />
The Ankhleshwar plan acquired from<br />
GlaxoSmithKline has now been<br />
upgraded to meet with USFDA<br />
certification. Another API<br />
manufacturing facility has been<br />
recently acquired at Solapur. The<br />
company has also recently<br />
commissioned a state-of-the-art<br />
formulations plant at Goa, built in<br />
stringent compliance with USFDA and<br />
other regulated market standards.<br />
Research and<br />
development<br />
Research lies at the heart of<br />
<strong>Glenmark</strong>’s existence. Over the years,<br />
the company has invested in a stateof-art<br />
R&D centre in Mahape on the<br />
outskirts of Mumbai. This R&D centre<br />
employs over 250 scientists who<br />
possess a vast experience across<br />
several disciplines and technology<br />
platforms. The company’s research<br />
encompasses new chemical entity<br />
research (NCE) and novel drug<br />
delivery systems (NDDS). This IPRgenerating<br />
activity is supplemented<br />
by dedicated teams that are engaged<br />
in process engineering of bulk drugs<br />
and developing formulations for<br />
launch in regulated markets. Another<br />
team situated at an exclusive facility<br />
at Sinnar focuses on the formulation<br />
of drugs to be launched in India and<br />
across semi-regulated markets.<br />
Revenues<br />
<strong>Glenmark</strong>’s revenues were Rs<br />
3806.60 million in 2003-04 compared<br />
to Rs 3336.40 million in 2002-03, a<br />
growth of 14.10 per cent. Its profit<br />
after tax grew from Rs 331.90 million<br />
in 2002-03 to Rs 420.04 million in<br />
2003-04, a growth of 27 per cent.
Corporate Information<br />
CHAIRMAN<br />
Gracias Saldanha<br />
MANAGING DIRECTOR & CEO<br />
Glenn Saldanha<br />
DIRECTORS<br />
B E Saldanha<br />
Cheryl Pinto<br />
J F Riberio<br />
Dr. Prasanna Gore<br />
R V Desai<br />
A S Mohanty<br />
J M Trivedi<br />
Sridhar Gorthi (Alternate Director to Dr. Prasanna Gore)<br />
M Gopal Krishnan<br />
Natvarlal B. Desai<br />
Steven Bates<br />
COMPANY SECRETARY<br />
Marshall Mendonza<br />
REGISTERED OFFICE<br />
B/2 Mahalaxmi Chambers<br />
22, Bhulabhai Desai Road<br />
Mumbai 400 026<br />
Tel: 496 4893-96<br />
Fax 022-493 2648<br />
Website: http://www.glenmarkpharma.com<br />
E-mail: webmaster@glenmarkpharma.com<br />
WORKS<br />
• E-37, MIDC Industria Area, D-Road, Satpur,<br />
Nasik 422 007, Maharashtra.<br />
• 3109-C, GIDC Industrial Estate, Ankleshwar,<br />
Dist. Bharuch, Gujarat 393 002.<br />
• Plot No. 163-165/170-172, Chandramouli Industrial<br />
Estate, Mohol, Mohal Bazarpeth, Sholapur,<br />
Maharashtra - 413213.<br />
• Plot No. A-80, MIDC Area, Kurkumbh, Daund,<br />
Pune 413 802, Maharashtra.<br />
• Plot No. 7, Colvale Industrial Estate, Bardez, Goa.<br />
R&D CENTRE<br />
• Plot No. C-152, MIDC Sinnar Industrial Area,<br />
Malegaon, Nasik District 421 103<br />
Maharashtra.<br />
• Plot No. A-607, TTC Industrial Area, MIDC,<br />
Mahape, Vashi, Navi Mumbai - 400 705<br />
Maharashtra.<br />
AUDITORS<br />
Pricewater House<br />
Chartered Accountants<br />
Mumbai.<br />
COST AUDITORS<br />
Sevekari, Maniar & Associates<br />
Cost Accounts<br />
Mumbai<br />
SOLICITOR<br />
Kanga & Co., Mumbai,<br />
Trilegal, Mumbai<br />
REGISTRAR AND TRANSFER AGENTS<br />
Karvy Computershare Pvt. Limited<br />
“Karvy House”, 46 Avenue 4, Street No. 1,<br />
Banjara Hills, Hyderabad 500 034<br />
BANKERS<br />
Bank of India<br />
Mahalaxmi Branch, Mumbai 400 026
MANUFACTURING SITE:<br />
INTERNATIONAL OPERATION (FORMULATIONS):<br />
India<br />
Brazil<br />
REGIONAL OFFICES :<br />
1. Brazil<br />
2. Kanya<br />
3. Malaysia<br />
4. Nigeria<br />
5. Philippines<br />
6. Russia<br />
7. South Africa<br />
8. USA<br />
9. UK<br />
10. Viatnam<br />
11. Afghanistan<br />
12. Angola<br />
13. Belarus<br />
14. Benin<br />
15. Botswana<br />
16. Brunei<br />
Darussalam<br />
17. Burkina Faso<br />
18. Cambodia<br />
19. Chad<br />
20. Congo Brazzaville<br />
21. Eritrea<br />
22. Ethiopia<br />
23. Fiji<br />
24. Ghana<br />
25. Hong Kong<br />
26. Indonesia<br />
27. Iraq<br />
28. Ivory Coast<br />
29. Kazakhstan<br />
30. Laos<br />
31. Latvia<br />
32. Madagascar<br />
33. Malawi<br />
34. Maldives<br />
35. Mali<br />
36. Mauritius<br />
37. Mozambique<br />
38. Myanmar<br />
39. Namibia<br />
40. Nepal<br />
41. Oman<br />
42. Papua New<br />
Guinea<br />
43. Peru<br />
44. Portugal<br />
45. R D Cango<br />
46. Rwanda<br />
47. Senegal<br />
48. Singapore<br />
49. Sri Lanka<br />
50. Sudan<br />
51. Tanzania<br />
52. Thailand<br />
53. Togo<br />
54. Trinidad &<br />
Tabago<br />
55. UAE<br />
56. Uganda<br />
57. Ukraine<br />
58. Venezuela<br />
59. Yemen<br />
60. Zambia<br />
61. Zimbabwe<br />
www.glenmarkpharma.com




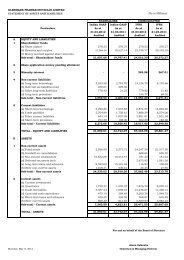

![Formulations [India] â Product List - Glenmark](https://img.yumpu.com/46601329/1/190x245/formulations-india-a-product-list-glenmark.jpg?quality=85)
![Formulation [India] â Product List - Glenmark](https://img.yumpu.com/44013338/1/190x245/formulation-india-a-product-list-glenmark.jpg?quality=85)




![Formulations [India] â Product List - Glenmark](https://img.yumpu.com/35994839/1/190x245/formulations-india-a-product-list-glenmark.jpg?quality=85)

