Quantum physics (quantum theory, quantum mechanics)
Quantum physics (quantum theory, quantum mechanics)
Quantum physics (quantum theory, quantum mechanics)
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
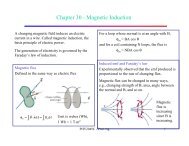
Splitting of atomic energy levels<br />
B 0 B 0<br />
m = +1<br />
m = 0<br />
m = -1<br />
(2l+1) states with same<br />
energy: m=-l,…+l<br />
B ≠ 0: (2l+1) states with<br />
distinct energies<br />
Predictions: should always get an odd number of<br />
levels. An s state (such as the ground state of<br />
hydrogen, n=1, l=0, m=0) should not be split.<br />
Splitting was observed by Zeeman<br />
(Hence the name<br />
“magnetic <strong>quantum</strong><br />
number” for m.)<br />
37