Periodic Table
Periodic Table
Periodic Table
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
Atomic History & Structure and <strong>Periodic</strong> <strong>Table</strong><br />
Practice Problems:<br />
1. Calculate the Average Atomic Mass<br />
Mass Abundance<br />
Isotope 1 80 amu 30%<br />
Isotope 2 82 amu 30%<br />
Isotope 3 84 amu 40%<br />
Average Atomic Mass = (Mass)(Abundance) + (Mass)(Abundance) + (Mass)(Abundance)<br />
Average Atomic Mass = (80 amu)(0.3) + (82 amu)(0.3) + (84 amu)(0.4)<br />
Average Mass: 82.2 amu<br />
2. There are 2 naturally occurring isotopes of boron. Calculate the abundance of B-10 and B-11.<br />
Average atomic mass of Boron = 10.8 amu<br />
Average Atomic Mass = (Mass)(Abundance) + (Mass)(Abundance)<br />
10.8 = (10 )(x) + (11)(1-x)<br />
10.8 = 10x + 11 – 11x<br />
0.2 = x<br />
B-10 20% abundant<br />
B-11 80% abundant<br />
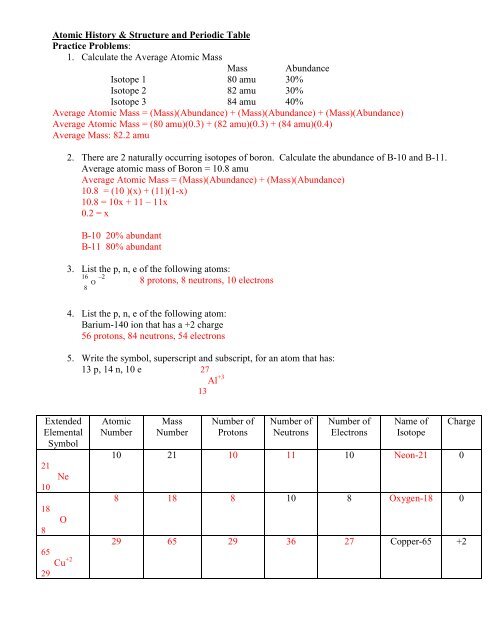
3. List the p, n, e of the following atoms:<br />
16<br />
O –2 8 protons, 8 neutrons, 10 electrons<br />
8<br />
4. List the p, n, e of the following atom:<br />
Barium-140 ion that has a +2 charge<br />
56 protons, 84 neutrons, 54 electrons<br />
5. Write the symbol, superscript and subscript, for an atom that has:<br />
13 p, 14 n, 10 e 27<br />
+3<br />
Al<br />
13<br />
Extended<br />
Elemental<br />
Symbol<br />
21<br />
10<br />
18<br />
8<br />
65<br />
29<br />
Ne<br />
O<br />
Cu<br />
+2<br />
Atomic<br />
Number<br />
Mass<br />
Number<br />
Number of<br />
Protons<br />
Number of<br />
Neutrons<br />
Number of<br />
Electrons<br />
Name of<br />
Isotope<br />
10 21 10 11 10 Neon-21 0<br />
8 18 8 10 8 Oxygen-18 0<br />
Charge<br />
29 65 29 36 27 Copper-65 +2
6. Does lithium have a larger value than potassium for:<br />
a. First ionization energy - YES<br />
b. Atomic radius - NO<br />
c. Electronegativity - YES<br />
d. Ionic radius - NO<br />
7. Arrange these elements in order of decreasing atomic size: sulfur, chlorine, aluminum, and<br />
sodium.<br />
Sodium > Aluminum > Sulfur > Chlorine<br />
8. Which element in each pair has the larger ionization energy<br />
a. sodium, potassium<br />
b. magnesium, phosphorus<br />
9. Which particle has the larger radius in each atom/ion pair?<br />
a. Na , Na +<br />
b. S , S -2<br />
10. Which element is more electronegative?<br />
a. Cl , F<br />
b. Mg , Ne *Most noble gases do not have electronegativity values<br />
c. As , Ca<br />
11. In each pair, which ion is larger?<br />
a. Ca +2 , Mg +2<br />
b. Cl -1 , P -3<br />
12. Name the family that (has):<br />
a. The highest ionization energies of any of the families - NOBLE GASES<br />
b. The largest atomic radii of any of the families - ALKALI METALS<br />
c. Gain 1 electron to fill its octet - HALOGENS<br />
d. Two valence electrons - ALKALINE EARTH METALS
13. Which group of elements is brittle, but can conduct electricity?<br />
METALLOIDS<br />
14. Of potassium, germanium, and bromine, which element loses electrons the easiest?<br />
Potassium – it has the lowest ionization energy<br />
15. Predict the melting point for potassium given the following melting points:<br />
Li = 181 o C, Na = 98 o C, Rb = 39 o C, Cs = 28 o C<br />
Li = 181 o C, Na = 98 o C, K = 63 o C, Rb = 39 o C, Cs = 28 o C<br />
(potassium is between Na and Rb on the periodic table, so the melting point needs to be<br />
between sodium’s and rubidium’s)