Conclusion Get more connected.
Conclusion Get more connected.
Conclusion Get more connected.
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
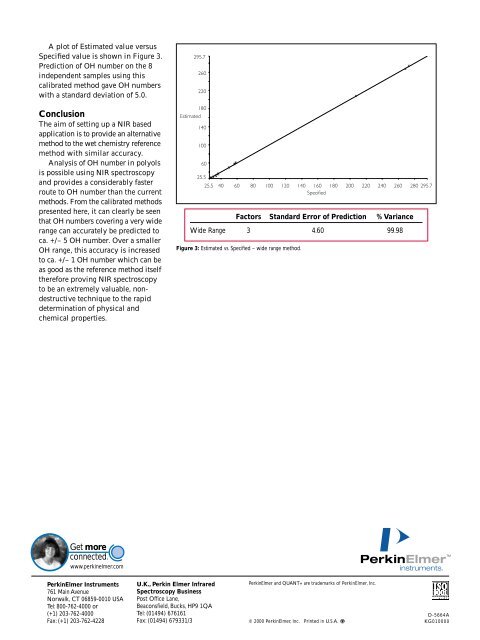
A plot of Estimated value versus<br />
Specified value is shown in Figure 3.<br />
Prediction of OH number on the 8<br />
independent samples using this<br />
calibrated method gave OH numbers<br />
with a standard deviation of 5.0.<br />
<strong>Conclusion</strong><br />
The aim of setting up a NIR based<br />
application is to provide an alternative<br />
method to the wet chemistry reference<br />
method with similar accuracy.<br />
Analysis of OH number in polyols<br />
is possible using NIR spectroscopy<br />
and provides a considerably faster<br />
route to OH number than the current<br />
methods. From the calibrated methods<br />
presented here, it can clearly be seen<br />
that OH numbers covering a very wide<br />
range can accurately be predicted to<br />
ca. +/– 5 OH number. Over a smaller<br />
OH range, this accuracy is increased<br />
to ca. +/– 1 OH number which can be<br />
as good as the reference method itself<br />
therefore proving NIR spectroscopy<br />
to be an extremely valuable, nondestructive<br />
technique to the rapid<br />
determination of physical and<br />
chemical properties.<br />
Factors Standard Error of Prediction % Variance<br />
Wide Range 3 4.60 99.98<br />
Figure 3: Estimated vs. Specified – wide range method.<br />
<strong>Get</strong> <strong>more</strong><br />
<strong>connected</strong>.<br />
www.perkinelmer.com<br />
PerkinElmer Instruments<br />
761 Main Avenue<br />
Norwalk, CT 06859-0010 USA<br />
Tel: 800-762-4000 or<br />
(+1) 203-762-4000<br />
Fax: (+1) 203-762-4228<br />
U.K., Perkin Elmer Infrared<br />
PerkinElmer and QUANT+ are trademarks of PerkinElmer, Inc.<br />
Spectroscopy Business<br />
Post Office Lane,<br />
Beaconsfield, Bucks, HP9 1QA<br />
Tel: (01494) 676161<br />
Fax: (01494) 679331/3 © 2000 PerkinElmer, Inc. Printed in U.S.A.<br />
D-5664A<br />
KG010000
FT-NIR SPECTROSCOPY<br />
application note<br />
The Determination of OH Number in<br />
Polyols Using FT-NIR Spectroscopy<br />
Summary<br />
NIR spectroscopy is an invaluable tool<br />
for the quantitative analysis of a wide<br />
range of chemical compounds. In<br />
combination with a number of<br />
chemometric methods, the technique<br />
provides a fast, non-destructive route<br />
to the determination of physical and<br />
chemical properties.<br />
This note describes the use of<br />
FT-NIR spectroscopy in a typical<br />
polyol analysis with an outline of<br />
the development of the quantitative<br />
method used for the application.<br />
Introduction<br />
Polyols are long-chain polymers<br />
which contain alcohol functional<br />
groups and are produced via<br />
reactions involving organic oxides,<br />
acids and multi-functional alcohols.<br />
A wide range of products<br />
including, surfactants, foams, paint<br />
additives, and adhesives are<br />
manufactured using polyols. The<br />
production of polyurethanes, for<br />
instance, involves polyol<br />
intermediates.<br />
Polyol products are typically<br />
produced via batch reactor processes<br />
held at high temperatures (>250˚C).<br />
Current analysis of the polyols<br />
produced normally take the form of<br />
back titrations. The resultant OH<br />
number is an average value over a<br />
number of titrations.<br />
This analysis generally takes<br />
place in an off-site laboratory, often<br />
far removed from the conditions<br />
present at the production site. The<br />
sample is extracted from the bulk,<br />
and the production process halted<br />
while the analysis is completed; a<br />
process that can take several hours.<br />
Care must be taken in handling the<br />
chemicals involved, especially when<br />
analyzing samples at elevated<br />
temperatures. The possibility of<br />
moisture being absorbed from the air<br />
is highly detrimental to the analysis<br />
of the OH value. A system whereby<br />
the sample must be heated is applied<br />
if the sample is a solid or highly<br />
viscous liquid. Even when all these<br />
factors have been minimized or<br />
taken into account, the process still<br />
depends on the analyst. The current<br />
method for determining OH number<br />
is expensive, time consuming, and<br />
prone to human error.<br />
A typical NIR based application<br />
is faster, <strong>more</strong> precise and reliable<br />
than other methods. It also reduces<br />
the need to handle potentially<br />
hazardous substances. The bands in<br />
the NIR region (ca. 15000 – 3000<br />
cm-1, 667 – 3333 nm) are primarily<br />
overtones and combination bands<br />
normally associated with C-H, N-H<br />
and O-H bonds. Since organic<br />
polymers are composed of carbon,<br />
hydrogen, nitrogen and oxygen<br />
atoms, the NIR spectra of polymers<br />
feature sharp, strong absorbance<br />
bands.<br />
Polymer manufacturers can use<br />
NIR spectroscopy to perform<br />
analyses on the production site.<br />
This has obvious advantages in the<br />
area of quality assurance. NIR<br />
analysis is also capable of process<br />
control as the analysis of<br />
intermediates occurs in real-time.<br />
Since many polyol reactions are<br />
irreversible, the analysis of such<br />
intermediates by NIR spectroscopy<br />
provides a commercially valuable<br />
tool.<br />
A typical NIR method<br />
development for OH determination<br />
consists of first assembling a set of<br />
preanalyzed samples which must<br />
span the OH range for which the<br />
calibration is intended. The number<br />
of samples used depends on the OH<br />
range and accuracy required, but for<br />
most applications in the range ca.<br />
20 – 200 OH approximately 20<br />
samples are adequate for calibration.
Once a set of reference samples is<br />
obtained, it is advisable to divide these<br />
into two sets; a set for calibration, and<br />
a set to test the calibration often<br />
referred to as the validation set. The<br />
suitability of the samples used in the<br />
calibration set is particularly important<br />
since it must closely match the<br />
product being manufactured at the<br />
production site. A calibration set must<br />
contain enough samples to enable the<br />
estimation of the calibration constants,<br />
and the range of the samples should<br />
be representative of the range of<br />
future samples.<br />
A calibration, based on ‘ideal’<br />
samples prepared in a completely<br />
stable environment, may give very<br />
small errors when used for similar<br />
samples. However, when the<br />
calibration is used to produce a value<br />
for a real product, the accuracy is<br />
often degraded. Further<strong>more</strong>, a tradeoff<br />
exists between calibration sets<br />
covering a very wide range of OH<br />
number and the accuracy with which<br />
OH number can be predicted.<br />
Therefore, it has been found that the<br />
accuracy increases when using a<br />
method calibrated over a <strong>more</strong><br />
restricted (ca. 50) OH range.<br />
The PLS (Partial Least Squares)<br />
method available in the Perkin-Elmer<br />
QUANT+ TM software uses the NIR<br />
spectra recorded to derive a<br />
calibration. This equation is then<br />
used to predict the value of the<br />
constituent of interest of future<br />
samples using their NIR spectra. An<br />
application that is analyzed by using<br />
spectroscopy is <strong>more</strong> precise than<br />
when measured by other methods.<br />
However, the accuracy of the method<br />
in predicting the true value of the<br />
future samples is dependant upon<br />
the accuracy of the reference method.<br />
It is therefore important to stress<br />
that the steps involved in setting up<br />
the calibration are crucial to the<br />
success of the NIR application. This<br />
is particularly true for the collection<br />
of suitable calibration standards,<br />
and the analysis of the calibration<br />
set by titration. This step should be<br />
carried out with an objective of<br />
producing the greatest possible<br />
accuracy.<br />
A suggestion for achieving this<br />
accuracy is to perform the titration<br />
in duplicate or triplicate. However,<br />
it is <strong>more</strong> important to have as many<br />
samples in the calibration set as<br />
possible. In comparison, it is better<br />
to collect twice as many calibration<br />
samples than it is to perform the<br />
reference analysis of the original<br />
calibration standards in duplicate.<br />
The most ideal, though costly and<br />
time consuming, situation would<br />
allow for the inclusion of a large set<br />
of calibration standards, with each<br />
sample analyzed in duplicate or<br />
triplicate.<br />
QUANT+ software was used to<br />
derive a calibration matrix between<br />
the calibration spectra and their<br />
Figure 1: Extremes of calibration set.<br />
reference OH values. The technique<br />
of PLS extracts those spectral<br />
contributions which are correlated<br />
with OH value, and eliminates the<br />
need to select specific wavelengths<br />
for the calibration.<br />
As previously stated, the near<br />
infrared region consists of many<br />
overlapping bands that contain<br />
overtone and combination bands<br />
involving the absorptions of mainly<br />
OH, NH and CH bonds. The bands<br />
of interest in the determination of<br />
OH value are due to the OH first<br />
overtone at 7150 – 6670 cm-1<br />
(1399 – 1499 nm), the OH<br />
combination bands of 5260 – 4760<br />
cm-1 (1900 – 2100 nm), and<br />
moisture content changes: ~5155<br />
cm-1 (1940 nm).<br />
Experimental<br />
Fourteen standards of known OH<br />
value were supplied to serve as a<br />
calibration set. A further eight<br />
samples were also supplied to act as<br />
an independent validation set. These<br />
were used to compare the reference<br />
values with those values predicted by<br />
the calibration equation. All samples<br />
had been analyzed previously in<br />
triplicate by the reference ‘wet’<br />
chemistry method (titration).<br />
The infrared spectra of all samples<br />
were recorded on a PerkinElmer<br />
FT-NIR Spectrometer. The samples<br />
were scanned in an NIR quartz<br />
transmission cell through a range<br />
of 10000 – 4000 cm -1 at a resolution<br />
of 8 cm -1 and strong apodization.<br />
All spectra were recorded in ratio<br />
mode using the shuttle accessory.<br />
The shuttle automatically collects<br />
alternate background and sample<br />
scans to eliminate the effects of<br />
water vapor in the air. A pathlength<br />
of 5 mm was considered to provide<br />
useful absorbance values. The<br />
cuvettes were cleaned with acetone<br />
and dried before refilling with the<br />
next sample.<br />
Two calibrated methods were<br />
built using the reference data and<br />
the spectra collected from the<br />
FT-NIR Spectrometer. These were<br />
setup using the QUANT+ software.<br />
Both methods used the range 9000 –<br />
4528 cm -1 with an upper threshold of<br />
1.5 A. The offset option was<br />
employed to minimize baseline drift
effects. The number of factors was<br />
set between 2 and 6. The validation<br />
option was set to Full Cross<br />
Validation (see later), and the Expert<br />
Assist (see later) was selected for the<br />
calibration. The first method was built<br />
using the full range (20 – 270 OH<br />
number) of samples. However, the<br />
second method was built covering a<br />
much smaller range of OH number<br />
(25 – 60). The second method was<br />
able to predict <strong>more</strong> accurately<br />
values within its low OH range. The<br />
first method however, was able to<br />
predict values better for the samples<br />
lying higher in the range, but was not<br />
as accurate for the mid-range values.<br />
Results and discussion<br />
The absorbance spectra for the<br />
polyol standards and the unknowns<br />
were collected and two examples,<br />
representing extremes of OH number,<br />
are shown in Figure 1. The spectral<br />
variations at approximately 7200 –<br />
6670 and 5300 – 4760 cm -1 (1389 –<br />
1499 and 1887 – 2100 nm) clearly<br />
show differing OH number, and<br />
correspond to polymeric O-H first<br />
overtone and O-H combination bands<br />
respectively. Temperature variation is<br />
important to consider in the analysis<br />
of polyols and other NIR applications.<br />
This application did not require<br />
temperature control, and heating<br />
was not necessary as all the samples<br />
were liquid at room temperature.<br />
However, care was taken to ensure<br />
that the sample temperatures were<br />
allowed to equilibrate and the<br />
samples were left in the beam for the<br />
same length of time before each<br />
measurement was made.<br />
The QUANT+ software is capable<br />
of modeling the concentrations in<br />
terms of the spectral contributions<br />
relevant to OH determination. This<br />
algorithm can extract the factors in<br />
the calibration that are not responsible<br />
for the specific OH value (the<br />
chemical constituent of interest)<br />
by eliminating the corresponding<br />
dimensions (spectral absorptions)<br />
from the model.<br />
The error present in an OH<br />
evaluation due to hydroxyl band of<br />
moisture absorbed in the sample can<br />
then therefore be minimized by<br />
Factors Standard Error of Prediction % Variance<br />
Limited Range 2 0.91 99.86<br />
Figure 2: Estimated vs. Specified – limited range method.<br />
digitally separating the different OH<br />
bands using this feature of the<br />
software. The calibration equation is<br />
modeled to exclude irrelevant bands<br />
from the analysis, and minimize the<br />
error in the prediction results.<br />
The Full Cross Validation feature<br />
used in this application further<br />
improves the reliability of results.<br />
The software drops one sample from<br />
the calibration set and performs an<br />
entire calibration with the remaining<br />
samples, then makes a prediction for<br />
the sample left. In normal<br />
applications the sample being<br />
predicted is not present in the<br />
calibration set, this gives a <strong>more</strong><br />
realistic if less accurate result. The<br />
Expert Assist option in the software<br />
makes intelligent decisions<br />
regarding the spectra in the<br />
calibration set. The Expert will<br />
decide whether to reject any spectra<br />
or specific regions of spectra in the<br />
calibration set, or disregard any<br />
property values on the basis of a set<br />
of rules developed for IR quantitative<br />
analysis. It will also identify any<br />
outliers in the set. Once these<br />
decisions have been made to exclude<br />
some data in the set, the software<br />
will decide whether or not the<br />
calibration needs to be repeated.<br />
The procedure continues iteratively<br />
until all the rules are satisfied.<br />
The method built over the narrow<br />
range of OH number was used to<br />
predict the samples of OH value<br />
ranging from 20 to 50. The error<br />
estimates are considerably better<br />
than the results obtained using the<br />
full calibration set. By looking at the<br />
results tabulated in the table in Figure<br />
2, 99.86 % of the data can be<br />
explained using 2 factors with a<br />
standard estimate of prediction (SEP)<br />
of 0.91 OH number. Figure 2 illustrates<br />
the accuracy of the method in the<br />
form of an Estimated value versus<br />
Specified value plot. Prediction of<br />
OH number on the 8 independent<br />
samples using this calibrated method<br />
gave OH numbers that were found to<br />
lie within ca. +/– 1 OH number of<br />
the value obtained from the<br />
reference method (titration).<br />
The full range method uses a larger<br />
calibration set to predict OH number<br />
over a much wider range (20 – 275).<br />
The values recorded in the table in<br />
Figure 3 show that in this method,<br />
99.98% of the data can be explained<br />
using three factors in this analysis.<br />
The SEP is much larger in this<br />
method (4.60 OH number) and was<br />
found to be better at predicting<br />
OH number of the higher valued<br />
unknowns.