efsa-opinion-chromium-food-drinking-water
efsa-opinion-chromium-food-drinking-water efsa-opinion-chromium-food-drinking-water
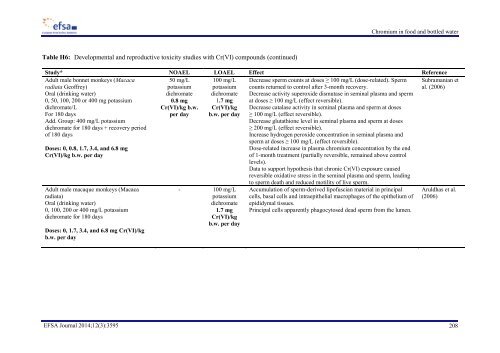
Chromium in food and bottled water Table H6: Developmental and reproductive toxicity studies with Cr(VI) compounds (continued) Study* NOAEL LOAEL Effect Reference Adult male bonnet monkeys (Macaca radiata Geoffrey) Oral (drinking water) 0, 50, 100, 200 or 400 mg potassium dichromate/L For 180 days Add. Group: 400 mg/L potassium dichromate for 180 days + recovery period of 180 days 50 mg/L potassium dichromate 0.8 mg Cr(VI)/kg b.w. per day 100 mg/L potassium dichromate 1.7 mg Cr(VI)/kg b.w. per day Doses: 0, 0.8, 1.7, 3.4, and 6.8 mg Cr(VI)/kg b.w. per day Adult male macaque monkeys (Macaca radiata) Oral (drinking water) 0, 100, 200 or 400 mg/L potassium dichromate for 180 days Doses: 0, 1.7, 3.4, and 6.8 mg Cr(VI)/kg b.w. per day - 100 mg/L potassium dichromate 1.7 mg Cr(VI)/kg b.w. per day Decrease sperm counts at doses ≥ 100 mg/L (dose-related). Sperm counts returned to control after 3-month recovery. Decrease activity superoxide dismutase in seminal plasma and sperm at doses ≥ 100 mg/L (effect reversible). Decrease catalase activity in seminal plasma and sperm at doses ≥ 100 mg/L (effect reversible). Decrease glutathione level in seminal plasma and sperm at doses ≥ 200 mg/L (effect reversible). Increase hydrogen peroxide concentration in seminal plasma and sperm at doses ≥ 100 mg/L (effect reversible). Dose-related increase in plasma chromium concentration by the end of 1-month treatment (partially reversible, remained above control levels). Data to support hypothesis that chronic Cr(VI) exposure caused reversible oxidative stress in the seminal plasma and sperm, leading to sperm death and reduced motility of live sperm. Accumulation of sperm-derived lipofuscian material in principal cells, basal cells and intraepithelial macrophages of the epithelium of epididymal tissues. Principal cells apparently phagocytosed dead sperm from the lumen. Subramanian et al. (2006) Aruldhas et al. (2006) EFSA Journal 2014;12(3):3595 208
Chromium in food and bottled water Table H6: Developmental and reproductive toxicity studies with Cr(VI) compounds (continued) Study* NOAEL LOAEL Effect Reference Adult male bonnet monkeys (Macaca radiata) Oral (drinking water) 0, 100, 200 or 400 mg/L potassium dichromate for 180 days + recovery period of 180 days (half of animals) - 100 mg/L potassium dichromate 1.7 mg Cr(VI)/kg b.w. per day Doses: 0, 1.7, 3.4, and 6.8 mg Cr(VI)/kg b.w. per day Adult male bonnet monkeys (Macaca radiata) Oral (drinking water) 0, 100, 200 or 400 mg/L potassium dichromate for 180 days + recovery period of 180 days (half of animals) Doses: 0, 1.7, 3.4, and 6.8 mg Cr(VI)/kg b.w. per day - 100 mg/L potassium dichromate 1.7 mg Cr(VI)/kg b.w. per day Increase plasma chromium levels at 24h following last day of treatment (up to 10 fold), values declined to control values after 180 days recovery. Decrease relative testes weights at end of treatment, returned to normal following 180 days recovery. Disorganized seminiferous tubules, dose-related decrease in diameter. Depletion of germ cells and hyperplasia of Leydig cells, absence of spermatids in some tubules, Sertoli cell fibrosis, vacuoles surrounding spermatids still adherent to the epithelium, multinucleate giant cells in adluminal compartment, lumen filled with prematurely released germ cells and cell debris and abnormal appearance of chromatin in postzygotene spermatocyte. These effects disappeared after recovery period. Treatment-related changes in testicular structure. The specific activities of testicular superoxide dismutase, catalase, glutathione peroxidase, glutathione reductase, and glucose-6- phosphate dehydrogenase, considered to indicate the status of oxidative stress in the testis, were all significantly decreased. The authors concluded that Cr(VI) disrupts spermatogenesis by inducing free-radical toxicity. Two types of ‘microcanals’ in epididymal epithelium. Effect doserelated. The authors hypothesize that the first type of microcanal provides passage for spermatozoa to bypass the blocked main duct. The second type of microcanal was proposed as a means by which spermatozoa reaching the core of the epithelium are sequestered, as a mechanism to avoid an autoimmune response. (effects were not quantified, but the authors’ believed that the incidence and severity of microcanalisation increased with increasing Cr(VI) concentration). They interpreted their findings as indicative of Cr(VI)-induced obstruction of the distal portion of the cauda epididymis. Aruldhas et al. (2005) Aruldhas et al. (2004) EFSA Journal 2014;12(3):3595 209
- Page 157 and 158: Chromium in food and drinking water
- Page 159 and 160: Chromium in food and drinking water
- Page 161 and 162: Chromium in food and drinking water
- Page 163 and 164: Chromium in food and drinking water
- Page 165 and 166: Chromium in food and drinking water
- Page 167 and 168: Chromium in food and drinking water
- Page 169 and 170: Chromium in food and drinking water
- Page 171 and 172: Chromium in food and drinking water
- Page 173 and 174: Chromium in food and bottled water
- Page 175 and 176: Chromium in food and bottled water
- Page 177 and 178: Chromium in food and bottled water
- Page 179 and 180: Chromium in food and bottled water
- Page 181 and 182: Chromium in food and bottled water
- Page 183 and 184: Chromium in food and bottled water
- Page 185 and 186: Chromium in food and bottled water
- Page 187 and 188: Chromium in food and bottled water
- Page 189 and 190: Chromium in food and bottled water
- Page 191 and 192: Chromium in food and bottled water
- Page 193 and 194: Chromium in food and bottled water
- Page 195 and 196: Chromium in food and bottled water
- Page 197 and 198: Chromium in food and bottled water
- Page 199 and 200: Chromium in food and bottled water
- Page 201 and 202: Chromium in food and bottled water
- Page 203 and 204: Chromium in food and bottled water
- Page 205 and 206: Chromium in food and bottled water
- Page 207: Chromium in food and bottled water
- Page 211 and 212: Chromium in food and bottled water
- Page 213 and 214: Chromium in food and bottled water
- Page 215 and 216: Chromium in food and bottled water
- Page 217 and 218: Chromium in food and bottled water
- Page 219 and 220: Chromium in food and bottled water
- Page 221 and 222: Chromium in food and bottled water
- Page 223 and 224: Chromium in food and bottled water
- Page 225 and 226: Chromium in food and bottled water
- Page 227 and 228: Chromium in food and bottled water
- Page 229 and 230: Chromium in food and bottled water
- Page 231 and 232: Chromium in food and bottled water
- Page 233 and 234: Chromium in food and bottled water
- Page 235 and 236: J.1.2. mice Chromium in food and bo
- Page 237 and 238: Chromium in food and bottled water
- Page 239 and 240: Chromium in food and bottled water
- Page 241 and 242: Chromium in food and bottled water
- Page 243 and 244: Chromium in food and bottled water
- Page 245 and 246: Chromium in food and bottled water
- Page 247 and 248: Chromium in food and bottled water
- Page 249 and 250: Chromium in food and bottled water
- Page 251 and 252: Chromium in food and bottled water
- Page 253 and 254: Chromium in food and bottled water
- Page 255 and 256: Chromium in food and bottled water
- Page 257 and 258: Chromium in food and bottled water
Chromium in <strong>food</strong> and bottled <strong>water</strong><br />
Table H6: Developmental and reproductive toxicity studies with Cr(VI) compounds (continued)<br />
Study* NOAEL LOAEL Effect Reference<br />
Adult male bonnet monkeys (Macaca<br />
radiata Geoffrey)<br />
Oral (<strong>drinking</strong> <strong>water</strong>)<br />
0, 50, 100, 200 or 400 mg potassium<br />
dichromate/L<br />
For 180 days<br />
Add. Group: 400 mg/L potassium<br />
dichromate for 180 days + recovery period<br />
of 180 days<br />
50 mg/L<br />
potassium<br />
dichromate<br />
0.8 mg<br />
Cr(VI)/kg b.w.<br />
per day<br />
100 mg/L<br />
potassium<br />
dichromate<br />
1.7 mg<br />
Cr(VI)/kg<br />
b.w. per day<br />
Doses: 0, 0.8, 1.7, 3.4, and 6.8 mg<br />
Cr(VI)/kg b.w. per day<br />
Adult male macaque monkeys (Macaca<br />
radiata)<br />
Oral (<strong>drinking</strong> <strong>water</strong>)<br />
0, 100, 200 or 400 mg/L potassium<br />
dichromate for 180 days<br />
Doses: 0, 1.7, 3.4, and 6.8 mg Cr(VI)/kg<br />
b.w. per day<br />
- 100 mg/L<br />
potassium<br />
dichromate<br />
1.7 mg<br />
Cr(VI)/kg<br />
b.w. per day<br />
Decrease sperm counts at doses ≥ 100 mg/L (dose-related). Sperm<br />
counts returned to control after 3-month recovery.<br />
Decrease activity superoxide dismutase in seminal plasma and sperm<br />
at doses ≥ 100 mg/L (effect reversible).<br />
Decrease catalase activity in seminal plasma and sperm at doses<br />
≥ 100 mg/L (effect reversible).<br />
Decrease glutathione level in seminal plasma and sperm at doses<br />
≥ 200 mg/L (effect reversible).<br />
Increase hydrogen peroxide concentration in seminal plasma and<br />
sperm at doses ≥ 100 mg/L (effect reversible).<br />
Dose-related increase in plasma <strong>chromium</strong> concentration by the end<br />
of 1-month treatment (partially reversible, remained above control<br />
levels).<br />
Data to support hypothesis that chronic Cr(VI) exposure caused<br />
reversible oxidative stress in the seminal plasma and sperm, leading<br />
to sperm death and reduced motility of live sperm.<br />
Accumulation of sperm-derived lipofuscian material in principal<br />
cells, basal cells and intraepithelial macrophages of the epithelium of<br />
epididymal tissues.<br />
Principal cells apparently phagocytosed dead sperm from the lumen.<br />
Subramanian et<br />
al. (2006)<br />
Aruldhas et al.<br />
(2006)<br />
EFSA Journal 2014;12(3):3595 208