Filed - Glenmark
Filed - Glenmark
Filed - Glenmark
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
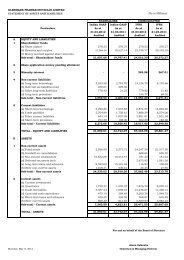
USA – Generic Formulations<br />
Current Status<br />
<br />
<br />
<br />
<br />
Rapidly built up a generic sales front end in the USA with investments in<br />
distribution, finance systems and sales team<br />
Organic and inorganic build-up of portfolio<br />
• <strong>Filed</strong> 10 ANDAs with US FDA towards FY07 filings (including partner filings<br />
and 2 Derma products and 2 FTFs) till Oct 06.<br />
In all, <strong>Glenmark</strong> has 25 ANDAs undergoing the US FDA approval process /<br />
launch.<br />
• 10 Generics on market – own and partnered<br />
• Focus on niche segment of controlled substances and dermatology<br />
6 controlled substances licensed: 2 on market and 1 to be launched in<br />
November<br />
Paul Cap partnerships to finance product development in dermatology<br />
• Strategic co-development tie-ups concluded with Shasun and InvaGen:<br />
Together accounting for 20 additional filings to which <strong>Glenmark</strong> has<br />
marketing access<br />
Excellent sales performance – locked in excess of 20-25% of the US market on<br />
most products within first year of launch<br />
Achieved operating cash break-even in Q4 FY06 – within 3 years of set-up<br />
15





![Formulations [India] â Product List - Glenmark](https://img.yumpu.com/46601329/1/190x245/formulations-india-a-product-list-glenmark.jpg?quality=85)
![Formulation [India] â Product List - Glenmark](https://img.yumpu.com/44013338/1/190x245/formulation-india-a-product-list-glenmark.jpg?quality=85)




![Formulations [India] â Product List - Glenmark](https://img.yumpu.com/35994839/1/190x245/formulations-india-a-product-list-glenmark.jpg?quality=85)

