Filed - Glenmark
Filed - Glenmark
Filed - Glenmark
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
Disclaimer<br />
This presentation has been prepared by <strong>Glenmark</strong><br />
Pharmaceuticals Ltd. All information, statements and analysis<br />
made in this presentation describing company’s objectives,<br />
projections and estimates are forward looking statements and<br />
progressive within the meaning of applicable security Laws<br />
and Regulations. The analysis contained herein is based on<br />
numerous assumptions. Actual result may vary from those<br />
Less Regulated Markets<br />
expressed or implied depending upon economic conditions,<br />
government policies and other incidental factors. No<br />
representation or warranty, either express or implied, is<br />
provided in relation to this presentation. This presentation<br />
should not be regarded by recipients as a substitute for the<br />
exercise of their own judgment.
Contents<br />
Background<br />
Strategy and Business Units<br />
Business Unit Overview<br />
Financials and Projections<br />
Summary
Background<br />
History<br />
<br />
29-year old company<br />
Went public on Indian bourses in 1999<br />
Identity / Scope<br />
<br />
<br />
Integrated, operating in Research,<br />
Manufacturing & Marketing of Pharmaceuticals<br />
Business interests span formulations and bulk<br />
activities<br />
Global Footprint<br />
Financial<br />
Performance<br />
Market<br />
Capitalisation<br />
<br />
<br />
<br />
Headquartered in Mumbai, India<br />
Operates in over 80 countries<br />
About 42% of revenues in FY 2006 from outside<br />
India<br />
Results in FY 2006 (ending March 31, 2006):<br />
• Consolidated revenues of ~ $171 Mn<br />
• Consolidated Profit after Tax of ~ $20 Mn<br />
~USD 1026 Mn (as at October 26, 2006)<br />
1
Strategy and Business<br />
Units<br />
2
Strategic roadmap: Transform into a specialty company<br />
through research<br />
Learning from Trends<br />
• Growing through generics alone is<br />
not a long-term value creator<br />
• Differentiation is key – but close to<br />
impossible in the generic space<br />
• Niche therapy segments offer good<br />
opportunity for value growth in<br />
generics space<br />
• Ownership of IP is critical to<br />
differentiation and value growth<br />
• NCE research is a large opportunity;<br />
but success in biologics can help<br />
leap-frog industry evolution<br />
• Taking proprietary molecules through<br />
all development phases is beyond our<br />
current skill-set and risk-appetite<br />
• Several markets are below the radar<br />
screen for bigger branded pharma<br />
players<br />
The Transformation Plan<br />
• Focus on building IP assets today, the<br />
key engine for this transformation<br />
• Out-license IP to drive/fund drug<br />
development<br />
• Build and launch portfolios of generic<br />
products in niche segments like<br />
Dermatology<br />
• Retain/acquire IP rights for markets<br />
under the “radar” screen<br />
• In parallel, seek out markets with growth<br />
potential and build a branded generic<br />
marketing foot-print<br />
• Subsequently, transform branded<br />
generics to specialty businesses: launch<br />
<strong>Glenmark</strong>-brand novel drugs<br />
3
Execute through Business Units aligned to Strategy<br />
Organized by Business Units<br />
CEO<br />
R&D<br />
Generic Formulations<br />
API<br />
NCE<br />
Biologics<br />
(Swiss)<br />
US<br />
Europe<br />
Latin<br />
America<br />
Less<br />
Regulated<br />
Markets<br />
India<br />
API<br />
4
Research and Development<br />
5
Build proprietary pipeline of NCEs/Biologics in high<br />
growth areas with good licensing potential<br />
RESEARCH FOCUS<br />
NCE RESEARCH<br />
Metabolic Disorders<br />
• Diabetes (Type II)<br />
• Obesity<br />
Inflammation<br />
• Asthma/COPD<br />
• Pain<br />
NCE Research – In Mumbai, India<br />
• Focused capability building over 5 years –<br />
170 scientists, spend ~5% of revenues<br />
• Eminent scientific advisory board<br />
• Five validated targets; 3 more planned<br />
• 6 leads – 2 in Ph II, 3 entering Ph I shortly<br />
and 1 in Pre-clinical<br />
• Target: One NCE into clinics every year<br />
BIOLOGICS<br />
Undisclosed Therapies and Targets<br />
Biologics Research – In Switzerland<br />
• Lab commissioned –15+ researchers<br />
by 2006<br />
• Invest to build capability until 2009-10<br />
• Start with bio-improvics; new drugs by<br />
2008<br />
• Target : Ph 1 on first lead by 2009<br />
6
Licensing Strategy<br />
Partner Selection<br />
• Selection criteria : (a) drug development skills (b) experience in therapy area<br />
(c) Marketing prowess (d) deal value (e) development program/speed to market<br />
• Preference for before Phase IIA/PoC deal timing;<br />
Sales $ Bn<br />
$400<br />
$300<br />
$200<br />
$100<br />
Licensing Focus<br />
US<br />
EU<br />
Jap<br />
Retain rights: high<br />
growth markets of<br />
Asia, Africa,<br />
Russia, Lat Am,<br />
Australia<br />
APPROACH TO LICENSING<br />
• Retain use of clinical data generated<br />
by partner<br />
• Retain exclusive /<br />
co- commercialization rights to<br />
products for Asia, Africa, Australia,<br />
Lat Am and Russia<br />
• Register product upon US/EU<br />
approval in these markets and launch<br />
$0<br />
0% 5% 10% 15% 20%<br />
Annual Sales Growth<br />
License out the leads to partners for the markets of North America, Europe and<br />
Japan; retain exclusive / co-commercialization rights for rest of the world<br />
7
Progress on 6 Compounds …<br />
Plan to progress 5 compounds in clinical trials by FY 2007<br />
Compound Target Primary Indications Status Target<br />
Launch<br />
GRC 3886<br />
PDE 4<br />
Asthma, COPD<br />
Ph II 2009/10<br />
(Oglemilast)<br />
GRC 8200<br />
DPP IV<br />
Diabetes (Type II)<br />
Ph II 2010<br />
GRC 6211 VR 1 Osteoarthritis, Dental Pain,<br />
Incontinence, Neuropathic<br />
Pain<br />
GRC 10693 CB 2 Neuropathic Pain,<br />
Osteoarthritis and other<br />
Inflammatory Pain<br />
Ph I in<br />
Nov 06<br />
Ph I in<br />
Jan 07<br />
2011<br />
2012<br />
GRC 10801 CB 1 Obesity Ph I in<br />
Q4 FY07<br />
2011<br />
GRC 4039 PDE 4 Rheumatoid Arthritis,<br />
Inflammation<br />
Ph I in<br />
Q1 FY08<br />
2012<br />
<strong>Glenmark</strong> will conclude 1 more licensing collaboration in FY 2007<br />
8
NCE Pipeline<br />
Lead Target Indication Stage Status<br />
GRC 3886<br />
(Oglemilast)<br />
PDE 4<br />
Asthma,<br />
COPD<br />
Ph II<br />
• Currently 2 Ph II trials underway for “exercise<br />
induced asthma” and “antigen challenge”;<br />
enrollment complete for the first study, top line<br />
results likely by Nov FY07<br />
• Phase II B for Asthma, COPD likely to begin<br />
early next year<br />
• Survey by R&D Directions classified GRC 3886<br />
as one of the Great 100 Investigational Drugs<br />
• $ 30 Mn expected from Forest Inc.,<br />
• In discussions with potential European Partners<br />
GRC 8200<br />
DPP IV<br />
Diabetes<br />
(Type II)<br />
Ph II<br />
• 2 Ph II trials underway in South Africa and India<br />
• Tied up with Merck KGaA of Germany in Oct 06<br />
to develop, register and commercialize GRC<br />
8200 and its combinations in US, Europe, Japan<br />
• <strong>Glenmark</strong> retains exclusive marketing rights for<br />
India and will co-commercialize the drug in RoW<br />
• Total deal size is EUR 190 Mn, with EUR 25 Mn<br />
as an up-front payment and the remaining<br />
milestone payments upon successful<br />
development, launch<br />
• Upon commercial launch, <strong>Glenmark</strong> will supply<br />
the API to Merck KGaA and will receive royalties<br />
on net sales of the product<br />
9
NCE Pipeline<br />
Lead Target Indication Stage Status<br />
GRC 6211 VR 1 Osteoarthritis,<br />
Dental Pain,<br />
Incontinence,<br />
Neuropathic<br />
pain<br />
GRC 10693 CB 2 Neuropathic<br />
pain,<br />
Osteoarthritis,<br />
Rheumatoid<br />
Arthritis and<br />
other anti -<br />
inflammatory<br />
pain<br />
Ph I in<br />
Nov 06<br />
Ph I in<br />
Dec /<br />
Jan 07<br />
• Exciting target – after Merck licensed<br />
compound from Neurogen (currently in<br />
Ph I) and Pfizer licensed compound from<br />
Renovis (currently in pre-clinicals)<br />
• <strong>Glenmark</strong> currently in discussions for<br />
potential licensing. Timing of licensing<br />
would depend on the deal terms and<br />
ability to get co-promotion rights in some<br />
of the regulated markets<br />
• <strong>Glenmark</strong> could be 2 nd in class with this<br />
compound<br />
• Likely to be first in class with an oral CB2<br />
Agonist. Currently Pharmos has an IV<br />
compound in Ph II and GSK has an oral<br />
compound in Ph I<br />
• Very exciting target as non-narcotic,<br />
peripherally acting, excellent pain-control<br />
and without addiction potential<br />
associated with Opoids. Potential<br />
applications in Rheumatoid Arthritis.<br />
• Currently in discussion with several bigpharma<br />
companies for potential licensing<br />
10
NCE Pipeline<br />
Lead Target Indication Stage Status<br />
GRC<br />
10801<br />
CB 1 Obesity Ph I in<br />
Q4<br />
FY07<br />
• Though Rimonabant (Sanofi-Aventis) is approved<br />
in EU, not an ideal candidate due to safety aspects<br />
• <strong>Glenmark</strong>’s lead target profile will clearly<br />
differentiate us on aspects of safety and drug<br />
metabolism<br />
• Though lead will enter Ph I in end-FY07, we expect<br />
to be third/fourth in class with a clear differential<br />
over existing compounds<br />
GRC<br />
4039<br />
PDE 4<br />
Rheumatoid<br />
Arthritis,<br />
Inflammation<br />
Ph I in<br />
Q1<br />
FY08<br />
• After the withdrawal/setback of COX-2s and the<br />
void that exists in RA therapy, we believe this<br />
could be a potential block-buster opportunity<br />
• The lead is also showing promise in Animal<br />
models in RA and other inflammatory conditions<br />
11
Biologics research in Switzerland in early phase<br />
Swiss Presence<br />
• Diversifying research activities<br />
to cover biologics<br />
• Bioimprovics and New biologics<br />
in the future – biosimilars to<br />
build skill-sets in the near term<br />
• Biologics Research Centre<br />
established<br />
• Employed scientists with<br />
Biologics Research experience<br />
from Switzerland and Europe<br />
12
Generic Formulations<br />
13
Overview: Market generics in the US market through own<br />
front-end ; Acquire branded generic footprint in Europe<br />
US Market<br />
• Remain focused on generics<br />
• Commercialize through own front end<br />
• Combination of Make/Partner/Buy to<br />
build depth of portfolio rapidly<br />
– Own development<br />
– Product development alliances<br />
– Acquire registrations<br />
– Licensing marketing rights<br />
• Focus own development on specific<br />
niches with greater<br />
margins/sustainability: Dermatology,<br />
modified release and controlled<br />
substances<br />
• Use own API for cost advantage<br />
Europe Market<br />
• Focus on select branded generic<br />
markets: Spain, Italy, Central and<br />
Eastern European Countries<br />
• Acquire privately held front ends<br />
– EUR 8-12 Mn Sales, cash generating<br />
– Pipeline in place for next 2-3 years<br />
– Good local brand equity<br />
– High overlap in prescriber base with<br />
our research focus<br />
• File and launch branded generics<br />
through own front-ends; also provide<br />
dossiers to partners for non-core<br />
markets<br />
• Partner with players to fund high cost<br />
developments – share marketing rights<br />
14
USA – Generic Formulations<br />
Current Status<br />
<br />
<br />
<br />
<br />
Rapidly built up a generic sales front end in the USA with investments in<br />
distribution, finance systems and sales team<br />
Organic and inorganic build-up of portfolio<br />
• <strong>Filed</strong> 10 ANDAs with US FDA towards FY07 filings (including partner filings<br />
and 2 Derma products and 2 FTFs) till Oct 06.<br />
In all, <strong>Glenmark</strong> has 25 ANDAs undergoing the US FDA approval process /<br />
launch.<br />
• 10 Generics on market – own and partnered<br />
• Focus on niche segment of controlled substances and dermatology<br />
6 controlled substances licensed: 2 on market and 1 to be launched in<br />
November<br />
Paul Cap partnerships to finance product development in dermatology<br />
• Strategic co-development tie-ups concluded with Shasun and InvaGen:<br />
Together accounting for 20 additional filings to which <strong>Glenmark</strong> has<br />
marketing access<br />
Excellent sales performance – locked in excess of 20-25% of the US market on<br />
most products within first year of launch<br />
Achieved operating cash break-even in Q4 FY06 – within 3 years of set-up<br />
15
USA – Generic Formulations<br />
Going forward: Multi-pronged build-out of Product Portfolio<br />
<br />
<br />
Own Filings: File 15-20 ANDAs in FY07; Includes 3-5 XRs, 6 Derma<br />
products and healthy & exciting mix of P-II, P-III & at least 3 FTF/P-IV<br />
opportunities<br />
Niche topical generics: working on 20+ steroidal, non-steroidal and<br />
combination topicals<br />
Niche controlled substances: Launched 2 controlled substances out of 5<br />
products licensed from two US based firms; remaining to be launched by<br />
Feb 07. Segment offers lower competitive pressures. Further licensing<br />
efforts ongoing.<br />
<br />
Partnerships: Already announced partnerships (Shasun and InvaGen) will<br />
file over 8 generics with the US FDA in FY07.<br />
Current status Target for FY 2007<br />
Marketed 10<br />
Under Approval 25<br />
Total Filings 35<br />
Own filings 21<br />
Marketed 18-22<br />
Under approval 38-40<br />
Total Filings 56-62<br />
Own filings 15-20<br />
Expect to Exceed April Guidance for FY07<br />
16
Europe – Start-up phase<br />
Strategy & Progress<br />
<br />
Entry through a niche / branded strategy<br />
• Developing niche generic products, and partnering & creating alliances<br />
with European companies<br />
• Entered into a joint developing, filing and marketing deal for 8 derma products<br />
with Merck Generics, Germany for the European region. The first of these<br />
products has already been filed and is targeted to be launched in 2007; the<br />
remaining products are forecasted to be launched by 2010.<br />
• Expanding Inorganically in select branded generics markets in Europe<br />
• Currently in discussions with Potential company for acquisition<br />
<br />
Establish front-end<br />
• Europe headquarters, <strong>Glenmark</strong> Pharmaceuticals (Europe) Ltd., set up in<br />
2004<br />
• Team building completed<br />
• Potential front-end acquisitions under review<br />
17
Latin America<br />
Current Status<br />
<br />
<br />
<br />
<br />
Integrated activities of acquired company, Servycal with <strong>Glenmark</strong> operations:<br />
launched multiple Servycal products into Brazil; registrations in Rest of the<br />
World<br />
Expanded field force and also commenced commercial operations in several<br />
additional countries: presence in 10 countries (including Brazil and Argentina)<br />
in Latin America<br />
Focused on specific therapeutic segments: Respiratory, Dermatology,<br />
Gynaecology and Oncology<br />
<strong>Filed</strong> 38 dossiers in Latin America and received approval to market 8 products<br />
18
Latin America<br />
Going forward<br />
<br />
<br />
<br />
<br />
Expand presence in Oncology to other Latin American markets as well<br />
as other key less regulated markets: use Argentina as the hub<br />
File an additional 10 dossiers in markets across Latin America in the<br />
second half of the year. Launch 10 new products in Brazil<br />
Margin expansion through increasing revenue for products filed from<br />
Indian parent<br />
Divisionalisation for greater focus – focus on high margin segments<br />
Achieve 100% growth in revenues over FY 2006<br />
Expect to Meet Guidance for FY07<br />
19
Less Regulated Markets<br />
Current Status (Apr-Sep 06)<br />
Plan going forward…<br />
<br />
<br />
Posted 96% growth in revenues<br />
fuelled by growth in Russia and<br />
acquisitions in South Africa<br />
Registered products in 2 new<br />
countries and started operations in 4<br />
<br />
<br />
<br />
Consolidate position in Russia<br />
Expand in South Africa – use the India<br />
advantage to file several products<br />
Focus on high potential country markets<br />
<br />
Completed 249 filings and received<br />
137 approvals for products across<br />
markets<br />
<br />
Diversify portfolio to include an<br />
oncology line – leverage the acquisition<br />
of Servycal in Argentina<br />
<br />
Consolidated position in existing<br />
markets: increased field force,<br />
launched many new products,<br />
reviewed portfolio to meet local<br />
requirements<br />
<br />
<br />
Continue registration of products across<br />
geographies<br />
Achieve over 40% growth in revenues<br />
over FY 2006<br />
<br />
Evaluate acquisition candidates in highpotential<br />
RoW markets<br />
Expect to Meet Guidance for FY07<br />
20
India Formulations<br />
Current Status<br />
<br />
Value growth of over 21% vis-à-vis IPM which grew at 16.2% (April – Sept<br />
06, ORG-IMS Sept 06)<br />
Ranked #2 in dermatology (after GSK), strong presence in over 10<br />
segments including female healthcare, paediatrics, respiratory, antiinfectives,<br />
cardiovascular, diabetology, oncology<br />
<br />
<br />
<br />
<br />
<br />
<br />
Increased focus on chronic lifestyle diseases<br />
Launched 17 new products and novel combinations in the first half of the<br />
year<br />
Created 7 retail divisions, 1 institutional division & 1 targeting family<br />
physicians for improved focus<br />
Grew field force (currently 1850 medical representatives) and increased<br />
focus on productivity improvements<br />
Driving supply chain cost down; new plant at Baddi for tax-efficiencies fully<br />
on-stream<br />
Commenced in-licensing and co-marketing tie-ups for novel products<br />
21
India Formulations<br />
Segment Contribution to Revenue: Diversified across therapies<br />
August ORG MAT 2005 August ORG MAT 2006<br />
5%<br />
11%<br />
5% 3% 3% 2% 3% 0%<br />
31%<br />
6%<br />
6%<br />
5% 4% 3% 2% 3%<br />
30%<br />
9%<br />
12%<br />
16%<br />
9%<br />
16%<br />
16%<br />
Derma Respiratory Anti-infectives Anti Diabetic<br />
Pain Cardiac Gynaec G.I<br />
Otologicals Stomatologicals Others<br />
22
India Formulations<br />
Moving forward, the plan…<br />
Grow revenues by 15% p.a. - revised upwards from earlier guidance of 10%<br />
<br />
<br />
<br />
<br />
<br />
Strengthen presence in respiratory, pain management and metabolic diseases<br />
– in preparation for own NCE launches<br />
Grow mass-market division, Milieus, through increased product launches,<br />
sales force hires and geographic expansion<br />
Increase in-licensing efforts for novel products to augment own pipeline<br />
Continue launching differentiated branded generics; focus on building<br />
power brands<br />
Improve cost-efficiencies and cash-cycle management<br />
Expect to Exceed April (Profit) Guidance for FY07<br />
23
Active Pharmaceutical<br />
Ingredients<br />
24
Active Pharmaceutical Ingredients<br />
Current Status<br />
Plan going forward…<br />
<br />
<br />
<br />
<br />
<br />
Revenue increase (28.7%) driven by:<br />
• Growth in regulated markets<br />
• New product introductions<br />
Mix of revenues from segments:<br />
• India and co-marketing – 55%<br />
• Exports – 45%<br />
Commenced commercial supplies of<br />
DMFs during year; several samples,<br />
validation, pivotal batches to global<br />
generic players for their<br />
ANDA/Dossier filings<br />
<strong>Filed</strong> 6 DMFs in H1<br />
Capacity de-bottlenecking initiated<br />
for Ankleshwar, commenced preconstruction<br />
work at new site<br />
Aurangabad<br />
Revenue growth in excess of 30%<br />
<br />
<br />
<br />
<br />
<br />
Complete capacity expansion in<br />
Ankleshwar, start construction in<br />
Aurangabad<br />
File 12-14 DMFs for the year<br />
Commercial supplies to regulated<br />
markets to increase dramatically with<br />
several ANDAs/Dossiers filed and<br />
pending from out 3rd party<br />
customers<br />
Develop several APIs annually to<br />
provide cost and timing advantage to<br />
complement our US and Europe<br />
generic filings<br />
Continue to be a preferred 3 rd party<br />
API supplier to the generic industry<br />
Expect to Meet Guidance for FY07<br />
25
Financials and Projections<br />
26
Financials and Projections<br />
Performance in FY 2006<br />
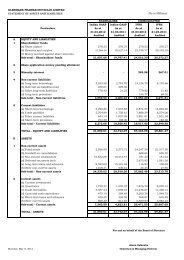
FY 2005 1 FY 2006 2 H1 FY 2007 3<br />
(In USD Million)<br />
Audited<br />
Audited<br />
Un Audited<br />
Operating income (A)<br />
139.87<br />
171.09<br />
97.90<br />
Expenditure (B)<br />
EBITDA (Operations) (C =A-B)<br />
EBT (Operations) (D)<br />
Other Income (E)<br />
PBT (F=D+E)<br />
PAT (G)<br />
104.27<br />
35.60<br />
27.90<br />
1.19<br />
29.09<br />
24.48<br />
140.11<br />
30.98<br />
22.41<br />
2.90<br />
25.31<br />
19.87<br />
77.72<br />
20.18<br />
12.72<br />
1.61<br />
14.33<br />
12.89<br />
Basic Earnings per share (in rupees) 8.97 7.28 4.89<br />
Diluted Earnings per share (in<br />
rupees) 8.16 6.41 4.31<br />
Note : 1. Average conversion rate for FY 2004-05 of Rs. 44.94 / USD 1.00<br />
2. Average conversion rate for FY 2005-06 of Rs. 44.28 / USD 1.00<br />
3. Average conversion rate for FY 2006-07 of Rs. 45.68/ USD 1.00 27
Financials and Projections – Net of Milestones<br />
Projections (in Mn USD)<br />
Revenues<br />
In USD Mn*<br />
164<br />
248 334 CAGR ~ 42%<br />
Business Mix<br />
55<br />
40<br />
108<br />
22<br />
98<br />
49<br />
90<br />
33<br />
47<br />
34<br />
24<br />
17 43<br />
13<br />
75<br />
FY 2006 FY 2007 FY 2008<br />
API<br />
India Formulation<br />
ROW<br />
Latam<br />
USA<br />
Net Profits<br />
15 35<br />
55 CAGR ~ 91%<br />
* In USD Mn (Assumed exchange rate of 44.28 for FY06 and 45.2 INR/USD for FY07 and FY08) 28
Financials and Projections<br />
Milestone Projections (in Mn USD)<br />
Revenues<br />
In USD Mn* 7 31 69<br />
GRC 3886<br />
7 M USD<br />
GRC 8200<br />
31 M USD<br />
All<br />
Milestones<br />
69 M USD<br />
FY 2006 FY 2007 FY 2008<br />
Note 1 : We believe that we will receive 3886 milestone in FY 2007; however due to Forest’s recent statement and<br />
conservative accounting principles, we have taken the milestone into next year<br />
Note 2 : Figures for Milestones now include only those pertaining to GRC 3886 (Oglemilast) and 8200<br />
* In USD Mn (Assumed exchange rate of 44.28 for FY06 and ~45.20 INR/USD and ~ 1.26 EUR/USD for FY07 and FY08) 29
Financials and Projections – Summary<br />
Revenues<br />
In USD Mn*<br />
Business Mix<br />
24<br />
13<br />
171<br />
7<br />
22<br />
90<br />
Projections (in Mn USD)<br />
17<br />
279 397 CAGR ~ 52%<br />
31<br />
40<br />
98<br />
33<br />
34<br />
43<br />
69<br />
55<br />
108<br />
49<br />
47<br />
75<br />
FY 2006 FY 2007 FY 2008<br />
Net profit (Mn. USD) 20.5 62 115<br />
EPS (INR) 7.28 23.60 43.78<br />
P/E (Based on Rs. 393<br />
Sh Price as of 26.10.06)<br />
53.95 16.64 8.97<br />
NCE<br />
API<br />
India Formulation<br />
ROW<br />
Latam<br />
USA<br />
CAGR ~ 137%<br />
* In USD Mn (Assumed exchange rate of 44.28 for FY06 and ~45.20 INR/USD and ~ 1.26 EUR/USD for FY07 and FY08)<br />
30
Summary<br />
31
Summary-Key target milestones by FY 2007<br />
<br />
<br />
<br />
Research<br />
Conclude 1 more licensing deal for NCEs by End FY 2007<br />
<br />
Aim to progress Phase II for GRC 8200 and GRC 3886; initiate Phase I trials<br />
on 3 NCE compounds (GRC 10693, 6211 and 10801)<br />
Formulations<br />
<br />
<br />
<br />
<br />
APIs<br />
<br />
<br />
Conclude 1 acquisition in Europe<br />
In-license novel products/co-marketing deals<br />
Market 18-22 ANDAs in the US market<br />
File 15-20 solid dose ANDAs and another 6 topicals; include XRs and P-IV<br />
opportunities<br />
File 12-14 DMFs<br />
Commence construction in Aurangabad, expand Ankhleshwar<br />
32
Thank you<br />
33





![Formulations [India] â Product List - Glenmark](https://img.yumpu.com/46601329/1/190x245/formulations-india-a-product-list-glenmark.jpg?quality=85)
![Formulation [India] â Product List - Glenmark](https://img.yumpu.com/44013338/1/190x245/formulation-india-a-product-list-glenmark.jpg?quality=85)




![Formulations [India] â Product List - Glenmark](https://img.yumpu.com/35994839/1/190x245/formulations-india-a-product-list-glenmark.jpg?quality=85)

