Diclofenac Sodium 4% Spray Gel - cyathus.cz
Diclofenac Sodium 4% Spray Gel - cyathus.cz
Diclofenac Sodium 4% Spray Gel - cyathus.cz
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
PAR <strong>Diclofenac</strong> <strong>Sodium</strong> <strong>4%</strong> <strong>Spray</strong> <strong>Gel</strong><br />
UK/H/0562-3/001/E01<br />
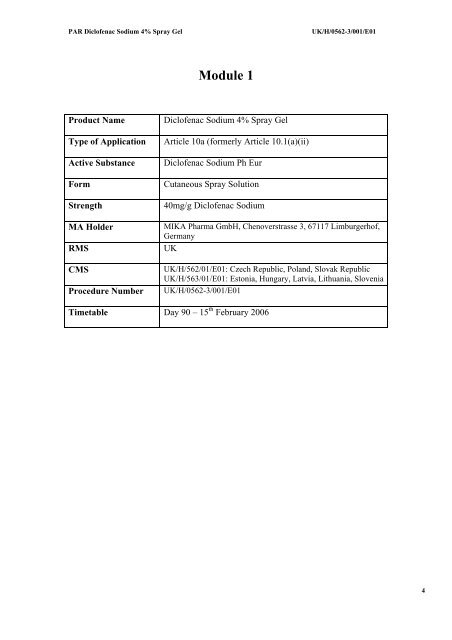
Module 1<br />
Product Name<br />
Type of Application<br />
Active Substance<br />
Form<br />
Strength<br />
MA Holder<br />
RMS<br />
CMS<br />
Procedure Number<br />
<strong>Diclofenac</strong> <strong>Sodium</strong> <strong>4%</strong> <strong>Spray</strong> <strong>Gel</strong><br />
Article 10a (formerly Article 10.1(a)(ii)<br />
<strong>Diclofenac</strong> <strong>Sodium</strong> Ph Eur<br />
Cutaneous <strong>Spray</strong> Solution<br />
40mg/g <strong>Diclofenac</strong> <strong>Sodium</strong><br />
MIKA Pharma GmbH, Chenoverstrasse 3, 67117 Limburgerhof,<br />
Germany<br />
UK<br />
UK/H/562/01/E01: Czech Republic, Poland, Slovak Republic<br />
UK/H/563/01/E01: Estonia, Hungary, Latvia, Lithuania, Slovenia<br />
UK/H/0562-3/001/E01<br />
Timetable Day 90 – 15 th February 2006<br />
4