Invasive breast carcinoma - IARC
Invasive breast carcinoma - IARC
Invasive breast carcinoma - IARC
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
A<br />
B<br />
absence of either microcalcifications or<br />
n e c rosis does not impact the diagnosis.<br />
UDH with necrosis, a rare event, may be<br />
mistaken for DCIS; the diagnosis should<br />
be based on the cytological features and<br />
not the presence of necrotic debris. UDH<br />
generally displays either diffuse or a<br />
mosaic pattern of positivity with high<br />
molecular weight cytokeratins {1963,<br />
2126} such as CK5, CK1/5/10/14 (clone<br />
CK34betaE12 or clone D5/16 B4); it is<br />
also positive for E-cadherin. In UDH, the<br />
p e rcentage of ER-positive cells was found<br />
slightly increased compared to the norm a l<br />
b reast {2667}. Increased levels of cyclin<br />
D1 expression were recently described in<br />
11-19% of UDH cases {1172,3264}.<br />
C<br />
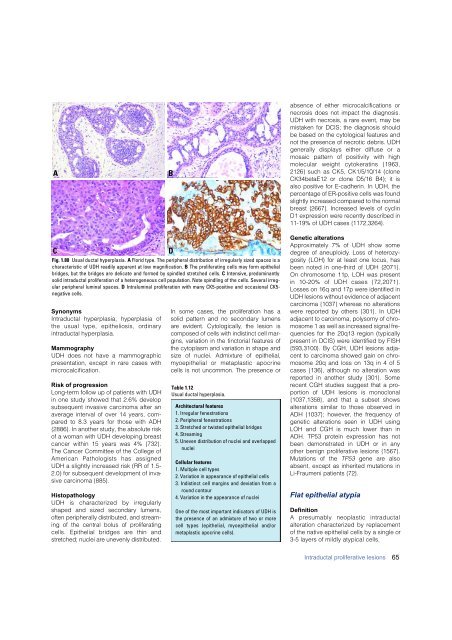
Fig. 1.80 Usual ductal hyperplasia. A Florid type. The peripheral distribution of irregularly sized spaces is a<br />
characteristic of UDH readily apparent at low magnification. B The proliferating cells may form epithelial<br />
bridges, but the bridges are delicate and formed by spindled stretched cells. C Intensive, predominantly<br />
solid intraductal proliferation of a heterogeneous cell population. Note spindling of the cells. Several irregular<br />
peripheral luminal spaces. D Intraluminal proliferation with many CK5-positive and occasional CK5-<br />
negative cells.<br />
Synonyms<br />
Intraductal hyperplasia, hyperplasia of<br />
the usual type, epitheliosis, ord i n a ry<br />
intraductal hyperplasia.<br />
Mammography<br />
UDH does not have a mammographic<br />
presentation, except in rare cases with<br />
microcalcification.<br />
Risk of progression<br />
Long-term follow up of patients with UDH<br />
in one study showed that 2.6% develop<br />
subsequent invasive <strong>carcinoma</strong> after an<br />
average interval of over 14 years, compared<br />
to 8.3 years for those with ADH<br />
{2886}. In another study, the absolute risk<br />
of a woman with UDH developing <strong>breast</strong><br />
cancer within 15 years was 4% {732}.<br />
The Cancer Committee of the College of<br />
American Pathologists has assigned<br />
UDH a slightly increased risk (RR of 1.5-<br />
2.0) for subsequent development of invasive<br />
<strong>carcinoma</strong> {885}.<br />
Histopathology<br />
UDH is characterized by irre g u l a r l y<br />
shaped and sized secondary lumens,<br />
often peripherally distributed, and stre a m-<br />
ing of the central bolus of pro l i f e r a t i n g<br />
cells. Epithelial bridges are thin and<br />
s t retched; nuclei are unevenly distributed.<br />
D<br />
In some cases, the proliferation has a<br />
solid pattern and no secondary lumens<br />
a re evident. Cytologically, the lesion is<br />
composed of cells with indistinct cell margins,<br />
variation in the tinctorial features of<br />
the cytoplasm and variation in shape and<br />
size of nuclei. Admixture of epithelial,<br />
myoepithelial or metaplastic apocrine<br />
cells is not uncommon. The presence or<br />
Table 1.12<br />
Usual ductal hyperplasia.<br />
Architectural features<br />
1. Irregular fenestrations<br />
2. Peripheral fenestrations<br />
3. Stretched or twisted epithelial bridges<br />
4. Streaming<br />
5. Uneven distribution of nuclei and overlapped<br />
nuclei<br />
Cellular features<br />
1. Multiple cell types<br />
2. Variation in appearance of epithelial cells<br />
3. Indistinct cell margins and deviation from a<br />
round contour<br />
4. Variation in the appearance of nuclei<br />
One of the most important indicators of UDH is<br />
the presence of an admixture of two or more<br />
cell types (epithelial, myoepithelial and/or<br />
metaplastic apocrine cells).<br />
Genetic alterations<br />
A p p roximately 7% of UDH show some<br />
d e g ree of aneuploidy. Loss of hetero z y-<br />
gosity (LOH) for at least one locus, has<br />
been noted in one-third of UDH {2071}.<br />
On chromosome 11p, LOH was pre s e n t<br />
in 10-20% of UDH cases {72,2071}.<br />
Losses on 16q and 17p were identified in<br />
UDH lesions without evidence of adjacent<br />
c a rcinoma {1037} whereas no alterations<br />
w e re re p o rted by others {301}. In UDH<br />
adjacent to <strong>carcinoma</strong>, polysomy of chromosome<br />
1 as well as increased signal frequencies<br />
for the 20q13 region (typically<br />
p resent in DCIS) were identified by FISH<br />
{593,3100}. By CGH, UDH lesions adjacent<br />
to <strong>carcinoma</strong> showed gain on chromosome<br />
20q and loss on 13q in 4 of 5<br />
cases {136}, although no alteration was<br />
re p o rted in another study {301}. Some<br />
recent CGH studies suggest that a prop<br />
o rtion of UDH lesions is monoclonal<br />
{1037,1358}, and that a subset shows<br />
alterations similar to those observed in<br />
ADH {1037}; however, the frequency of<br />
genetic alterations seen in UDH using<br />
LOH and CGH is much lower than in<br />
ADH. TP53 protein expression has not<br />
been demonstrated in UDH or in any<br />
other benign proliferative lesions {1567}.<br />
Mutations of the T P 5 3 gene are also<br />
absent, except as inherited mutations in<br />
Li-Fraumeni patients {72}.<br />
Flat epithelial atypia<br />
Definition<br />
A presumably neoplastic intraductal<br />
alteration characterized by replacement<br />
of the native epithelial cells by a single or<br />
3-5 layers of mildly atypical cells.<br />
Intraductal proliferative lesions<br />
65