Invasive breast carcinoma - IARC
Invasive breast carcinoma - IARC
Invasive breast carcinoma - IARC
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
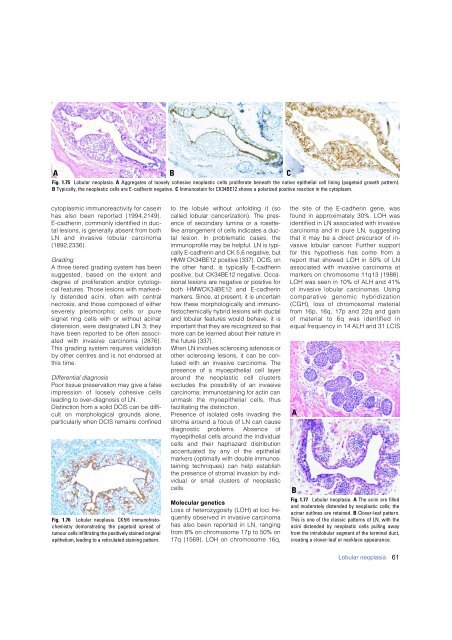
A<br />
B<br />
C<br />
Fig. 1.75 Lobular neoplasia. A Aggregates of loosely cohesive neoplastic cells proliferate beneath the native epithelial cell lining (pagetoid growth pattern).<br />
B Typically, the neoplastic cells are E-cadherin negative. C Immunostain for CK34BE12 shows a polarized positive reaction in the cytoplasm.<br />
cytoplasmic immunoreactivity for casein<br />
has also been re p o rted {1994,2149}.<br />
E-cadherin, commonly identified in ductal<br />
lesions, is generally absent from both<br />
LN and invasive lobular carc i n o m a<br />
{ 1 8 9 2 , 2 3 3 6 } .<br />
Grading<br />
A three tiered grading system has been<br />
suggested, based on the extent and<br />
d e g ree of proliferation and/or cytological<br />
features. Those lesions with markedly<br />
distended acini, often with central<br />
n e c rosis, and those composed of either<br />
s e v e rely pleomorphic cells or pure<br />
signet ring cells with or without acinar<br />
distension, were designated LIN 3; they<br />
have been re p o rted to be often associated<br />
with invasive <strong>carcinoma</strong> {2876}.<br />
This grading system re q u i res validation<br />
by other centres and is not endorsed at<br />
this time.<br />
Differential diagnosis<br />
Poor tissue preservation may give a false<br />
i m p ression of loosely cohesive cells<br />
leading to over-diagnosis of LN.<br />
Distinction from a solid DCIS can be diff i-<br />
cult on morphological grounds alone,<br />
p a rticularly when DCIS remains confined<br />
Fig. 1.76 Lobular neoplasia. CK5/6 immunohistochemistry<br />
demonstrating the pagetoid spread of<br />
tumour cells infiltrating the positively stained original<br />
epithelium, leading to a reticulated staining pattern.<br />
to the lobule without unfolding it (so<br />
called lobular cancerization). The pre s-<br />
ence of secondary lumina or a ro s e t t e -<br />
like arrangement of cells indicates a ductal<br />
lesion. In problematic cases, the<br />
i m m u n o p rofile may be helpful. LN is typically<br />
E-cadherin and CK 5,6 negative, but<br />
HMW CK34BE12 positive {337}. DCIS, on<br />
the other hand, is typically E-cadherin<br />
positive, but CK34BE12 negative. Occasional<br />
lesions are negative or positive for<br />
both HMWCK34BE12 and E-cadherin<br />
markers. Since, at present, it is uncert a i n<br />
how these morphologically and immunohistochemically<br />
hybrid lesions with ductal<br />
and lobular features would behave, it is<br />
i m p o rtant that they are recognized so that<br />
m o re can be learned about their nature in<br />
the future {337}.<br />
When LN involves sclerosing adenosis or<br />
other sclerosing lesions, it can be confused<br />
with an invasive <strong>carcinoma</strong>. The<br />
presence of a myoepithelial cell layer<br />
a round the neoplastic cell clusters<br />
excludes the possibility of an invasive<br />
<strong>carcinoma</strong>; immunostaining for actin can<br />
unmask the myoepithelial cells, thus<br />
facilitating the distinction.<br />
Presence of isolated cells invading the<br />
stroma around a focus of LN can cause<br />
diagnostic problems. Absence of<br />
myoepithelial cells around the individual<br />
cells and their haphazard distribution<br />
accentuated by any of the epithelial<br />
markers (optimally with double immunostaining<br />
techniques) can help establish<br />
the presence of stromal invasion by individual<br />
or small clusters of neoplastic<br />
cells.<br />
Molecular genetics<br />
Loss of heterozygosity (LOH) at loci frequently<br />
observed in invasive carc i n o m a<br />
has also been re p o rted in LN, ranging<br />
f rom 8% on chromosome 17p to 50% on<br />
17q {1569}. LOH on chromosome 16q,<br />
the site of the E-cadherin gene, was<br />
found in approximately 30%. LOH was<br />
identified in LN associated with invasive<br />
c a rcinoma and in pure LN, suggesting<br />
that it may be a direct precursor of invasive<br />
lobular cancer. Further support<br />
for this hypothesis has come from a<br />
re p o rt that showed LOH in 50% of LN<br />
associated with invasive <strong>carcinoma</strong> at<br />
markers on chromosome 11q13 {1988}.<br />
LOH was seen in 10% of ALH and 41%<br />
of invasive lobular <strong>carcinoma</strong>s. Using<br />
comparative genomic hybridization<br />
(CGH), loss of chromosomal material<br />
f rom 16p, 16q, 17p and 22q and gain<br />
of material to 6q was identified in<br />
equal frequency in 14 ALH and 31 LCIS<br />
A<br />
B<br />
Fig. 1.77 Lobular neoplasia. A The acini are filled<br />
and moderately distended by neoplastic cells; the<br />
acinar outlines are retained. B Clover-leaf pattern.<br />
This is one of the classic patterns of LN, with the<br />
acini distended by neoplastic cells pulling away<br />
from the intralobular segment of the terminal duct,<br />
creating a clover-leaf or necklace appearance.<br />
Lobular neoplasia<br />
61