Invasive breast carcinoma - IARC
Invasive breast carcinoma - IARC
Invasive breast carcinoma - IARC
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
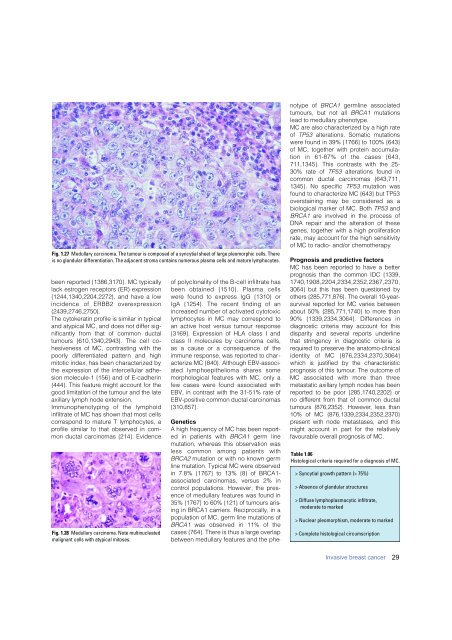
Fig. 1.27 Medullary <strong>carcinoma</strong>. The tumour is composed of a syncytial sheet of large pleomorphic cells. There<br />
is no glandular differentiation. The adjacent stroma contains numerous plasma cells and mature lymphocytes.<br />
been reported {1386,3170}. MC typically<br />
lack estrogen receptors (ER) expression<br />
{1244,1340,2204,2272}, and have a low<br />
incidence of ERBB2 overe x p re s s i o n<br />
{2439,2746,2750}.<br />
The cytokeratin profile is similar in typical<br />
and atypical MC, and does not differ significantly<br />
from that of common ductal<br />
tumours {610,1340,2943}. The cell cohesiveness<br />
of MC, contrasting with the<br />
poorly diff e rentiated pattern and high<br />
mitotic index, has been characterized by<br />
the expression of the intercellular adhesion<br />
molecule-1 {156} and of E-cadherin<br />
{444}. This feature might account for the<br />
good limitation of the tumour and the late<br />
axillary lymph node extension.<br />
Immunophenotyping of the lymphoid<br />
infiltrate of MC has shown that most cells<br />
correspond to mature T lymphocytes, a<br />
profile similar to that observed in common<br />
ductal <strong>carcinoma</strong>s {214}. Evidence<br />
Fig. 1.28 Medullary <strong>carcinoma</strong>. Note multinucleated<br />
malignant cells with atypical mitoses.<br />
of polyclonality of the B-cell infiltrate has<br />
been obtained {1510}. Plasma cells<br />
w e re found to express IgG {1310} or<br />
IgA {1254}. The recent finding of an<br />
i n c reased number of activated cytotoxic<br />
lymphocytes in MC may correspond to<br />
an active host versus tumour response<br />
{3169}. Expression of HLA class I and<br />
class II molecules by <strong>carcinoma</strong> cells,<br />
as a cause or a consequence of the<br />
immune response, was reported to characterize<br />
MC {840}. Although EBV-associated<br />
lymphoepithelioma shares some<br />
morphological features with MC, only a<br />
few cases were found associated with<br />
EBV, in contrast with the 31-51% rate of<br />
EBV-positive common ductal <strong>carcinoma</strong>s<br />
{310,857}.<br />
Genetics<br />
A high frequency of MC has been reported<br />
in patients with BRCA1 germ line<br />
mutation, whereas this observation was<br />
less common among patients with<br />
BRCA2 mutation or with no known germ<br />
line mutation. Typical MC were observed<br />
in 7.8% {1767} to 13% {8} of BRCA1-<br />
associated <strong>carcinoma</strong>s, versus 2% in<br />
control populations. However, the presence<br />
of medullary features was found in<br />
35% {1767} to 60% {121} of tumours arising<br />
in BRCA1 carriers. Reciprocally, in a<br />
population of MC, germ line mutations of<br />
BRCA1 was observed in 11% of the<br />
cases {764}. There is thus a large overlap<br />
between medullary features and the phenotype<br />
of BRCA1 germline associated<br />
tumours, but not all BRCA1 mutations<br />
lead to medullary phenotype.<br />
MC are also characterized by a high rate<br />
of TP53 alterations. Somatic mutations<br />
were found in 39% {1766} to 100% {643}<br />
of MC, together with protein accumulation<br />
in 61-87% of the cases {643,<br />
711,1345}. This contrasts with the 25-<br />
30% rate of TP53 alterations found in<br />
common ductal <strong>carcinoma</strong>s {643,711,<br />
1345}. No specific TP53 mutation was<br />
found to characterize MC {643} but TP53<br />
overstaining may be considered as a<br />
biological marker of MC. Both TP53 and<br />
BRCA1 are involved in the process of<br />
DNA repair and the alteration of these<br />
genes, together with a high proliferation<br />
rate, may account for the high sensitivity<br />
of MC to radio- and/or chemotherapy.<br />
Prognosis and predictive factors<br />
MC has been re p o rted to have a better<br />
p rognosis than the common IDC {1339,<br />
1 7 4 0 , 1 9 0 8 , 2 2 0 4 , 2 3 3 4 , 2 3 5 2 , 2 3 6 7 , 2 3 7 0 ,<br />
3064} but this has been questioned by<br />
others {285,771,876}. The overall 10-yearsurvival<br />
re p o rted for MC varies between<br />
about 50% {285,771,1740} to more than<br />
90% {1339,2334,3064}. Diff e rences in<br />
diagnostic criteria may account for this<br />
disparity and several re p o rts underline<br />
that stringency in diagnostic criteria is<br />
re q u i red to preserve the anatomo-clinical<br />
identity of MC {876,2334,2370,3064}<br />
which is justified by the characteristic<br />
p rognosis of this tumour. The outcome of<br />
MC associated with more than thre e<br />
metastatic axillary lymph nodes has been<br />
re p o rted to be poor {285,1740,2202} or<br />
no diff e rent from that of common ductal<br />
tumours {876,2352}. However, less than<br />
10% of MC {876,1339,2334,2352,2370}<br />
p resent with node metastases, and this<br />
might account in part for the re l a t i v e l y<br />
favourable overall prognosis of MC.<br />
Table 1.06<br />
Histological criteria required for a diagnosis of MC.<br />
> Syncytial growth pattern (> 75%)<br />
> Absence of glandular structures<br />
> Diffuse lymphoplasmacytic infiltrate,<br />
moderate to marked<br />
> Nuclear pleomorphism, moderate to marked<br />
> Complete histological circumscription<br />
<strong>Invasive</strong> <strong>breast</strong> cancer<br />
29