2. ENVIRONMENTAL ChEMISTRy & TEChNOLOGy 2.1. Lectures
2. ENVIRONMENTAL ChEMISTRy & TEChNOLOGy 2.1. Lectures
2. ENVIRONMENTAL ChEMISTRy & TEChNOLOGy 2.1. Lectures
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
Chem. Listy, 102, s265–s1311 (2008) Environmental Chemistry & Technology<br />
areas. The surface areas are relatively low because they were<br />
measured only for comparison of the changes. These samples<br />
were heated only at temperature 105 o C for two hours. In our<br />
next study the surface area will be measured also after heating<br />
at the higher temperatures up to 400 °C.<br />
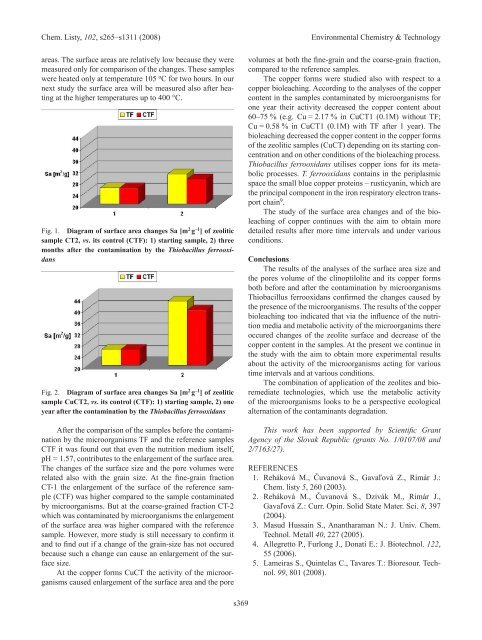
Fig. 1. Diagram of surface area changes Sa [m 2 g –1 ] of zeolitic<br />
sample CT2, vs. its control (CTF): 1) starting sample, 2) three<br />
months after the contamination by the Thiobacillus ferrooxidans<br />
Fig. <strong>2.</strong> Diagram of surface area changes Sa [m 2 g –1 ] of zeolitic<br />
sample CuCT2, vs. its control (CTF): 1) starting sample, 2) one<br />
year after the contamination by the Thiobacillus ferrooxidans<br />
After the comparison of the samples before the contamination<br />
by the microorganisms TF and the reference samples<br />
CTF it was found out that even the nutrition medium itself,<br />
pH = 1.57, contributes to the enlargement of the surface area.<br />
The changes of the surface size and the pore volumes were<br />
related also with the grain size. At the fine-grain fraction<br />
CT-1 the enlargement of the surface of the reference sample<br />
(CTF) was higher compared to the sample contaminated<br />
by microorganisms. But at the coarse-grained fraction CT-2<br />
which was contaminated by microorganisms the enlargement<br />
of the surface area was higher compared with the reference<br />
sample. However, more study is still necessary to confirm it<br />
and to find out if a change of the grain-size has not occured<br />
because such a change can cause an enlargement of the surface<br />
size.<br />
At the copper forms CuCT the activity of the microorganisms<br />
caused enlargement of the surface area and the pore<br />
s369<br />
volumes at both the fine-grain and the coarse-grain fraction,<br />
compared to the reference samples.<br />
The copper forms were studied also with respect to a<br />
copper bioleaching. According to the analyses of the copper<br />
content in the samples contaminated by microorganisms for<br />
one year their activity decreased the copper content about<br />
60–75 % (e.g. Cu = <strong>2.</strong>17 % in CuCT1 (0.1M) without TF;<br />
Cu = 0.58 % in CuCT1 (0.1M) with TF after 1 year). The<br />
bioleaching decreased the copper content in the copper forms<br />
of the zeolitic samples (CuCT) depending on its starting concentration<br />
and on other conditions of the bioleaching process.<br />
Thiobacillus ferrooxidans utilises copper ions for its metabolic<br />
processes. T. ferrooxidans contains in the periplasmic<br />
space the small blue copper proteins – rusticyanin, which are<br />
the principal component in the iron respiratory electron transport<br />
chain 9 .<br />
The study of the surface area changes and of the bioleaching<br />
of copper continues with the aim to obtain more<br />
detailed results after more time intervals and under various<br />
conditions.<br />
Conclusions<br />
The results of the analyses of the surface area size and<br />
the pores volume of the clinoptilolite and its copper forms<br />
both before and after the contamination by microorganisms<br />
Thiobacillus ferrooxidans confirmed the changes caused by<br />
the presence of the microorganisms. The results of the copper<br />
bioleaching too indicated that via the influence of the nutrition<br />
media and metabolic activity of the microorganims there<br />
occured changes of the zeolite surface and decrease of the<br />
copper content in the samples. At the present we continue in<br />
the study with the aim to obtain more experimental results<br />
about the activity of the microorganisms acting for various<br />
time intervals and at various conditions.<br />
The combination of application of the zeolites and bioremediate<br />
technologies, which use the metabolic activity<br />
of the microorganisms looks to be a perspective ecological<br />
alternation of the contaminants degradation.<br />
This work has been supported by Scientific Grant<br />
Agency of the Slovak Republic (grants No. 1/0107/08 and<br />
2/7163/27).<br />
REFEREnCES<br />
1. Reháková M., Čuvanová S., Gavaľová Z., Rimár J.:<br />
Chem. listy 5, 260 (2003).<br />
<strong>2.</strong> Reháková M., Čuvanová S., Dzivák M., Rimár J.,<br />
Gavaľová Z.: Curr. Opin. Solid State Mater. Sci. 8, 397<br />
(2004).<br />
3. Masud Hussain S., Anantharaman n.: J. Univ. Chem.<br />
Technol. Metall 40, 227 (2005).<br />
4. Allegretto P., Furlong J., Donati E.: J. Biotechnol. 122,<br />
55 (2006).<br />
5. Lameiras S., Quintelas C., Tavares T.: Bioresour. Technol.<br />
99, 801 (2008).