Experimental infection and protection against ... - TI Pharma
Experimental infection and protection against ... - TI Pharma Experimental infection and protection against ... - TI Pharma
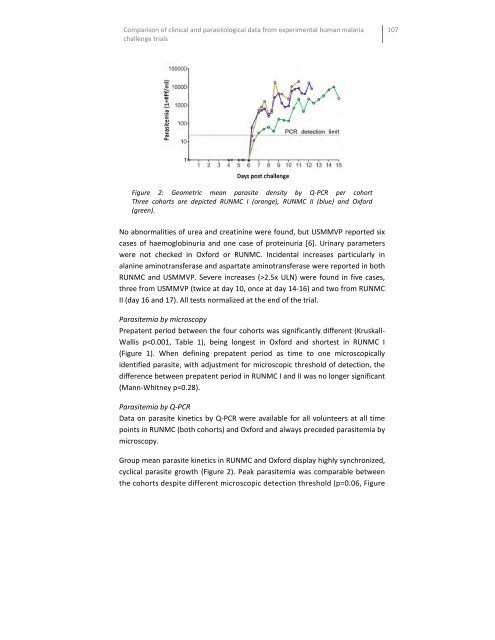
Comparison of clinical and parasitological data from experimental human malaria challenge trials Figure 2: Geometric mean parasite density by Q-PCR per cohort Three cohorts are depicted RUNMC I (orange), RUNMC II (blue) and Oxford (green). No abnormalities of urea and creatinine were found, but USMMVP reported six cases of haemoglobinuria and one case of proteinuria [6]. Urinary parameters were not checked in Oxford or RUNMC. Incidental increases particularly in alanine aminotransferase and aspartate aminotransferase were reported in both RUNMC and USMMVP. Severe increases (>2.5x ULN) were found in five cases, three from USMMVP (twice at day 10, once at day 14-16) and two from RUNMC II (day 16 and 17). All tests normalized at the end of the trial. Parasitemia by microscopy Prepatent period between the four cohorts was significantly different (Kruskall- Wallis p
108 Chapter 5 Figure 3. Statistics of Q-PCR parasitemia per cohort. Peak parasitemia (A, one-way ANOVA p=0.13), geometric mean parasitemia during the first blood stage parasite multiplication cycle, day 6.5 to 8.5 (B, one-way ANOVA p45 was assigned a parasitemia of 10 parasites/ml. 3A), although there seemed to be a trend towards higher parasitemia at RUNMC after the threshold for microscopic parasite detection changed. The first blood stage parasite growth cycle was significantly lower in Oxford as compared to RUNMC (p
- Page 58 and 59: Chapter 3 Humoral immune responses
- Page 60 and 61: Humoral immune responses to a singl
- Page 62 and 63: Humoral immune responses to a singl
- Page 64 and 65: Humoral immune responses to a singl
- Page 66 and 67: Humoral immune responses to a singl
- Page 68 and 69: Humoral immune responses to a singl
- Page 70 and 71: Humoral immune responses to a singl
- Page 72 and 73: Humoral immune responses to a singl
- Page 74 and 75: Humoral immune responses to a singl
- Page 76 and 77: Humoral immune responses to a singl
- Page 78 and 79: Humoral immune responses to a singl
- Page 80: Humoral immune responses to a singl
- Page 83 and 84: Chapter 4 82 Abstract The functiona
- Page 85 and 86: 84 Chapter 4 Figure 1. Schematic re
- Page 87 and 88: 86 Chapter 4 Concentration (mg/ml,
- Page 89 and 90: 88 Chapter 4 Figure 4: Correlation
- Page 91 and 92: 90 Chapter 4 positively correlated
- Page 93 and 94: 92 Chapter 4 Acknowledgements The a
- Page 95 and 96: 94 Chapter 4 determinant of subsequ
- Page 98 and 99: Chapter 5 Comparison of clinical an
- Page 100 and 101: Comparison of clinical and parasito
- Page 102 and 103: Comparison of clinical and parasito
- Page 104 and 105: Comparison of clinical and parasito
- Page 106 and 107: Comparison of clinical and parasito
- Page 110 and 111: Comparison of clinical and parasito
- Page 112 and 113: Comparison of clinical and parasito
- Page 114 and 115: Comparison of clinical and parasito
- Page 116 and 117: Comparison of clinical and parasito
- Page 118 and 119: Chapter 6 Efficacy of pre-erythrocy
- Page 120 and 121: Efficacy of pre-erythrocytic and bl
- Page 122 and 123: Efficacy of pre-erythrocytic and bl
- Page 124 and 125: Efficacy of pre-erythrocytic and bl
- Page 126 and 127: Efficacy of pre-erythrocytic and bl
- Page 128 and 129: Efficacy of pre-erythrocytic and bl
- Page 130 and 131: Chapter 7 NF135.C10: a new Plasmodi
- Page 132 and 133: NF135.C10: a new Plasmodium falcipa
- Page 134 and 135: NF135.C10: a new Plasmodium falcipa
- Page 136 and 137: NF135.C10: a new Plasmodium falcipa
- Page 138 and 139: NF135.C10: a new Plasmodium falcipa
- Page 140 and 141: NF135.C10: a new Plasmodium falcipa
- Page 142 and 143: NF135.C10: a new Plasmodium falcipa
- Page 144 and 145: NF135.C10: a new Plasmodium falcipa
- Page 146 and 147: NF135.C10: a new Plasmodium falcipa
- Page 148 and 149: NF135.C10: a new Plasmodium falcipa
- Page 150 and 151: NF135.C10: a new Plasmodium falcipa
- Page 152 and 153: Chapter 8 Induction of malaria in v
- Page 154 and 155: Induction of malaria in volunteers
- Page 156 and 157: Induction of malaria in volunteers
Comparison of clinical <strong>and</strong> parasitological data from experimental human malaria<br />
challenge trials<br />
Figure 2: Geometric mean parasite density by Q-PCR per cohort<br />
Three cohorts are depicted RUNMC I (orange), RUNMC II (blue) <strong>and</strong> Oxford<br />
(green).<br />
No abnormalities of urea <strong>and</strong> creatinine were found, but USMMVP reported six<br />
cases of haemoglobinuria <strong>and</strong> one case of proteinuria [6]. Urinary parameters<br />
were not checked in Oxford or RUNMC. Incidental increases particularly in<br />
alanine aminotransferase <strong>and</strong> aspartate aminotransferase were reported in both<br />
RUNMC <strong>and</strong> USMMVP. Severe increases (>2.5x ULN) were found in five cases,<br />
three from USMMVP (twice at day 10, once at day 14-16) <strong>and</strong> two from RUNMC<br />
II (day 16 <strong>and</strong> 17). All tests normalized at the end of the trial.<br />
Parasitemia by microscopy<br />
Prepatent period between the four cohorts was significantly different (Kruskall-<br />
Wallis p