towards improved death receptor targeted therapy for ... - TI Pharma
towards improved death receptor targeted therapy for ... - TI Pharma
towards improved death receptor targeted therapy for ... - TI Pharma
You also want an ePaper? Increase the reach of your titles
YUMPU automatically turns print PDFs into web optimized ePapers that Google loves.
Combination study: TRAIL and TFT<br />
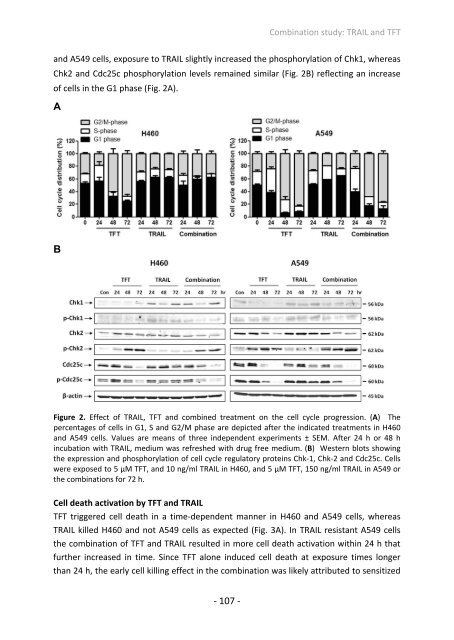
and A549 cells, exposure to TRAIL slightly increased the phosphorylation of Chk1, whereas<br />
Chk2 and Cdc25c phosphorylation levels remained similar (Fig. 2B) reflecting an increase<br />
of cells in the G1 phase (Fig. 2A).<br />
A<br />
B<br />
Figure 2. Effect of TRAIL, TFT and combined treatment on the cell cycle progression. (A) The<br />
percentages of cells in G1, S and G2/M phase are depicted after the indicated treatments in H460<br />
and A549 cells. Values are means of three independent experiments ± SEM. After 24 h or 48 h<br />
incubation with TRAIL, medium was refreshed with drug free medium. (B) Western blots showing<br />
the expression and phosphorylation of cell cycle regulatory proteins Chk‐1, Chk‐2 and Cdc25c. Cells<br />
were exposed to 5 µM TFT, and 10 ng/ml TRAIL in H460, and 5 µM TFT, 150 ng/ml TRAIL in A549 or<br />
the combinations <strong>for</strong> 72 h.<br />
Cell <strong>death</strong> activation by TFT and TRAIL<br />
TFT triggered cell <strong>death</strong> in a time‐dependent manner in H460 and A549 cells, whereas<br />
TRAIL killed H460 and not A549 cells as expected (Fig. 3A). In TRAIL resistant A549 cells<br />
the combination of TFT and TRAIL resulted in more cell <strong>death</strong> activation within 24 h that<br />
further increased in time. Since TFT alone induced cell <strong>death</strong> at exposure times longer<br />
than 24 h, the early cell killing effect in the combination was likely attributed to sensitized<br />
‐ 107 ‐