A systematic review of the effectiveness of adalimumab
A systematic review of the effectiveness of adalimumab
A systematic review of the effectiveness of adalimumab
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
40<br />
Effectiveness<br />
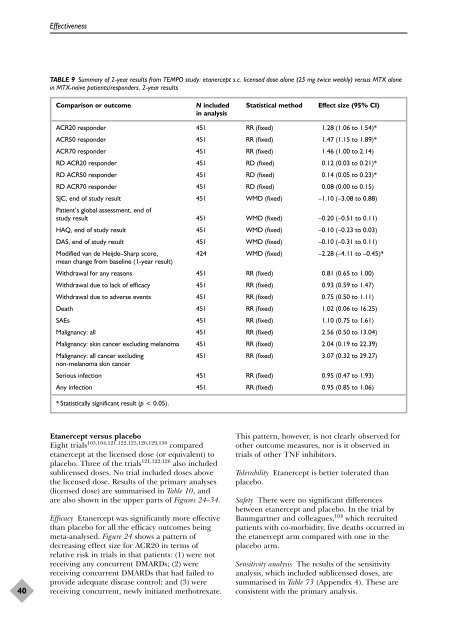
TABLE 9 Summary <strong>of</strong> 2-year results from TEMPO study: etanercept s.c. licensed dose alone (25 mg twice weekly) versus MTX alone<br />
in MTX-naïve patients/responders, 2-year results<br />
Comparison or outcome N included Statistical method Effect size (95% CI)<br />
in analysis<br />
ACR20 responder 451 RR (fixed) 1.28 (1.06 to 1.54)*<br />
ACR50 responder 451 RR (fixed) 1.47 (1.15 to 1.89)*<br />
ACR70 responder 451 RR (fixed) 1.46 (1.00 to 2.14)<br />
RD ACR20 responder 451 RD (fixed) 0.12 (0.03 to 0.21)*<br />
RD ACR50 responder 451 RD (fixed) 0.14 (0.05 to 0.23)*<br />
RD ACR70 responder 451 RD (fixed) 0.08 (0.00 to 0.15)<br />
SJC, end <strong>of</strong> study result<br />
Patient’s global assessment, end <strong>of</strong><br />
451 WMD (fixed) –1.10 (–3.08 to 0.88)<br />
study result 451 WMD (fixed) –0.20 (–0.51 to 0.11)<br />
HAQ, end <strong>of</strong> study result 451 WMD (fixed) –0.10 (–0.23 to 0.03)<br />
DAS, end <strong>of</strong> study result 451 WMD (fixed) –0.10 (–0.31 to 0.11)<br />
Modified van de Heijde–Sharp score,<br />
mean change from baseline (1-year result)<br />
424 WMD (fixed) –2.28 (–4.11 to –0.45)*<br />
Withdrawal for any reasons 451 RR (fixed) 0.81 (0.65 to 1.00)<br />
Withdrawal due to lack <strong>of</strong> efficacy 451 RR (fixed) 0.93 (0.59 to 1.47)<br />
Withdrawal due to adverse events 451 RR (fixed) 0.75 (0.50 to 1.11)<br />
Death 451 RR (fixed) 1.02 (0.06 to 16.25)<br />
SAEs 451 RR (fixed) 1.10 (0.75 to 1.61)<br />
Malignancy: all 451 RR (fixed) 2.56 (0.50 to 13.04)<br />
Malignancy: skin cancer excluding melanoma 451 RR (fixed) 2.04 (0.19 to 22.39)<br />
Malignancy: all cancer excluding<br />
non-melanoma skin cancer<br />
451 RR (fixed) 3.07 (0.32 to 29.27)<br />
Serious infection 451 RR (fixed) 0.95 (0.47 to 1.93)<br />
Any infection 451 RR (fixed) 0.95 (0.85 to 1.06)<br />
* Statistically significant result (p < 0.05).<br />
Etanercept versus placebo<br />
Eight trials 103,104,121,122,125,126,129,130 compared<br />
etanercept at <strong>the</strong> licensed dose (or equivalent) to<br />
placebo. Three <strong>of</strong> <strong>the</strong> trials 121,122,126 also included<br />
sublicensed doses. No trial included doses above<br />
<strong>the</strong> licensed dose. Results <strong>of</strong> <strong>the</strong> primary analyses<br />
(licensed dose) are summarised in Table 10, and<br />
are also shown in <strong>the</strong> upper parts <strong>of</strong> Figures 24–34.<br />
Efficacy Etanercept was significantly more effective<br />
than placebo for all <strong>the</strong> efficacy outcomes being<br />
meta-analysed. Figure 24 shows a pattern <strong>of</strong><br />
decreasing effect size for ACR20 in terms <strong>of</strong><br />
relative risk in trials in that patients: (1) were not<br />
receiving any concurrent DMARDs; (2) were<br />
receiving concurrent DMARDs that had failed to<br />
provide adequate disease control; and (3) were<br />
receiving concurrent, newly initiated methotrexate.<br />
This pattern, however, is not clearly observed for<br />
o<strong>the</strong>r outcome measures, nor is it observed in<br />
trials <strong>of</strong> o<strong>the</strong>r TNF inhibitors.<br />
Tolerability Etanercept is better tolerated than<br />
placebo.<br />
Safety There were no significant differences<br />
between etanercept and placebo. In <strong>the</strong> trial by<br />
Baumgartner and colleagues, 104 which recruited<br />
patients with co-morbidity, five deaths occurred in<br />
<strong>the</strong> etanercept arm compared with one in <strong>the</strong><br />
placebo arm.<br />
Sensitivity analysis The results <strong>of</strong> <strong>the</strong> sensitivity<br />
analysis, which included sublicensed doses, are<br />
summarised in Table 73 (Appendix 4). These are<br />
consistent with <strong>the</strong> primary analysis.