ehr onc final certification - Department of Health Care Services
ehr onc final certification - Department of Health Care Services ehr onc final certification - Department of Health Care Services
moved the CPOE certification criteria to the general certification criteria section. Rather, we have kept the certification criteria for CPOE separate because we anticipate that these certification criteria could in the future include different requirements, specific to the settings for which Complete EHRs and EHR Modules are developed. Comment. A commenter repeated a question it raised with respect to CPOE for eligible professionals. The commenter requested that we clarify whether only imaging and radiology reports were intended to be included in this capability, or, if we intended to include the images themselves in addition to the imaging reports as part of the certification criteria. The commenter recommended that we further clarify the criterion and requested that the DICOM standard be adopted in the initial set of standards, as an essential step in meeting the CPOE capability. this issue. Response. We refer this commenter to our previous response above regarding §170.306(b) - Record demographics Meaningful Use Stage 1 Objective Record demographics • preferred language • gender • race • ethnicity • date of birth • date and preliminary cause of death in the event of mortality in the eligible hospital or CAH Meaningful Use Stage 1 Measure More than 50% of all unique patients seen by the EP or admitted to the eligible hospital’s or CAH’s inpatient or emergency department (POS 21 or 23) have demographics recorded as structured data Page 162 of 228 Certification Criterion Interim Final Rule Text: Enable a user to electronically record, modify, and retrieve patient demographic data including preferred language, insurance type, gender, race, ethnicity, date of birth, and date and cause of death in the event of mortality. Final Rule Text: §170.306(b) Record demographics. Enable a user to electronically record, modify, and retrieve patient demographic data including preferred language, gender, race, ethnicity, date of birth, and date and preliminary cause of death in the event of mortality. Enable race and ethnicity to be recorded in accordance with the standard specified at §170.207(f). Many commenters expressed the same comments with respect to this certification criterion as they did for the record demographics certification criterion for Complete
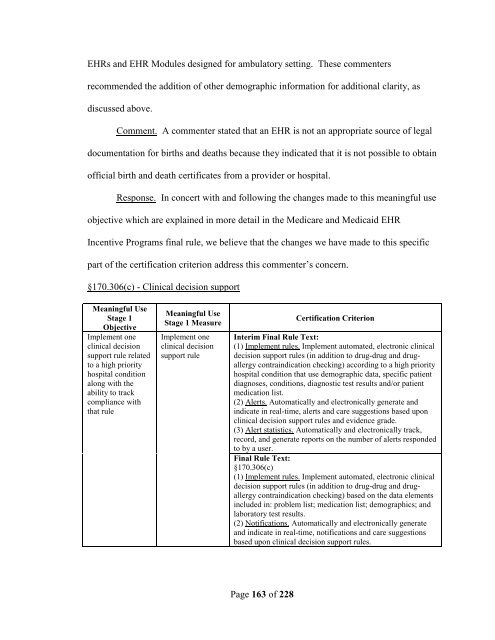
EHRs and EHR Modules designed for ambulatory setting. These commenters recommended the addition of other demographic information for additional clarity, as discussed above. Comment. A commenter stated that an EHR is not an appropriate source of legal documentation for births and deaths because they indicated that it is not possible to obtain official birth and death certificates from a provider or hospital. Response. In concert with and following the changes made to this meaningful use objective which are explained in more detail in the Medicare and Medicaid EHR Incentive Programs final rule, we believe that the changes we have made to this specific part of the certification criterion address this commenter’s concern. §170.306(c) - Clinical decision support Meaningful Use Stage 1 Objective Implement one clinical decision support rule related to a high priority hospital condition along with the ability to track compliance with that rule Meaningful Use Stage 1 Measure Implement one clinical decision support rule Page 163 of 228 Certification Criterion Interim Final Rule Text: (1) Implement rules. Implement automated, electronic clinical decision support rules (in addition to drug-drug and drugallergy contraindication checking) according to a high priority hospital condition that use demographic data, specific patient diagnoses, conditions, diagnostic test results and/or patient medication list. (2) Alerts. Automatically and electronically generate and indicate in real-time, alerts and care suggestions based upon clinical decision support rules and evidence grade. (3) Alert statistics. Automatically and electronically track, record, and generate reports on the number of alerts responded to by a user. Final Rule Text: §170.306(c) (1) Implement rules. Implement automated, electronic clinical decision support rules (in addition to drug-drug and drugallergy contraindication checking) based on the data elements included in: problem list; medication list; demographics; and laboratory test results. (2) Notifications. Automatically and electronically generate and indicate in real-time, notifications and care suggestions based upon clinical decision support rules.
- Page 111 and 112: Response. As discussed above, we ha
- Page 113 and 114: SHA-1 and other secure hash algorit
- Page 115 and 116: misinterpreted our example and stat
- Page 117 and 118: Other commenters also expressed con
- Page 119 and 120: eferenced in FIPS 140-2 Annex A, wh
- Page 121 and 122: of the most secure encryption algor
- Page 123 and 124: the disclosure was made (recipient)
- Page 125 and 126: Use CPOE for medication orders dire
- Page 127 and 128: equire EHRs to build custom interfa
- Page 129 and 130: esult, we do not believe that this
- Page 131 and 132: was needed before RxNorm could be a
- Page 133 and 134: • MDDB - Medi-Span Master Drug Da
- Page 135 and 136: Response. We do not believe that it
- Page 137 and 138: Send reminders to patients per pati
- Page 139 and 140: specified data elements and CMS’s
- Page 141 and 142: what would qualify as a "response."
- Page 143 and 144: Comment. A commenter recommended th
- Page 145 and 146: in accordance with one of the adopt
- Page 147 and 148: Comments. Many commenters suggested
- Page 149 and 150: flexibility in this certification c
- Page 151 and 152: productive, confusing, time-consumi
- Page 153 and 154: include in this initial set. Accord
- Page 155 and 156: to the HITSP C32 implementation spe
- Page 157 and 158: Response. Again, we do not believe
- Page 159 and 160: e achieved without these and recomm
- Page 161: electronically record, store, retri
- Page 165 and 166: §170.205(a)(2)(iii); and (v) The s
- Page 167 and 168: Response. We disagree, as doing so
- Page 169 and 170: Dental Terminology as a condition o
- Page 171 and 172: However, we do not preclude Complet
- Page 173 and 174: ability of CCD and CCR to support t
- Page 175 and 176: ability to receive these reports. M
- Page 177 and 178: commenters acknowledged and express
- Page 179 and 180: a meaningful use objective they wou
- Page 181 and 182: CMS and ONC had worked together to
- Page 183 and 184: The eligible professional or eligib
- Page 185 and 186: EHR technology with the needs of us
- Page 187 and 188: The RFA requires agencies to analyz
- Page 189 and 190: values seemed low and that the gap
- Page 191 and 192: commenter provided), our assumption
- Page 193 and 194: absolute low we estimated for a per
- Page 195 and 196: number of previously CCHIT-certifie
- Page 197 and 198: for Certification Low High Page 197
- Page 199 and 200: Finally, the third type of cost we
- Page 201 and 202: 2012 15% $10.10 $30.80 $20.45 3-Yea
- Page 203 and 204: The RFA requires agencies to analyz
- Page 205 and 206: The Office of Management and Budget
- Page 207 and 208: The standards and implementation sp
- Page 209 and 210: The Secretary adopts the following
- Page 211 and 212: any edition other than that specifi
EHRs and EHR Modules designed for ambulatory setting. These commenters<br />
recommended the addition <strong>of</strong> other demographic information for additional clarity, as<br />
discussed above.<br />
Comment. A commenter stated that an EHR is not an appropriate source <strong>of</strong> legal<br />
documentation for births and deaths because they indicated that it is not possible to obtain<br />
<strong>of</strong>ficial birth and death certificates from a provider or hospital.<br />
Response. In c<strong>onc</strong>ert with and following the changes made to this meaningful use<br />
objective which are explained in more detail in the Medicare and Medicaid EHR<br />
Incentive Programs <strong>final</strong> rule, we believe that the changes we have made to this specific<br />
part <strong>of</strong> the <strong>certification</strong> criterion address this commenter’s c<strong>onc</strong>ern.<br />
§170.306(c) - Clinical decision support<br />
Meaningful Use<br />
Stage 1<br />
Objective<br />
Implement one<br />
clinical decision<br />
support rule related<br />
to a high priority<br />
hospital condition<br />
along with the<br />
ability to track<br />
compliance with<br />
that rule<br />
Meaningful Use<br />
Stage 1 Measure<br />
Implement one<br />
clinical decision<br />
support rule<br />
Page 163 <strong>of</strong> 228<br />
Certification Criterion<br />
Interim Final Rule Text:<br />
(1) Implement rules. Implement automated, electronic clinical<br />
decision support rules (in addition to drug-drug and drugallergy<br />
contraindication checking) according to a high priority<br />
hospital condition that use demographic data, specific patient<br />
diagnoses, conditions, diagnostic test results and/or patient<br />
medication list.<br />
(2) Alerts. Automatically and electronically generate and<br />
indicate in real-time, alerts and care suggestions based upon<br />
clinical decision support rules and evidence grade.<br />
(3) Alert statistics. Automatically and electronically track,<br />
record, and generate reports on the number <strong>of</strong> alerts responded<br />
to by a user.<br />
Final Rule Text:<br />
§170.306(c)<br />
(1) Implement rules. Implement automated, electronic clinical<br />
decision support rules (in addition to drug-drug and drugallergy<br />
contraindication checking) based on the data elements<br />
included in: problem list; medication list; demographics; and<br />
laboratory test results.<br />
(2) Notifications. Automatically and electronically generate<br />
and indicate in real-time, notifications and care suggestions<br />
based upon clinical decision support rules.