Tuning Reactivity of Platinum(II) Complexes
Tuning Reactivity of Platinum(II) Complexes Tuning Reactivity of Platinum(II) Complexes
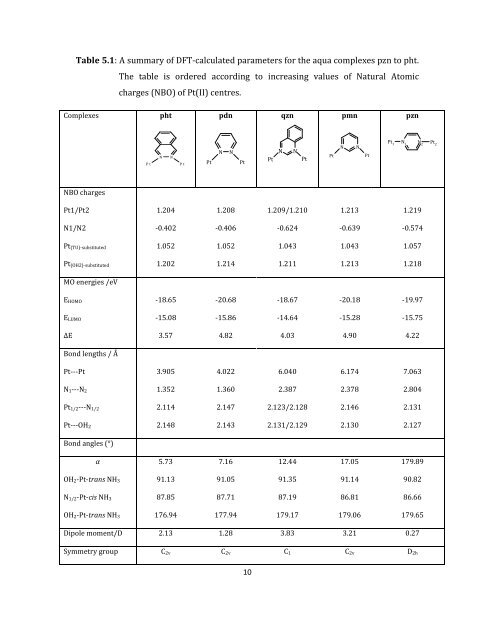
Table 5.1: A summary of DFT-calculated parameters for the aqua complexes pzn to pht. Complexes pht NBO charges The table is ordered according to increasing values of Natural Atomic charges (NBO) of Pt(II) centres. N N P t P t pdn N N Pt Pt 10 qzn N N Pt Pt pmn N N Pt Pt Pt1/Pt2 1.204 1.208 1.209/1.210 1.213 1.219 N1/N2 -0.402 -0.406 -0.624 -0.639 -0.574 Pt(TU)-substituted 1.052 1.052 1.043 1.043 1.057 Pt(OH2)-substituted 1.202 1.214 1.211 1.213 1.218 MO energies /eV EHOMO -18.65 -20.68 -18.67 -20.18 -19.97 ELUMO -15.08 -15.86 -14.64 -15.28 -15.75 ∆E 3.57 4.82 4.03 4.90 4.22 Bond lengths / Å Pt---Pt 3.905 4.022 6.040 6.174 7.063 N1---N2 1.352 1.360 2.387 2.378 2.804 Pt1/2---N1/2 2.114 2.147 2.123/2.128 2.146 2.131 Pt---OH2 2.148 2.143 2.131/2.129 2.130 2.127 Bond angles (°) α 5.73 7.16 12.44 17.05 179.89 OH2-Pt-trans NH3 91.13 91.05 91.35 91.14 90.82 N1/2-Pt-cis NH3 87.85 87.71 87.19 86.81 86.66 OH2-Pt-trans NH3 176.94 177.94 179.17 179.06 179.65 Dipole moment/D 2.13 1.28 3.83 3.21 0.27 Symmetry group C2v C2v C1 C2v D2h Pt 1 N 1 pzn N 2 Pt 2
Structure HOMO LUMO pzn (D2h) pmn (C2v) pdn (C2v) qzn (C1) pht (C2v) Figure 5.1: Frontier molecular orbitals HOMO–LUMO of the complexes 11
- Page 176 and 177: The decrease in reactivity of 2,6pz
- Page 178 and 179: Table 4.5: DFT calculated (NBO) cha
- Page 180 and 181: eaction proceeds via bimolecular pa
- Page 182 and 183: References 1 T. Storr, K. H.Thomson
- Page 184 and 185: 36 D. Jaganyi, D. Reddy, J.A. Gerte
- Page 186 and 187: Appendix 4 THE INFLUENCE OF THE PYR
- Page 188 and 189: Absorbance at 368. 0 nm 0. 0 8 0. 0
- Page 190 and 191: Table S4.3: Average observed rate c
- Page 192 and 193: k nd obs(2 ) , s-1 0.003 TU DMTU TM
- Page 194 and 195: Table S4.7: Average observed rate c
- Page 196 and 197: Table S4.8: Average observed rate c
- Page 198 and 199: k obs2 , s -1 2.40x10 -4 2.20x10 -4
- Page 200 and 201: Table S4.13: Average observed rate
- Page 202 and 203: Table S4.14: Average observed rate
- Page 204 and 205: Table S4.18: Average observed rate
- Page 206 and 207: k obs(1 st ) , s -1 0.06 0.04 0.02
- Page 208 and 209: Table S4.23: Average observed rate
- Page 210 and 211: ln(k 2(3 rd ) /T) -10.0 -10.5 -11.0
- Page 212 and 213: SpinWorks 2.5: 2,6 pznClO4 in D2O N
- Page 214 and 215: Table of Contents-5 Chapter 5 .....
- Page 216 and 217: List of Tables Table 5.1: A summary
- Page 218 and 219: 5.1 Introduction Multinuclear plati
- Page 220 and 221: onding. For this reason, pKa titrat
- Page 222 and 223: 400-300 cm -1): 3308, 3117, 3071 (N
- Page 224 and 225: 5.2.6 Spectrophotometric pKa Titrat
- Page 228 and 229: However, because the highest occupi
- Page 230 and 231: Table 5.2: Acid dissociation consta
- Page 232 and 233: Table 5.3: A summary of DFT calcula
- Page 234 and 235: H3N 6 eq TU 0 eq TU Ha NH3 Ha Cl TU
- Page 236 and 237: third step due to the trans-effect
- Page 238 and 239: [H 2 O-Pt-(NN)-Pt-OH 2 ] +4 [NU-Pt-
- Page 240 and 241: k obs(1st) / s -1 0.20 TU DMTU TMTU
- Page 242 and 243: thiourea nucleophile is large enoug
- Page 244 and 245: ln(k st 2(1 ) /T) -3 -4 -5 -6 -7 -8
- Page 246 and 247: is the same as the electron-withdra
- Page 248 and 249: associative mode of substitution me
- Page 250 and 251: 16 H. Ertürk, J. Maigut, R. Puchta
- Page 252 and 253: 43 (a) D. Jaganyi, A. Hofmann and R
- Page 254 and 255: 276 nm Absorbance 0 . 6 5 0 . 6 4 0
- Page 256 and 257: k obs(1 st ) , s -1 0.4 0.3 0.2 0.1
- Page 258 and 259: Table S5.5: Average observed rate c
- Page 260 and 261: ln(k 2(2 nd ) /T) -8.0 TU -8.5 -9.0
- Page 262 and 263: pzn PPM -1750.0 -1850.0 -1950.0 -20
- Page 264 and 265: Table S5.9: Average observed rate c
- Page 266 and 267: Figure S5.13: UV/Visible spectra fo
- Page 268 and 269: k obs(1 st ) in s -1 0.030 0.025 0.
- Page 270 and 271: Table S5.17: Average observed rate
- Page 272 and 273: ln(k 2(2 nd ) /T) -10 -11 -12 -13 -
- Page 274 and 275: 9.61 ppm Ha PPM 9.8 9.6 9.4 9.2 9.0
Table 5.1: A summary <strong>of</strong> DFT-calculated parameters for the aqua complexes pzn to pht.<br />
<strong>Complexes</strong> pht<br />
NBO charges<br />
The table is ordered according to increasing values <strong>of</strong> Natural Atomic<br />
charges (NBO) <strong>of</strong> Pt(<strong>II</strong>) centres.<br />
N N<br />
P t P t<br />
pdn<br />
N N<br />
Pt Pt<br />
10<br />
qzn<br />
N N<br />
Pt Pt<br />
pmn<br />
N N<br />
Pt Pt<br />
Pt1/Pt2 1.204 1.208 1.209/1.210 1.213 1.219<br />
N1/N2 -0.402 -0.406 -0.624 -0.639 -0.574<br />
Pt(TU)-substituted 1.052 1.052 1.043 1.043 1.057<br />
Pt(OH2)-substituted 1.202 1.214 1.211 1.213 1.218<br />
MO energies /eV<br />
EHOMO -18.65 -20.68 -18.67 -20.18 -19.97<br />
ELUMO -15.08 -15.86 -14.64 -15.28 -15.75<br />
∆E 3.57 4.82 4.03 4.90 4.22<br />
Bond lengths / Å<br />
Pt---Pt 3.905 4.022 6.040 6.174 7.063<br />
N1---N2 1.352 1.360 2.387 2.378 2.804<br />
Pt1/2---N1/2 2.114 2.147 2.123/2.128 2.146 2.131<br />
Pt---OH2 2.148 2.143 2.131/2.129 2.130 2.127<br />
Bond angles (°)<br />
α 5.73 7.16 12.44 17.05 179.89<br />
OH2-Pt-trans NH3 91.13 91.05 91.35 91.14 90.82<br />
N1/2-Pt-cis NH3 87.85 87.71 87.19 86.81 86.66<br />
OH2-Pt-trans NH3 176.94 177.94 179.17 179.06 179.65<br />
Dipole moment/D 2.13 1.28 3.83 3.21 0.27<br />
Symmetry group C2v C2v C1 C2v D2h<br />
Pt 1<br />
N 1<br />
pzn<br />
N 2<br />
Pt 2