laporan bersama perwakilan malaysia - Food Safety and Quality ...
laporan bersama perwakilan malaysia - Food Safety and Quality ...
laporan bersama perwakilan malaysia - Food Safety and Quality ...
Create successful ePaper yourself
Turn your PDF publications into a flip-book with our unique Google optimized e-Paper software.
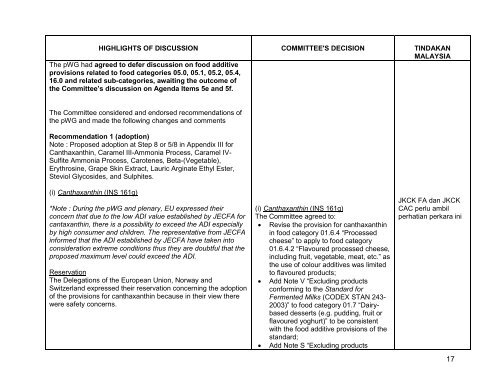
HIGHLIGHTS OF DISCUSSION<br />
The pWG had agreed to defer discussion on food additive<br />
provisions related to food categories 05.0, 05.1, 05.2, 05.4,<br />
16.0 <strong>and</strong> related sub-categories, awaiting the outcome of<br />
the Committee’s discussion on Agenda Items 5e <strong>and</strong> 5f.<br />
The Committee considered <strong>and</strong> endorsed recommendations of<br />
the pWG <strong>and</strong> made the following changes <strong>and</strong> comments<br />
Recommendation 1 (adoption)<br />
Note : Proposed adoption at Step 8 or 5/8 in Appendix III for<br />
Canthaxanthin, Caramel III-Ammonia Process, Caramel IV-<br />
Sulfite Ammonia Process, Carotenes, Beta-(Vegetable),<br />
Erythrosine, Grape Skin Extract, Lauric Arginate Ethyl Ester,<br />
Steviol Glycosides, <strong>and</strong> Sulphites.<br />
(i) Canthaxanthin (INS 161g)<br />
*Note : During the pWG <strong>and</strong> plenary, EU expressed their<br />
concern that due to the low ADI value established by JECFA for<br />
cantaxanthin, there is a possibility to exceed the ADI especially<br />
by high consumer <strong>and</strong> children. The representative from JECFA<br />
informed that the ADI established by JECFA have taken into<br />
consideration extreme conditions thus they are doubtful that the<br />
proposed maximum level could exceed the ADI.<br />
Reservation<br />
The Delegations of the European Union, Norway <strong>and</strong><br />
Switzerl<strong>and</strong> expressed their reservation concerning the adoption<br />
of the provisions for canthaxanthin because in their view there<br />
were safety concerns.<br />
COMMITTEE'S DECISION<br />
(i) Canthaxanthin (INS 161g)<br />
The Committee agreed to:<br />
Revise the provision for canthaxanthin<br />
in food category 01.6.4 “Processed<br />
cheese” to apply to food category<br />
01.6.4.2 “Flavoured processed cheese,<br />
including fruit, vegetable, meat, etc.” as<br />
the use of colour additives was limited<br />
to flavoured products;<br />
Add Note V “Excluding products<br />
conforming to the St<strong>and</strong>ard for<br />
Fermented Milks (CODEX STAN 243-<br />
2003)” to food category 01.7 “Dairybased<br />
desserts (e.g. pudding, fruit or<br />
flavoured yoghurt)” to be consistent<br />
with the food additive provisions of the<br />
st<strong>and</strong>ard;<br />
Add Note S “Excluding products<br />
TINDAKAN<br />
MALAYSIA<br />
JKCK FA dan JKCK<br />
CAC perlu ambil<br />
perhatian perkara ini<br />
17