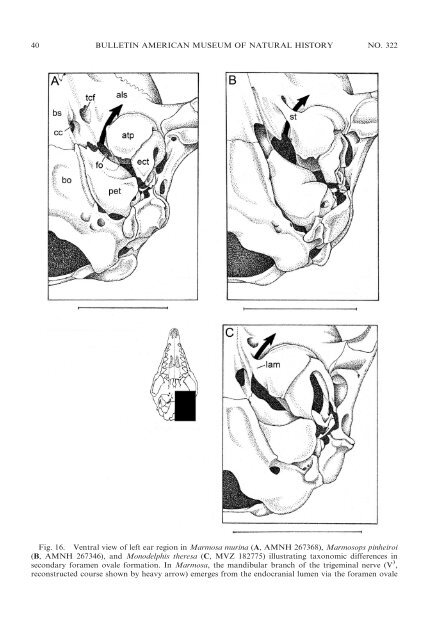
40 BULLETIN AMERICAN MUSEUM OF NATURAL HISTORY NO. 322 Fig. 16. Ventral view <strong>of</strong> left ear region in Marmosa murina (A, AMNH 267368), Marmosops pinheiroi (B, AMNH 267346), <strong>and</strong> Monodelphis theresa (C, MVZ 182775) illustrating taxonomic differences in secondary foramen ovale formation. In Marmosa, the m<strong>and</strong>ibular branch <strong>of</strong> the trigeminal nerve (V 3 , reconstructed course shown by heavy arrow) emerges from the endocranial lumen via the foramen ovale
2009 VOSS AND JANSA: DIDELPHID MARSUPIALS 41 opment; subadults <strong>and</strong> young adults show progressively more complete enclosure; <strong>and</strong> old adults (large specimens with heavily worn teeth) usually have completely enclosed foramina <strong>and</strong> canals (Abdala et al., 2001). All other <strong>didelphid</strong> taxa (Caluromys, Chacodelphys, Cryptonanuns, Glironia, Hyladelphys, Marmosa, Tlacuatzin, <strong>and</strong> most species <strong>of</strong> Monodelphis) lack secondary enclosures <strong>of</strong> the m<strong>and</strong>ibular nerve except as rare (usually unilateral) variants. Although many non<strong>didelphid</strong> <strong>marsupials</strong> (e.g., caenolestids, Dromiciops, most dasyuromorphians) have only a primary foramen ovale, secondary foramina ovales are also widely distributed. Unfortunately, these traits remain to be widely surveyed, <strong>and</strong> inconsistent terminology (discussed by Gaudin et al., 1996) makes it difficult to interpret some published descriptions <strong>of</strong> marsupial basicrania. Although other developmental mechanisms have obviously been responsible for secondary enclosures <strong>of</strong> the m<strong>and</strong>ibular nerve in some taxa (e.g., Phascolarctos), putative homologues <strong>of</strong> both <strong>didelphid</strong> patterns <strong>of</strong> secondary foramen formation can be recognized among certain Old World <strong>marsupials</strong>. At least some specimens <strong>of</strong> Thylacinus, for example, have a secondary foramen formed by an anteromedial bullar strut that spans the transverse canal foramen, whereas the secondary foramen ovale <strong>of</strong> Recent peramelemorphians (e.g., Echymipera, Perameles) is formed by a broad medial bullar lamina. EAR REGION: All <strong>didelphid</strong>s have an osteologically well-defined middle ear cavity or hypotympanic sinus (sensu van der Klaauw, 1931: 19). A cup-shaped tympanic process (or ‘‘wing’’) <strong>of</strong> the alisphenoid invariably forms the anterior part <strong>of</strong> the r floor <strong>of</strong> the middle ear, but this structure exhibits significant taxonomic variation in size. The alisphenoid tympanic process is large <strong>and</strong> extends far enough posteriorly to closely approximate or contact the rostral tympanic process <strong>of</strong> the petrosal in Caluromys <strong>and</strong> Caluromysiops (see Reig et al., 1987: fig. 44B, D). In other opossums, a distinct gap separates the alisphenoid tympanic process from the rostral tympanic process <strong>of</strong> the petrosal, such that at least part <strong>of</strong> the floor <strong>of</strong> the middle ear is membranous (e.g., in Marmosa <strong>and</strong> Monodelphis; Reig et al., 1987: fig. 41B, D). In Lestodelphys <strong>and</strong> Thylamys a rather indistinct medial process <strong>of</strong> the ectotympanic makes a small contribution to the floor <strong>of</strong> the middle ear, partially filling in the gap between the tympanic processes <strong>of</strong> the alisphenoid <strong>and</strong> petrosal (Reig et al., 1987: fig. 42B, D). The ro<strong>of</strong> <strong>of</strong> the hypotympanic sinus in all Recent <strong>didelphid</strong>s is almost exclusively formed by the alisphenoid, with the petrosal making only a small, <strong>of</strong>ten negligible, contribution. The <strong>didelphid</strong> squamosal does not participate in forming any part <strong>of</strong> the floor or ro<strong>of</strong> <strong>of</strong> the middle ear cavity. Caenolestids, dasyurids, peramelemorphians, <strong>and</strong> Dromiciops resemble <strong>didelphid</strong>s in that the alisphenoid forms most <strong>of</strong> the anterior part <strong>of</strong> the floor <strong>of</strong> the middle ear. However, whereas caenolestids <strong>and</strong> some peramelemorphians (e.g., Echymipera) have a small alisphenoid tympanic process that does not contact the rostral tympanic process <strong>of</strong> the petrosal, the alisphenoid tympanic process is large <strong>and</strong> broadly contacts the petrosal in dasyurids, Dromiciops, <strong>and</strong> other peramelemorphians (e.g., Perameles). The hypotympanic sinus ro<strong>of</strong> <strong>of</strong> dasyurids <strong>and</strong> (fo), which is bordered by the alisphenoid (als) <strong>and</strong> the petrosal (pet); the extracranial course <strong>of</strong> the nerve is unenclosed in this taxon. In Marmosops, however, the extracranial course <strong>of</strong> V 3 is partially enclosed by a bony strut (st) that extends from the anteromedial surface <strong>of</strong> the alisphenoid tympanic process (atp) across the transverse canal foramen (tcf); the nerve then emerges from a so-called secondary foramen ovale. Another kind <strong>of</strong> secondary enclosure is seen in Monodelphis, where the nerve emerges from a secondary foramen ovale formed by a medial lamina (lam) <strong>of</strong> the alisphenoid tympanic process. Other abbreviations: bs, basisphenoid; bo, basioccipital; cc, carotid canal; ect, ectotympanic. Scale bars 5 5 mm.