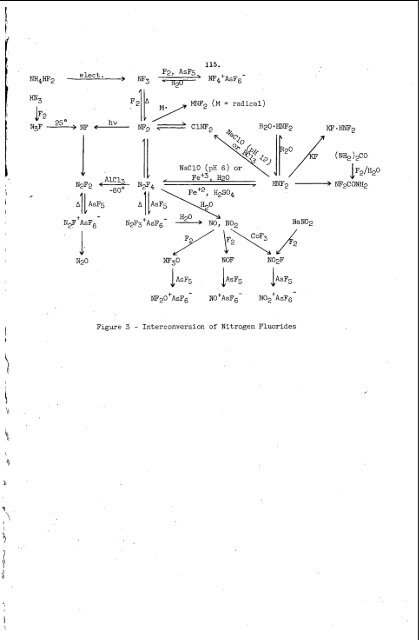
114. weaker than those in NF02. The W3O has good thermal <strong>and</strong> hydrolytic stability, but undergoes the fluoride abstraction reaction <strong>with</strong> strong Lewis acids, to give <strong>salt</strong>s such as E7?20+AsF6-, <strong>and</strong> adds to perfluoroolefins (under BF3 catalysis) to give stable RfONF2 compounds. It apparently reacts slowly <strong>with</strong> NO to give NOF. Difluoramine has an ammonia-like structure <strong>with</strong> the following parameters: N-H, 1.03-1.08; N-F, 1.38; FNF, 103', HNF, 102'; dipole moment, 1.93 D. It is best prepared from difluorourea (which is obtained by aqueous fluorination of urea) by treatment <strong>with</strong> H2SO4 at 90' or by the reaction of N2F4 <strong>with</strong> CgHgSH at 50'. The HNF2 is stable <strong>and</strong> can be stored, but the usual procedure is to generate it as needed <strong>and</strong> pass it directly into a reaction vessel, since it has a tendency to ex- plode when frozen. The reactions of HNF2 are usually complex, but it undergoes three general types of reactions as follows: oxidation, for example <strong>with</strong> aqueous &e+3 solution to give N2F4 (perhaps the NF2- ion is involved); reduction, as in the reaction <strong>with</strong> aqueous HI to give NH4F <strong>and</strong> HF; complex formation <strong>with</strong> ethers, Lewis acids, 2nd metal fluorides. Chlorodifluoramine ClNF2 is well known, C12NF <strong>and</strong> BrNF2 are known as unstable compounds, <strong>and</strong> the other halogen fluoramines appear to be very unstable. The CLNF2 (or BrNF2) can be prepared by the reaction of aqueous NaOCl (or NaOBr) <strong>with</strong> N,N-difluoroureas or N,N-dif!luorosulfuryl amide. The C 1 9 is prepared by the reaction of CLF <strong>with</strong> ClN3 at 25'C or <strong>with</strong> NaN3 at 0'. The CUW2 is stable but dissociates readily to give C1 atoms <strong>and</strong> NF2 radicals (which defines the reaction chemistry) while C12NF is explosively unstable in the liquid state. A few remaining N-F compounds such as NF2NO <strong>and</strong> N3F are of limited inter- est, but a number of inorganic compounds <strong>and</strong> a host of organic compounds have been prepared in recent years in which NF2 groups may be regarded as substituents, e.g., SF5NF2, C(NF2)4, C(NF)(NF2)2, CF3ONF2 <strong>and</strong> CFzN(O)=NF. Figure 3. A summary of the interconversions of the nitrogen fluorides is found in Chlorine Fluorides <strong>and</strong> Related Compounds The halogen fluorides of prime interest as propellant oxidizers have been C@,CLF5, C103F <strong>and</strong> BrF5, but a number of other halogen fluorides have been studied. Properties of halogen fluorides are summarized in Table 111. Chlorine monofluoride appears to have considerable ionic character as reflected in the The C1-F bond NMR chemical shift of @ = +441 ppm (vs. CFC13). distance is 1.63 d, the dipole moment, 0.88 D, the bnd dissociation energy, 60.4 kcal/mol, <strong>and</strong> the heat of formation, -13.5 kcal/mol. The CLF is an energetic fluorinating agent. It reacts <strong>with</strong> fluorides such as CsF or NOF to give Cs+CLF2- <strong>and</strong> NO+ClF2-, respectively, <strong>and</strong> has been reported to react <strong>with</strong> the Lewis acid AsF5 to give Cl+AsF6- but substantiating evidence is lacking. The high volatility of CLF (b.p. -1OOOC) suggests that little or no association or self-ionization (to Clf <strong>and</strong> CUP- ions or to C12F' <strong>and</strong> CLF2- ions) exists, but the electrical conductivity is higher than that of CLF3.' c12. Chlorine monofluoride is prepared by reaction of Cu3 ana ,
B I 3 m4*2 elect. I 11 115. Figure 3 - Interconversion of Nitrogen Fluorides
- Page 1 and 2:
Introduction 1. SECONDARY CELLS WIT
- Page 3 and 4:
I time curves at constant current d
- Page 5 and 6:
I 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11
- Page 7 and 8:
7. IV IV- Equivalent Weight, gr/ Eq
- Page 9 and 10:
1 3 9 SOYO FILLER,HQT PRESS Fig. 4.
- Page 11 and 12:
11. COAL PYROLYSIS USING LASER IRRA
- Page 13 and 14:
13. Macerals. Macerals from a singl
- Page 15 and 16:
i 1 I Photochemistry. A fundamental
- Page 17 and 18:
t P li. al ’ i ._ m LL
- Page 19 and 20:
' 19. PYROLYSIS OF COAL IN A MICROW
- Page 21 and 22:
I 21. In the third stage, the gas e
- Page 23 and 24:
.4 4 0 W 0 m .d m x .-( 0 x w M m s
- Page 25 and 26:
' 25. CONCLUSIONS The principal rea
- Page 28 and 29:
' .4 b s tract 28.
- Page 30 and 31:
30. the course of the experiment Ex
- Page 32 and 33:
d (Sulfur] dt m i trogenj dt 32. E
- Page 34 and 35:
34. Table 1, Properties of Feed Mat
- Page 36 and 37:
0 0 0 m 0 VI b N 0 c, VI N 0 v, N h
- Page 38 and 39:
- Literature Cited 38 1. Gordon, K
- Page 40 and 41:
40 z - B 30 w 6 20 yl w U 10 40. 10
- Page 42 and 43:
Z 0 In 80 CK W > 6 60- 0 I- 40- Z W
- Page 44 and 45:
44. 2.0 I 1.2 - 0 2 1.0- 0.8 i TIME
- Page 46 and 47:
- 2.81 1.NITROGEN 2. SULFUR 3. GASO
- Page 48 and 49:
48. The oil from the separator is v
- Page 50 and 51:
. 50 . Table I . Properties of Pitc
- Page 52 and 53:
52. Coke yield A - - - - 0 800 900
- Page 54 and 55:
FIGURE 8. 54. t 0.5 1 800 900 1,000
- Page 56 and 57:
Introduction 56. FLUORODINITROETUNO
- Page 58 and 59:
chloride extractant without other h
- Page 60 and 61:
60. identified (Reference 7) as the
- Page 62 and 63:
62. to FEFO -e quite high (80 to 85
- Page 64 and 65: 64. RECENT CHEMISTRY OF THE OXYGEN
- Page 66 and 67: polymers for the conventional fuel
- Page 68 and 69: 68. In summary, two general methods
- Page 70 and 71: 70. Table XI1 Differential Thermal
- Page 72 and 73: vapor Pressure (psia) Figure 4. Vap
- Page 74 and 75: C H -0-C-NHF, - 2 5 II 0 74. H 9304
- Page 76 and 77: 76. The infrared spectrum is descri
- Page 78 and 79: , 78. PREPARATION AND POLYMERIZATIO
- Page 80 and 81: . .- . - ..... . . I ,caving the re
- Page 82 and 83: If it ~ 3 ~ o~t 7 s Y'2t the therm1
- Page 84 and 85: Chlorine Fentafluaride T q D OC 252
- Page 86 and 87: , 86. Zeections of Cl30 and AsF5. M
- Page 88 and 89: aa . - The rrost difficult rctionel
- Page 90 and 91: 90. DENSITY, VISCOSITY AND SURFACE
- Page 92 and 93: 92. If it is assumed that the syste
- Page 94 and 95: 94. After condensation of oxidizer
- Page 96 and 97: Stirring Solenoid LHe 7. Cryostat 9
- Page 98 and 99: Introduction 98. RFACTIONS OF OxYcm
- Page 100 and 101: , Acknowledgement 100. This work wa
- Page 102 and 103: 102. volume and then by pumping to
- Page 104 and 105: 104. The x-ray powder pattern (Tabl
- Page 106 and 107: Irredie tion Time, mi&) 106. Table
- Page 108 and 109: I. Introduction 108. RE7IIEw OF ADV
- Page 110 and 111: 1.40 w F O/) 103-4O 'F 110. /H 0 21
- Page 112 and 113: !P, "c -- -2118.5 -195 -172 -16.1 .
- Page 116 and 117: Compound C102F ~ 1 , 0 ~ -146 ~ 116
- Page 118 and 119: 118. fom such salts as cS+m8- have
- Page 120 and 121: 120. DETONABILITY TESTING AT NONAMB
- Page 122 and 123: I 122. top) is closed with caps whi
- Page 124 and 125: 124. five pieces of MDF, each 20.00
- Page 126 and 127: 126. auxiliary equipment. For conve
- Page 128 and 129: 128. reliability. Details of most o
- Page 130 and 131: 130. The time required for the deto
- Page 132 and 133: 132. Fig. 2 Typical Witness Plate
- Page 134 and 135: I ll 134. L, ALUMINUM 011+ ‘7 I-
- Page 136: W (3 3 a (3 W m 3 0 v) W E k- / W 3